Keywords
$\mathrm{S_N^H{Ar}}$ reaction
3-phenyl-2-propynenitrile
isoquinoline
N-vinylation/C-phosphorylation
pyridinoids
secondary phosphine oxides
Abstract
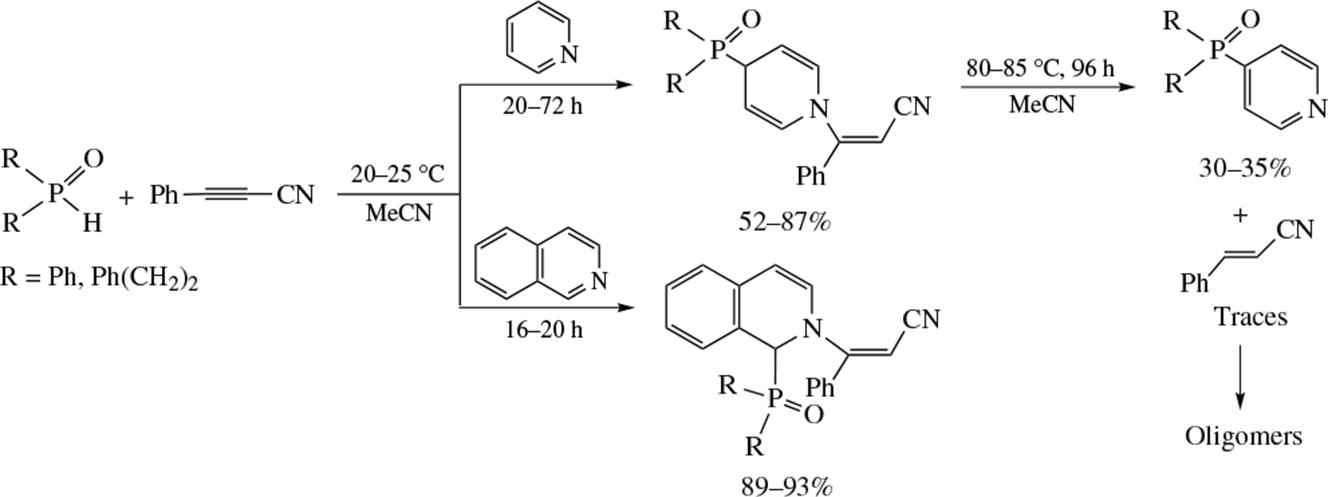
Reactions between 3-phenyl-2-propynenitrile, secondary phosphine oxides and pyridinoids have been implemented and studied. Pyridine and isoquinoline react with propynenitrile and phosphine oxides at room temperature according to the N-vinylation/C-phosphorylation scheme to afford (Z)-N-(2-cyano-1-phenyl)ethenyl phosphoryl-1,4-dihydropyridines or -1,2-dihydroiso quinolines. In the case of pyridine on heating (80-85 °C), the reaction gives 4-phosphorylpyridines (\( \mathrm{S_N^H{Ar}} \) reaction) and 3-phenyl-acrylonitrile oligomers.
References
1.
Arbuzova S.N., Gusarova N.K., Bogdanova M.V., Ivanova N.I., Ushakov I.A., Mal’kina A.G., Trofimov B.A.
Mendeleev Communications,
2005
2.

Trofimov B.A., Volkov P.A., Khrapova K.O., Telezhkin A.A., Ivanova N.I., Albanov A.I., Gusarova N.K., Chupakhin O.N.
Chemical Communications,
2018
3.

Trofimov B.A., Volkov P.A., Khrapova K.O., Telezhkin A.A., Ivanova N.I., Albanov A.I., Gusarova N.K., Belogolova A.M., Trofimov A.B.
Journal of Organic Chemistry,
2019
4.
10.1016/j.mencom.2021.09.026_h0020
Selective Organic Transformations,
1970
5.
10.1016/j.mencom.2021.09.026_h0025
The Chemistry of the Carbon-Carbon Triple Bond,
1978
6.

Volkov P.A., Telezhkin A.A., Khrapova K.O., Ivanova N.I., Albanov A.I., Gusarova N.K., Trofimov B.A.
New Journal of Chemistry,
2021
7.

Gusarova N.K., Volkov P.A., Ivanova N.I., Arbuzova S.N., Khrapova K.O., Albanov A.I., Smirnov V.I., Borodina T.N., Trofimov B.A.
Tetrahedron Letters,
2015
8.
Gusarova N.K., Volkov P.A., Ivanova N.I., Khrapova K.O., Telezhkin A.A., Albanov A.I., Trofimov B.A.
Mendeleev Communications,
2017
9.

