Keywords
allylic substitution
asymmetric catalysis
homogeneous catalysis
P,S-ligands
Palladium complexes
phosphoramidites
Abstract
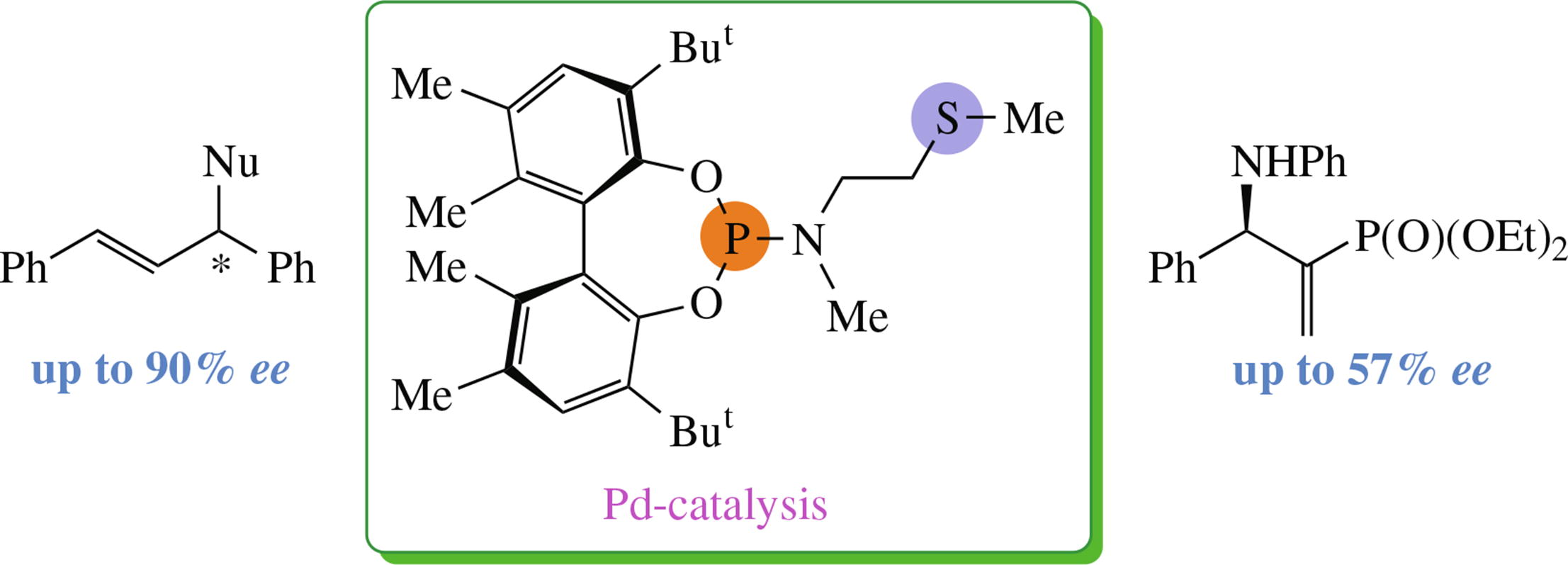
The reaction of P,S-phosphoramidite bearing (Ra)-BIPHEN H2 core and exocyclic amino sulfide as the new ligand with [Pd(ppp-allyl)Cl]2 in the presence of AgBF4 yielded a cationic metallochelate [Pd(ppp-allyl)(L)]BF4. This new chiral inducer provided up to 90% ee in the Pd-mediated allylic substitution reaction of (E)-1,3-diphenylallyl acetate with various C- and N-nucleophiles. In the Pd-catalyzed amination of 2-diethoxyphosphoryl-1-phenylallyl acetate with aniline, ee values up to 57% were achieved.
References
1.

Hua Z., Vassar V.C., Choi H., Ojima I.
Proceedings of the National Academy of Sciences of the United States of America,
2004
2.

