Keywords
3-c]pyridines
3-azaxanthenes
anticancer activity
chromeno[2
Cyclization
cytotoxicity
naphthol
pyridoxal
Abstract
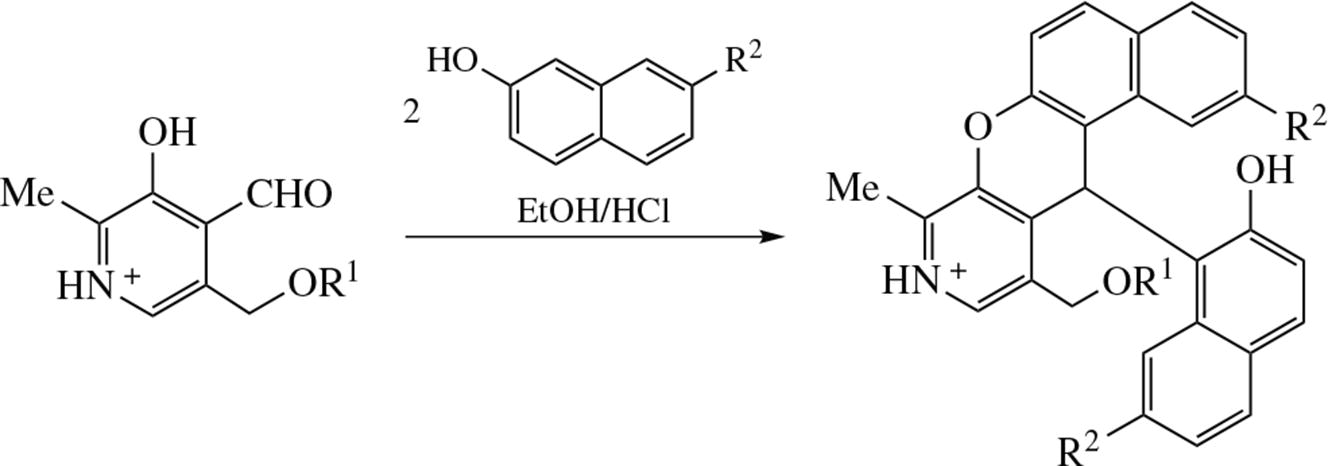
Novel 3-azaxanthenes have been synthesized via acid-catalyzed one-pot Friedel–Crafts reaction of pyridoxal/pyridoxal-5'-phosphate with naphthols followed by dehydration of the intermediate bis(2-hydroxyaryl)-(pyridin-4-yl)methane derivatives. The cytotoxicity of the obtained 3-azaxanthenes against M-HeLa and HuTu-80 tumor cell lines is comparable to that of tamoxifen, whereas the cytotoxicity against normal cell line is absent in the tested concentrations range, which makes this class of compounds interesting for further studies.
References
1.

Maia M., Resende D.I., Durães F., Pinto M.M., Sousa E.
European Journal of Medicinal Chemistry,
2021
2.

Chaudhary A., Khurana J.M.
Current Organic Synthesis,
2017
3.

Rarig R.F., Tran M.N., Chenoweth D.M.
Journal of the American Chemical Society,
2013
4.

Pavia M.R., Taylor C.P., Hershenson F.M., Lobbestael S.J., Butler D.E.
Journal of Medicinal Chemistry,
1988
5.

Villani F.J., Mann T.A., Wefer E.A., Hannon J., Larca L.L., Landon M.J., Spivak W., Vashi D., Tozzi S.
Journal of Medicinal Chemistry,
1975
6.
10.1016/j.mencom.2021.09.024_b0030
Cheng
Chem. Lett.,
2015
7.

Chen J.J., Liu Q., Yuan C., Gore V., Lopez P., Ma V., Amegadzie A., Qian W., Judd T.C., Minatti A.E., Brown J., Cheng Y., Xue M., Zhong W., Dineen T.A., et. al.
Bioorganic and Medicinal Chemistry Letters,
2015
8.

Qian W., Brown J., Chen J.J., Cheng Y.
Tetrahedron Letters,
2014
9.

Familoni O., Ionica I., Bower J., Snieckus V.
Synlett,
1997
10.

Kuhakarn C., Surapanich N., Kamtonwong S., Pohmakotr M., Reutrakul V.
European Journal of Organic Chemistry,
2011
11.

Szatmári I., Belasri K., Heydenreich M., Koch A., Kleinpeter E., Fülöp F.
ChemistryOpen,
2019
12.

Resende D.I., Almeida M.C., Maciel B., Carmo H., Sousa Lobo J., Dal Pozzo C., Cravo S.M., Rosa G.P., Kane-Pagès A., do Carmo Barreto M., Almeida I.F., de Sousa M.E., Pinto M.M.
Molecules,
2020
13.

Gazizov A.S., Burilov A.R., Pudovik M.A., Sinyashin O.G.
Russian Chemical Reviews,
2017
14.

Smolobochkin A.V., Gazizov A.S., Burilov A.R., Pudovik M.A., Sinyashin O.G.
Russian Chemical Reviews,
2021
15.
10.1016/j.mencom.2021.09.024_b0075
Kibardina
Synthesis,
2015
16.
Trifonov A.V., Kibardina L.K., Dobrynin A.B., Akhunov A.A., Pudovik M.A., Burilov A.R.
Mendeleev Communications,
2020
17.
A. D. Voloshina, V. E. Semenov, A. S. Strobykina, N. V. Kulik, E. S. Krylova, V. V. Zobov and V. S. Reznik, Russ. J. Bioorg. Chem., 2017, 43, 170 (Bioorg. Khim., 2017, 43, 197).
18.
10.1016/j.mencom.2021.09.024_b0090
Sheldrick
SADABS,
1997
19.

Altomare A., Cascarano G., Giacovazzo C., Viterbo D.
Acta Crystallographica Section A Foundations of Crystallography,
1991
20.
G. M. Sheldrick, SHELX-97, Programs for Crystal Structure Analysis (Release 97-2), University of Göttingen, 1997.
21.

Farrugia L.J.
Journal of Applied Crystallography,
1999
22.
APEX2 (Version 2.1), SAINTPlus, Data Reduction and Correction Program (Version 7.31A), Bruker AXS, Madison, WI, 2006.