Keywords
1-amino-2-alkynylcyclopropanes
alkylation
alkynyl(chloro)cyclopropanes
cyclopropenes
cyclopropylamines
directing groups
lithiation
protecting groups
stereoselectivity
Abstract
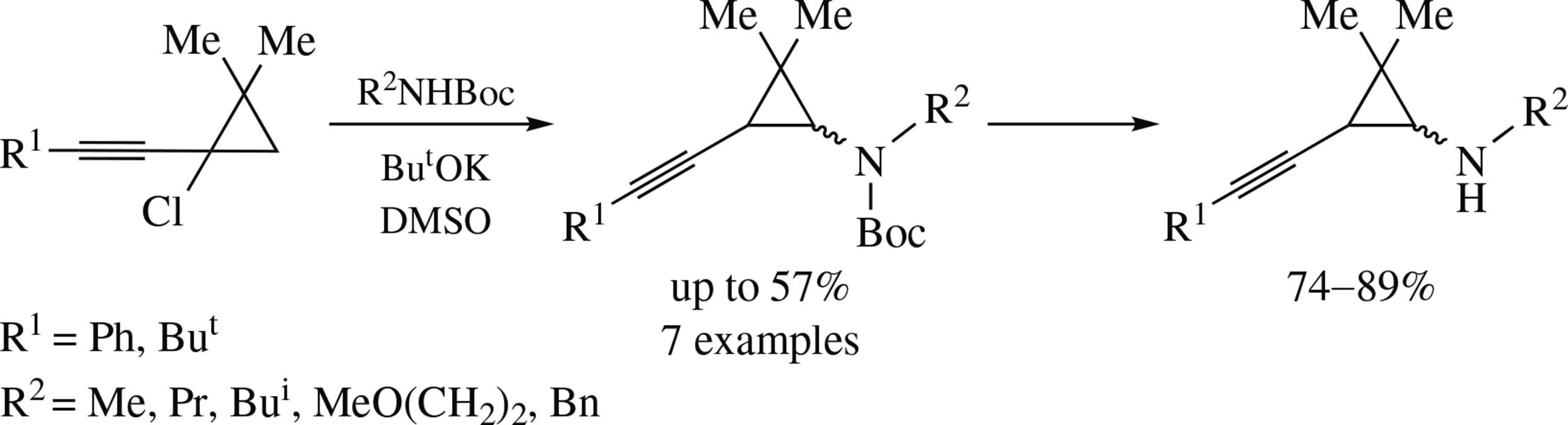
New N-Boc-alkyl(2-alkynylcyclopropyl)amines were synthesized from 1-alkynyl-1-chlorocyclopropanes and N-Boc- alkylamines under the action of ButOK in DMSO, the intermediates having been the corresponding conjugated alkynylcyclopropenes. The Boc-derivatives can be converted into free secondary 2-alkynylcyclopropylamines, as well as β-lithiated with subsequent alkylation.
References
1.
(a) K. N. Shavrin, V. D. Gvozdev and O. M. Nefedov, Russ. Chem. Bull., Int. Ed., 2010, 59, 1451 (Izv. Akad. Nauk, Ser. Khim., 2010, 1418); (b) E. M. Otero, J. M. Fernández-García, M. A. Fernández-Rodríguez and E. Aguilar, Tetrahedron Lett., 2015, 56, 195; (c) H. Chen, J. Zhang and D. Z. Wang, Org. Lett., 2015, 17, 2098; (d) W. Yuan, X. Tang, Y. Wei and M. Shi, Chem. - Eur. J., 2014, 20, 3198; (e) D. Pan, Y. Wei and M. Shi, Org. Lett., 2016, 18, 3930; (f) Y. Zhang, Y. Xiao and J. Zhang, Synthesis, 2016, 48, 512; (g) J. E. C. Tejeda, B. K. Landschoot and M. A. Kerr, Org. Lett., 2016, 18, 2142; (h) A. Urbanaité and I. Čikotiené, Eur. J. Org. Chem., 2016, 5294; (i) J. M. Fernández-García, H. A. Garro, L. Fernández-García, P. García-García, M. A. Fernández-Rodríguez, I. Merino and E. Aguilar, Adv. Synth. Catal., 2017, 359, 3035; (j) D. Pan, Y. Wei and M. Shi, Org. Lett., 2017, 19, 3584; (k) V. D. Gvozdev, K. N. Shavrin, A. A. Ageshina and O. M. Nefedov, Russ. Chem. Bull., Int. Ed., 2018, 67, 1862 (Izv. Akad. Nauk, Ser. Khim., 2018, 1862); (l) V. D. Gvozdev, K. N. Shavrin and O. M. Nefedov, Russ. Chem. Bull., Int. Ed., 2019, 68, 1384 (Izv. Akad. Nauk, Ser. Khim., 2019, 1384); (m) M. S. Garre, D. Sucunza, E. Aguilar, P. García-García and J. J. Vaquero, J. Org. Chem., 2019, 84, 5712; (n) W. Zang, L. Wang, Y. Wei, M. Shi and Y. Guo, Adv. Synth. Catal., 2019, 361, 2321.
2.