Chapsal B.D., Hua Z., Ojima I.
Tetrahedron Asymmetry,
2006
3.

Shi C., Ojima I.
Tetrahedron,
2007
4.

Giacomina F., Meetsma A., Panella L., Lefort L., de Vries A. ., de Vries J.
Angewandte Chemie - International Edition,
2007
5.

Harada H., Thalji R.K., Bergman R.G., Ellman J.A.
Journal of Organic Chemistry,
2008
6.

Costin S., Rath N., Bauer E.
Advanced Synthesis and Catalysis,
2008
7.
![Total Synthesis of Indolizidine Alkaloid (−)-209D: Overriding Substrate Bias in the Asymmetric Rhodium-Catalyzed [2+2+2] Cycloaddition](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Yu R., Lee E., Malik G., Rovis T.
Angewandte Chemie - International Edition,
2009
8.

Zhang X., Cao B., Yan Y., Yu S., Ji B., Zhang X.
Chemistry - A European Journal,
2009
9.

Chien C., Shi C., Lin C., Ojima I.
Tetrahedron,
2011
10.
10.1016/j.mencom.2021.09.019_h0050
Athan
in Kirk-Othmer Encyclopedia of Chemical Technology, ed,
2013
11.

Chen L., Huang J., Xu Z., Zheng Z., Yang K., Cui Y., Cao J., Xu L.
RSC Advances,
2016
12.
![(S)-2-[(N-arylamino)methyl]pyrrolidines-Based Phosphoramidite P,N-Ligand Library for Asymmetric Metal-Catalyzed Allylic Substitution and Conjugate 1,4-Addition](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Gavrilov K.N., Mikhel I.S., Chuchelkin I.V., Zheglov S.V., Gavrilov V.K., Birin K.P., Tafeenko V.A., Chernyshev V.V., Goulioukina N.S., Beletskaya I.P.
ChemistrySelect,
2016
13.

Feng B., Pu X., Liu Z., Xiao W., Chen J.
Organic Chemistry Frontiers,
2016
14.
![P,S Ligands for the Asymmetric Construction of Quaternary Stereocenters in Palladium-Catalyzed Decarboxylative [4+2] Cycloadditions](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Wei Y., Lu L., Li T., Feng B., Wang Q., Xiao W., Alper H.
Angewandte Chemie - International Edition,
2016
15.
![Sequential Visible-Light Photoactivation and Palladium Catalysis Enabling Enantioselective [4+2] Cycloadditions](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Li M., Wei Y., Liu J., Chen H., Lu L., Xiao W.
Journal of the American Chemical Society,
2017
16.

Feng B., Chen J., Yang Y., Lu B., Xiao W.
Chemistry - A European Journal,
2018
17.
![Palladium‐Catalyzed Asymmetric [8+2] Dipolar Cycloadditions of Vinyl Carbamates and Photogenerated Ketenes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Zhang Q., Xiong Q., Li M., Xiong W., Shi B., Lan Y., Lu L., Xiao W.
Angewandte Chemie - International Edition,
2020
18.

Pellissier H.
Tetrahedron,
2007
19.

Mellah M., Voituriez A., Schulz E.
Chemical Reviews,
2007
20.

Pavlov V.A.
Tetrahedron,
2008
21.

Lam F.L., Kwong F.Y., Chan A.S.
Chemical Communications,
2010
22.

Bayardon J., Maronnat M., Langlois A., Rousselin Y., Harvey P.D., Jugé S.
Organometallics,
2015
23.

Berthelot-Bréhier A., Panossian A., Colobert F., Leroux F.R.
Organic Chemistry Frontiers,
2015
24.

Margalef J., Borràs C., Alegre S., Pàmies O., Diéguez M.
Dalton Transactions,
2019
25.