Gusarova N.K., Trofimov B.A.
Russian Chemical Reviews,
2020
10.

Gusarova N.K., Volkov P.A., Ivanova N.I., Khrapova K.O., Albanov A.I., Trofimov B.A.
Russian Journal of General Chemistry,
2016
11.

Gusarova N.K., Volkov P.A., Ivanova N.I., Khrapova K.O., Albanov A.I., Afonin A.V., Borodina T.N., Trofimov B.A.
Tetrahedron Letters,
2016
12.

Glotova T.E., Dvorko M.Y., Gusarova N.K., Arbuzova S.N., Ushakov I.A., Kazantseva T.I., Trofimov B.A.
Phosphorus, Sulfur and Silicon and the Related Elements,
2008
13.

Dvorko M.Y., Glotova T.E., Ushakov I.A., Gusarova N.K.
Russian Journal of Organic Chemistry,
2010
14.

Hong S.J., Sung K., Lim S., Kim S., Kim W., Shin J., Park S., Kim H., Rhee M.
Drug Design, Development and Therapy,
2020
15.

Kalar I., Xu H., Secnik J., Schwertner E., Kramberger M.G., Winblad B., Euler M., Eriksdotter M., Garcia‐Ptacek S.
Journal of Internal Medicine,
2020
16.

Li C., Du P., Zhou M., Yang L., Zhang H., Wang J., Yang C.
Chemical and Pharmaceutical Bulletin,
2020
17.
10.1016/j.mencom.2021.09.026_h0075
Ullas
J. Drug Delivery Sci. Technol.,
2020
18.

Selyutina O.Y., Mastova A.V., Shelepova E.A., Polyakov N.E.
Molecules,
2021
19.
10.1016/j.mencom.2021.09.026_h0085
Anwar
Syst. Rev. Pharm.,
2021
20.

Lal C., Garg R., Gupta G.D.
Current Drug Delivery,
2020
21.

Sadaat S.S., Stankzi N., Tawfeeq M.M., Tanin F.A., Aziz A., Sasaki K.
Open Journal of Veterinary Medicine,
2020
22.

Praski Alzrigat L., Huseby D.L., Brandis G., Hughes D.
Journal of Antimicrobial Chemotherapy,
2020
23.

Cheng Y., Chang Y., Ko Y., Liu C.J.
International Journal of Biological Macromolecules,
2021
24.

Alffenaar J.C., Jongedijk E.M., van Winkel C.A., Sariko M., Heysell S.K., Mpagama S., Touw D.J.
Journal of Antimicrobial Chemotherapy,
2020
25.

Guk J., Guedj J., Burdet C., Andremont A., Gunzburg J., Ducher A., Mentré F.
Clinical Pharmacology and Therapeutics,
2020
26.

Alseqely M., Newton-Foot M., Khalil A., El-Nakeeb M., Whitelaw A., Abouelfetouh A.
Scientific Reports,
2021
27.

Wu C., Wei P., Shen Y., Chang H., Tsai Y., Pan H., Chang Y., Wei Y., Yang C.
Antibiotics,
2019
28.

Yıldırım S., Karakoç H.N., Yaşar A., Köksal İ.
Biomedical Chromatography,
2020
29.

Abbas H.S., El Sayed W.A., Fathy N.M.
European Journal of Medicinal Chemistry,
2010
30.

Prasanthi G., Prasad K.V., Bharathi K.
European Journal of Medicinal Chemistry,
2014
31.

Kumar R.S., Idhayadhulla A., Abdul Nasser A.J., Selvin J.
European Journal of Medicinal Chemistry,
2011
32.

Fassihi A., Azadpour Z., Delbari N., Saghaie L., Memarian H.R., Sabet R., Alborzi A., Miri R., Pourabbas B., Mardaneh J.
European Journal of Medicinal Chemistry,
2009
33.

Fleming F.F., Fleming F.F.
Natural Product Reports,
1999
34.
(b) P. Zhan, X. Chen, D. Li, Z. Fang, E. De Clercq and X. Liu, Med. Res. Rev., 2013, 33, E1
35.

Fleming F.F., Yao L., Ravikumar P.C., Funk L., Shook B.C.
Journal of Medicinal Chemistry,
2010
36.

Sergeyev S., Yadav A.K., Franck P., Michiels J., Lewi P., Heeres J., Vanham G., Ariën K.K., Vande Velde C.M., De Winter H., Maes B.U.
Journal of Medicinal Chemistry,
2016