Zampella A., D'Auria M.V., Minale L., Debitus C., Roussakis C.
Journal of the American Chemical Society,
1996
3.
10.1016/j.mencom.2021.09.020_h0015
Tedford
J. Pharmacol. Exp. Ther.,
1999
4.
10.1016/j.mencom.2021.09.020_h0020
Corbett
J. Med. Chem.,
2019
5.
(a) J. Salaün, Top. Curr. Chem., 2000, 207, 1
6.

Salaon J., Baird M.S.
Current Medicinal Chemistry,
2022
7.

Gnad F., Reiser O.
Chemical Reviews,
2003
8.

H. Stammer C.
Tetrahedron,
1990
9.

Burgess K., Ho K., Moye-Sherman D.
Synlett,
1994
10.

Sokolova O.O., Bower J.F.
Chemical Reviews,
2020
11.

Liu J., An Y., Jiang H., Chen Z.
Tetrahedron Letters,
2008
12.

de Meijere A., Kozhushkov S., Wagner-Gillen K., Khlebnikov A.
Synthesis,
2010
13.

Mons E., Jansen I.D., Loboda J., van Doodewaerd B.R., Hermans J., Verdoes M., van Boeckel C.A., van Veelen P.A., Turk B., Turk D., Ovaa H.
Journal of the American Chemical Society,
2019
14.

Panchaud P., Surivet J., Diethelm S., Blumstein A., Gauvin J., Jacob L., Masse F., Mathieu G., Mirre A., Schmitt C., Enderlin-Paput M., Lange R., Gnerre C., Seeland S., Herrmann C., et. al.
Journal of Medicinal Chemistry,
2019
15.
L. Blatt, L. Pan, S. Seiwert, S. Andrews, P. Martin, A. Schumacher, L. Beigelman, J. Liu, K. Condroski, Y. Jiang, R. Kaus, A. Kennedy, T. Kercher, M. Lyon and B. Wang, Patent WO 2008/137779 A2, 2008.
16.
Shavrin K.N., Gvozdev V.D., Budanov D.V., Yurov S.V., Nefedov O.M.
Mendeleev Communications,
2006
17.
K. N. Shavrin, V. D. Gvozdev and O. M. Nefedov, Russ. Chem. Bull., Int. Ed., 2010, 59, 396 (Izv. Akad. Nauk, Ser. Khim., 2010, 388).
18.
Shavrin K.N., Gvozdev V.D., Yurov S.V., Nefedov O.M.
Mendeleev Communications,
2008
19.

Park Y.S., Beak P.
Tetrahedron,
1996
20.
K. N. Shavrin, V. D. Gvozdev and O. M. Nefedov, Russ. Chem. Bull., Int. Ed., 2008, 57, 2117 (Izv. Akad. Nauk, Ser. Khim., 2008, 2078).