Keywords
in vitro studies
anticancer agents
antitumor activity
etoposide
N-heterocyclic compounds
natural products
phaeosphaeride A
sulfides
Abstract
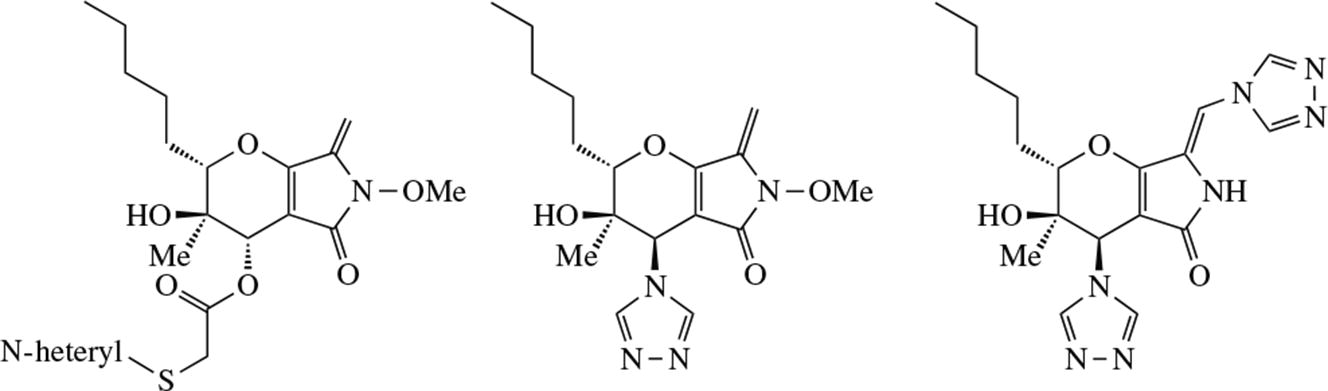
New C(6)-derivatives of natural phaeosphaeride A (PPA) modified with pharmacophoric nitrogen heterocyclic groups have been synthesized. The reaction of 1,2,4-triazole with PPA mesylate produced both the product of the expected substitution of the methanesulfonate group and the PPA derivative with two 1,2,4-triazole groups: at the C(6) atom and the exocyclic C=C bond. The synthesized compounds, with the exception of those containing two 1,2,4-triazole groups, are superior in cytotoxic activity to the original phaeosphaeride and the positive control, etoposide.
References
1.
Bhavana V., Sudharshan S.J., Madhu D.
2017
2.

Cragg G.M., Newman D.J.
Journal of Ethnopharmacology,
2005
3.

Cragg G.M., Newman D.J.
Biochimica et Biophysica Acta - General Subjects,
2013
4.

Newman D.J., Cragg G.M.
Journal of Natural Products,
2016
5.

Maloney K.N., Hao W., Xu J., Gibbons J., Hucul J., Roll D., Brady S.F., Schroeder F.C., Clardy J.
Organic Letters,
2006
6.
A. O. Berestetskiy, E. V. Poluektova, S. V. Sokornova and Yu. S. Tokarev, Patent RF 2596928, 2016.
7.

Abzianidze V.V., Prokofieva D.S., Chisty L.A., Bolshakova K.P., Berestetskiy A.O., Panikorovskii T.L., Bogachenkov A.S., Holder A.A.
Bioorganic and Medicinal Chemistry Letters,
2015
8.
Abzianidze V.V., Bolshakova K.P., Prokofieva D.S., Berestetskiy A.O., Kuznetsov V.A., Trishin Y.G.
Mendeleev Communications,
2017
9.
Abzianidze V.V., Efimova K.P., Poluektova E.V., Trishin Y.G., Kuznetsov V.A.
Mendeleev Communications,
2017
10.

Abzianidze V., Beltyukov P., Zakharenkova S., Moiseeva N., Mejia J., Holder A., Trishin Y., Berestetskiy A., Kuznetsov V.
Molecules,
2018
11.

Abzianidze V.V., Zakharenkova S.A., Moiseeva N.I., Beltyukov P.P., Polukeev V.A., Dubrovskii Y.A., Kuznetsov V.A., Trishin Y.G., Mejia J.E., Holder A.A.
Bioorganic and Medicinal Chemistry Letters,
2019
12.

Ferreira P.M., Maia H.L., Monteiro L.S., Sacramento J.
Tetrahedron Letters,
2000
13.

Abdel-Aziz H.A., Ghabbour H.A., Eldehna W.M., Al-Rashood S.T., Al-Rashood K.A., Fun H., Al-Tahhan M., Al-Dhfyan A.
European Journal of Medicinal Chemistry,
2015
14.

Turky A., Bayoumi A.H., Sherbiny F.F., El-Adl K., Abulkhair H.S.
Molecular Diversity,
2020