Keywords
alcohols
donor–acceptor cyclopropanes
imidazolones
nucleophilic ring opening
spirocyclic compounds
Abstract
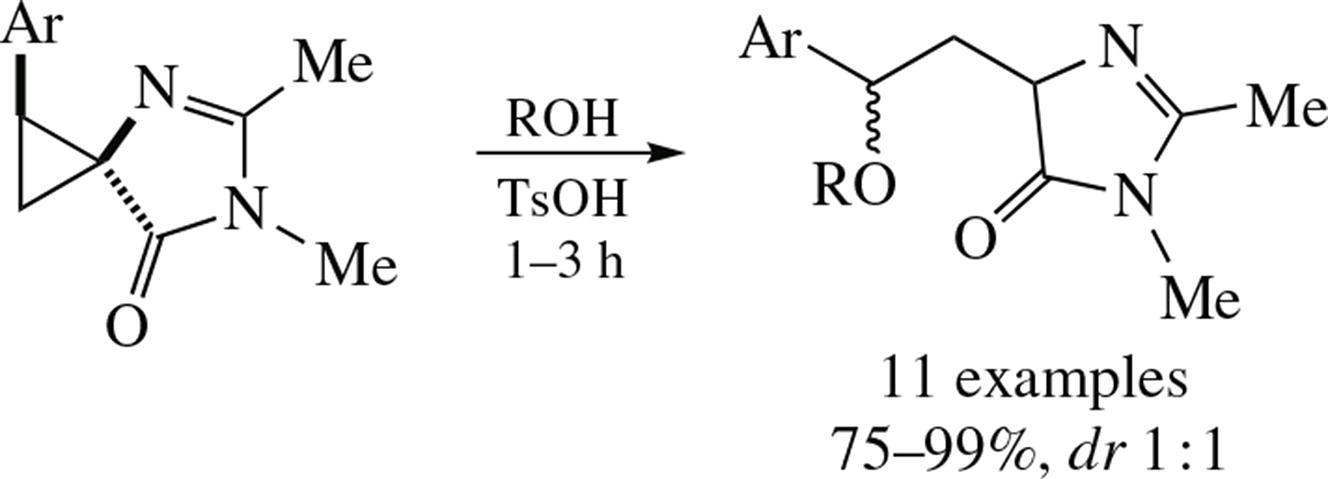
Imidazolone-activated donor–acceptor cyclopropanes undergo alcohol-assisted ring opening under the co-action of p-toluenesulfonic acid. Under the optimized conditions, cyclopropanes and alcohols are coupled in 1,3-fashion with the retention of heterocyclic fragment. Substrates with aromatic donor groups provide the addition products in 75–99% yields as mixtures of two diastereomers.
References
1.

Reissig H., Zimmer R.
Chemical Reviews,
2003
2.

Schneider T.F., Kaschel J., Werz D.B.
Angewandte Chemie - International Edition,
2014
3.

Craig A.J., Hawkins B.C.
Synthesis,
2019
4.

Pirenne V., Muriel B., Waser J.
Chemical Reviews,
2020
5.

Xia Y., Liu X., Feng X.
Angewandte Chemie - International Edition,
2020
6.

Akaev A.A., Melnikov M.Y., Budynina E.M.
Organic Letters,
2019
7.

Borisova I.A., Ratova D.V., Potapov K.V., Tarasova A.V., Novikov R.A., Tomilov Y.V.
Journal of Organic Chemistry,
2021
8.

Budynina E.M., Ivanov K.L., Tukhtaev H.B., Tukhtaeva F.O., Bezzubov S.I., Melnikov M.Y.
Synthesis,
2020
9.

Augustin A.U., Merz J.L., Jones P.G., Mlostoń G., Werz D.B.
Organic Letters,
2019
10.

Petzold M., Jones P.G., Werz D.B.
Angewandte Chemie - International Edition,
2019
11.
10.1016/j.mencom.2021.09.021_b0055
Li
Org. Lett.,
1903
12.

Werz D.B., Biju A.T.
Angewandte Chemie - International Edition,
2019
13.

Andreev I.A., Ratmanova N.K., Augustin A.U., Ivanova O.A., Levina I.I., Khrustalev V.N., Werz D.B., Trushkov I.V.
Angewandte Chemie - International Edition,
2021
14.

Novikov R.A., Denisov D.A., Potapov K.V., Tkachev Y.V., Shulishov E.V., Tomilov Y.V.
Journal of the American Chemical Society,
2018
15.

Suleymanov A.A., Le Du E., Dong Z., Muriel B., Scopelliti R., Fadaei-Tirani F., Waser J., Severin K.
Organic Letters,
2020
16.
10.1016/j.mencom.2021.09.021_b0080
Werz
Angew. Chem., Int. Ed.,
2021
17.

Mikhaylov A.A., Kuleshov A.V., Solyev P.N., Korlyukov A.A., Dorovatovskii P.V., Mineev K.S., Baranov M.S.
Organic Letters,
2020
18.

Mikhaylov A.А., Solyev P.N., Kuleshov A.V., Kublitskii V.S., Korlyukov A.А., Lushpa V.А., Baranov M.S.
Chemistry of Heterocyclic Compounds,
2020
19.

King S.W., Riordan J.M., Holt E.M., Stammer C.H.
Journal of Organic Chemistry,
1982
20.

Hines J.W., Breitholle E.G., Sato M., Stammer C.H.
Journal of Organic Chemistry,
1976
21.

Arenal I., Bernabé M., Fernández-Alvarez E., Penadés S.
Synthesis,
1985
22.

Arenal I., Bernabé M., Alvarez E.F., Izquierdo M.L., Penadés S.
Journal of Heterocyclic Chemistry,
1983
23.

Zhu C., Yang L., Li S., Zheng Y., Ma J.
Organic Letters,
2015
24.

Kreft A., Lücht A., Grunenberg J., Jones P.G., Werz D.B.
Angewandte Chemie - International Edition,
2019