Keywords
alkyl complexes
catalysis
dehydrocoupling
hydrophosphination
organophosphorus compounds
organosilicon compounds
scandium
Abstract
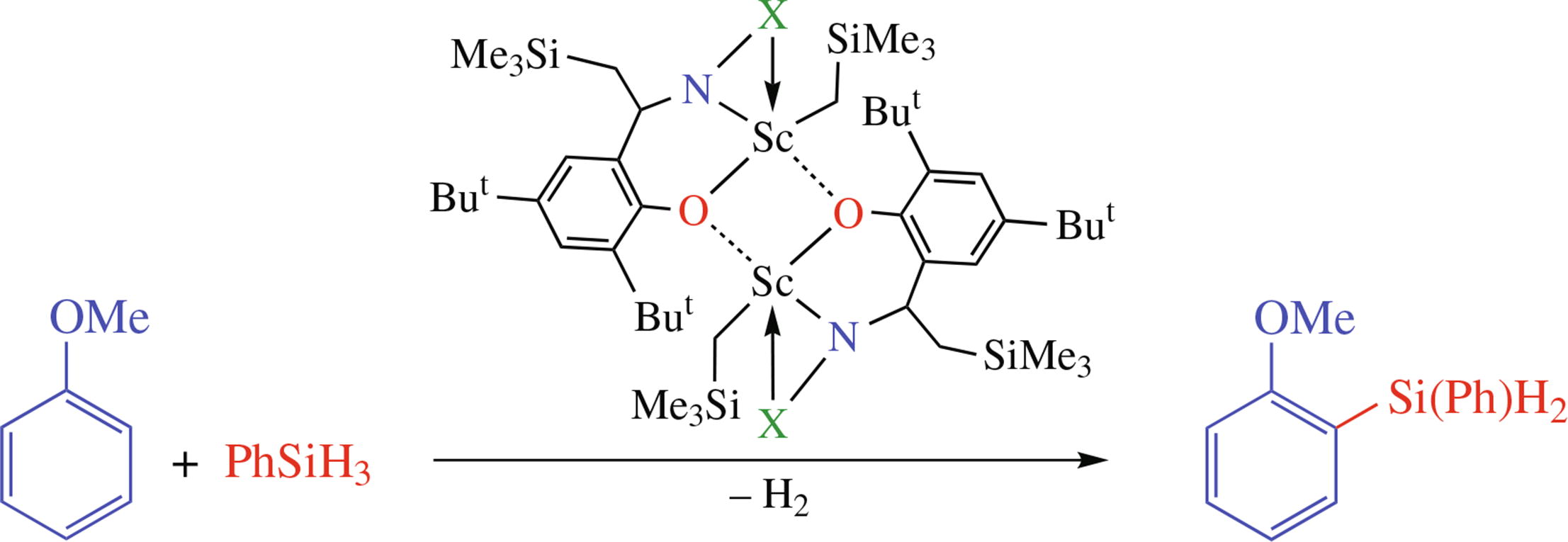
The reactions of imino phenols 3,5-But2-2-HOC6H2CH=NX (X = 8-C9H6N, 2-MeO-5-MeC6H3 and 2-PhOC6H4) with Sc(CH2SiMe3)3(THF)2 in toluene proceed with silane elimination and reductive alkylation of the C=N group affording dimeric base-free monoalkyl scandium complexes. X-ray analysis of the two complexes revealed their dimeric structures due to μ-bridging amidophenolato dianions. The complexes catalyze hydrophosphination of styrene, phenylacetylene and tolane with Ph2PH as well as dehydrogenative coupling of anisole with hydrosilanes.
References
1.

Trifonov A.A.
Coordination Chemistry Reviews,
2010
2.

Pellissier H.
Coordination Chemistry Reviews,
2016
3.

Trifonov A.A., Lyubov D.M.
Coordination Chemistry Reviews,
2017
4.

Zimmermann M., Anwander R.
Chemical Reviews,
2010
5.

Teng H., Luo Y., Wang B., Zhang L., Nishiura M., Hou Z.
Angewandte Chemie - International Edition,
2016
6.
10.1016/j.mencom.2021.09.013_b0030
Arndt
Chem. Rev.,
1953
7.
10.1016/j.mencom.2021.09.013_b0035
Li
Applied Homogeneous Catalysis with Organometallic Compounds,
2017
8.

Guo F., Meng R., Li Y., Hou Z.
Polymer,
2015
9.