Margalef J., Pàmies O., Pericàs M.A., Diéguez M.
Chemical Communications,
2020
26.
Z. Lu and S. Ma, Angew. Chem., Int. Ed., 2008, 47, 258; (b) M. Diéguez and O. Pàmies, Acc. Chem. Res., 2010, 43, 312; (c) J. F. Teichert and B. L. Feringa, Angew. Chem., Int. Ed., 2010, 49, 2486; (d) Privileged Chiral Ligands and Catalysts, ed. Q.-L. Zhou, Wiley-VCH, Weinheim, 2011; (e) H. Fernández-Pérez, P. Etayo, A. Panossian and A. Vidal-Ferran, Chem. Rev., 2011, 111, 2119; (f) I. G. Rios, A. Rosas-Hernandez and E. Martin, Molecules, 2011, 16, 970; (g) T. Nemoto and Y. Hamada, Tetrahedron, 2011, 67, 667; (h) Transition Metal Catalyzed Enantioselective Allylic Substitution in Organic Synthesis, ed. U. Kazmaier, Springer, Berlin, Heidelberg, 2012; (i) J. Kraft, M. Golkowski and T. Ziegler, Beilstein J. Org. Chem., 2016, 12, 166; (j) M. Biosca, J. Margalef, X. Caldentey, M. Besora, C. Rodríguez-Escrich, J. Saltó, X. Cambeiro, F. Maseras, O. Pàmies, M. Diéguez and M. A. Pericàs, ACS Catal., 2018, 8, 3587; (k) S. Noreen, A. F. Zahoor, S. Ahmad, I. Shahzadi, A. Irfan and S. Faiz, Curr. Org. Chem., 2019, 23, 1168.
27.

Lafrance D., Bowles P., Leeman K., Rafka R.
Organic Letters,
2011
28.

Nag S., Batra S.
Tetrahedron,
2011
29.

Chavan S.P., Khairnar L.B., Chavan P.N.
Tetrahedron Letters,
2014
30.

Evans P., Grange R., Clizbe E.
Synthesis,
2016
31.

Wang X., Wang X., Han Z., Wang Z., Ding K.
Organic Chemistry Frontiers,
2017
32.

Hölscher M., Franciò G., Leitner W.
Organometallics,
2004
33.
![Diastereomerism in palladium(II) allyl complexes of P,P-, P,S- and S,S-donor ligands, Ph2P(E)N(R)P(E′)Ph2 [R=CHMe2 or (S)-*CHMePh; E=E′=lone pair or S]: solution behaviour, X-ray crystal structure and catalytic allylic alkylation reactions](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Mandal S.K., Nagana Gowda G.A., Krishnamurthy S.S., Zheng C., Li S., Hosmane N.S.
Journal of Organometallic Chemistry,
2003
34.

Herrmann J., Pregosin P.S., Salzmann R., Albinati A.
Organometallics,
1995
35.

Gavrilov K.N., Zheglov S.V., Gavrilov V.K., Maksimova M.G., Tafeenko V.A., Chernyshev V.V., Birin K.P., Mikhel I.S.
Tetrahedron,
2017
36.

Benetskiy E.B., Bolm C.
Tetrahedron Asymmetry,
2011
37.

Thiesen K.E., Maitra K., Olmstead M.M., Attar S.
Organometallics,
2010
38.
![Enantiomers of dimethyl [(2E)-1,3-diphenylprop-2-en-1-yl]propanedioate resulting from allylic alkylation reaction: Elution order on major high-performance liquid chromatography chiral columns](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ramillien M., Vanthuyne N., Jean M., Gherase D., Giorgi M., Naubron J., Piras P., Roussel C.
Journal of Chromatography A,
2012
39.

Naganawa Y., Abe H., Nishiyama H.
Chemical Communications,
2018
40.

Liu J., Chen G., Xing J., Liao J.
Tetrahedron Asymmetry,
2011
41.
10.1016/j.mencom.2021.09.019_h0205
Chen
Tetrahedron: Asymmetry,
1953
42.

Majdecki M., Jurczak J., Bauer T.
ChemCatChem,
2015