Zeimentz P.M., Arndt S., Elvidge B.R., Okuda J.
Chemical Reviews,
2006
10.

Nishiura M., Hou Z.
Nature Chemistry,
2010
11.

Lyubov D.M., Tolpygin A.O., Trifonov A.A.
Coordination Chemistry Reviews,
2019
12.

Ma H., Spaniol T.P., Okuda J.
Angewandte Chemie,
2006
13.

Stanford M.J., Dove A.P.
Chemical Society Reviews,
2010
14.

Nishiura M., Guo F., Hou Z.
Accounts of Chemical Research,
2015
15.

Arnold P.L., McMullon M.W., Rieb J., Kühn F.E.
Angewandte Chemie - International Edition,
2014
16.

Guan B., Hou Z.
Journal of the American Chemical Society,
2011
17.

Song G., O W.W., Hou Z.
Journal of the American Chemical Society,
2014
18.

19.

Zhang J., Xu L., Wong W.
Coordination Chemistry Reviews,
2018
20.

Che C.
Coordination Chemistry Reviews,
2003
21.

Gupta K.C., Sutar A.K.
Coordination Chemistry Reviews,
2008
22.

Das P., Linert W.
Coordination Chemistry Reviews,
2016
23.

Kaczmarek M.T., Zabiszak M., Nowak M., Jastrzab R.
Coordination Chemistry Reviews,
2018
24.

Cozzi P.G.
Chemical Society Reviews,
2004
25.

Segura J.L., Mancheño M.J., Zamora F.
Chemical Society Reviews,
2016
26.

Jia Y., Li J.
Chemical Reviews,
2014
27.

Lara-Sanchez A., Rodriguez A., Hughes D.L., Schormann M., Bochmann M.
Journal of Organometallic Chemistry,
2002
28.

Miao W., Li S., Cui D., Huang B.
Journal of Organometallic Chemistry,
2007
29.

Ren W., Chen L., Zhao N., Wang Q., Hou G., Zi G.
Journal of Organometallic Chemistry,
2014
30.

Qu L., Roisnel T., Cordier M., Yuan D., Yao Y., Zhao B., Kirillov E.
Inorganic Chemistry,
2020
31.

Emslie D.J., Piers W.E., Parvez M.
Dalton Transactions,
2003
32.

Qin D., Han F., Yao Y., Zhang Y., Shen Q.
Dalton Transactions,
2009
33.
10.1016/j.mencom.2021.09.013_b0165
Cui
Sci. Technol.,
2014
34.

Emslie D.J., Piers W.E., Parvez M., McDonald R.
Organometallics,
2002
35.

Han F., Teng Q., Zhang Y., Wang Y., Shen Q.
Inorganic Chemistry,
2011
36.

Meermann C., Törnroos K.W., Anwander R.
Inorganic Chemistry,
2009
37.

Yang Y., Li S., Cui D., Chen X., Jing X.
Organometallics,
2006
38.

Zhang G., Wei Y., Guo L., Zhu X., Wang S., Zhou S., Mu X.
Chemistry - A European Journal,
2014
39.

Allen F.H., Kennard O., Watson D.G., Brammer L., Orpen A.G., Taylor R.
Journal of the Chemical Society Perkin Transactions 2,
1987
40.
10.1016/j.mencom.2021.09.013_b0200
Yacobi
Semiconductor Materials: An Introduction to Basic Principles (Microdevices),
2013
41.
Organosilicon Chemistry VI: From Molecules to Materials, eds. N. Auner and J. Weis, Wiley-VCH, 2008.
42.
10.1016/j.mencom.2021.09.013_b0210
Rösch
Wiley-VCH,
2000
43.

44.

Fujii S., Hashimoto Y.
Future Medicinal Chemistry,
2017
45.

Franz A.K., Wilson S.O.
Journal of Medicinal Chemistry,
2012
46.

Sharma U., Sharma R., Kumar R., Kumar I., Singh B.
Synthesis,
2015
47.

Xu L., Xu Z., Huang W., Zhang J.
Synthesis,
2015
48.

Oyamada J., Nishiura M., Hou Z.
Angewandte Chemie - International Edition,
2011
49.
Bruker APEX3, SAINT, Bruker AXS, Madison, WI, 2018.
50.
CrysAlisPro 1.171.38.46, Rigaku Oxford Diffraction, Rigaku Corporation, Wroclaw, Poland, 2018.
51.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
52.

Krause L., Herbst-Irmer R., Sheldrick G.M., Stalke D.
Journal of Applied Crystallography,
2015