Keywords
crystal structure
heteroleptic complex
iron complex
silver
solid phase thermolysis
Abstract
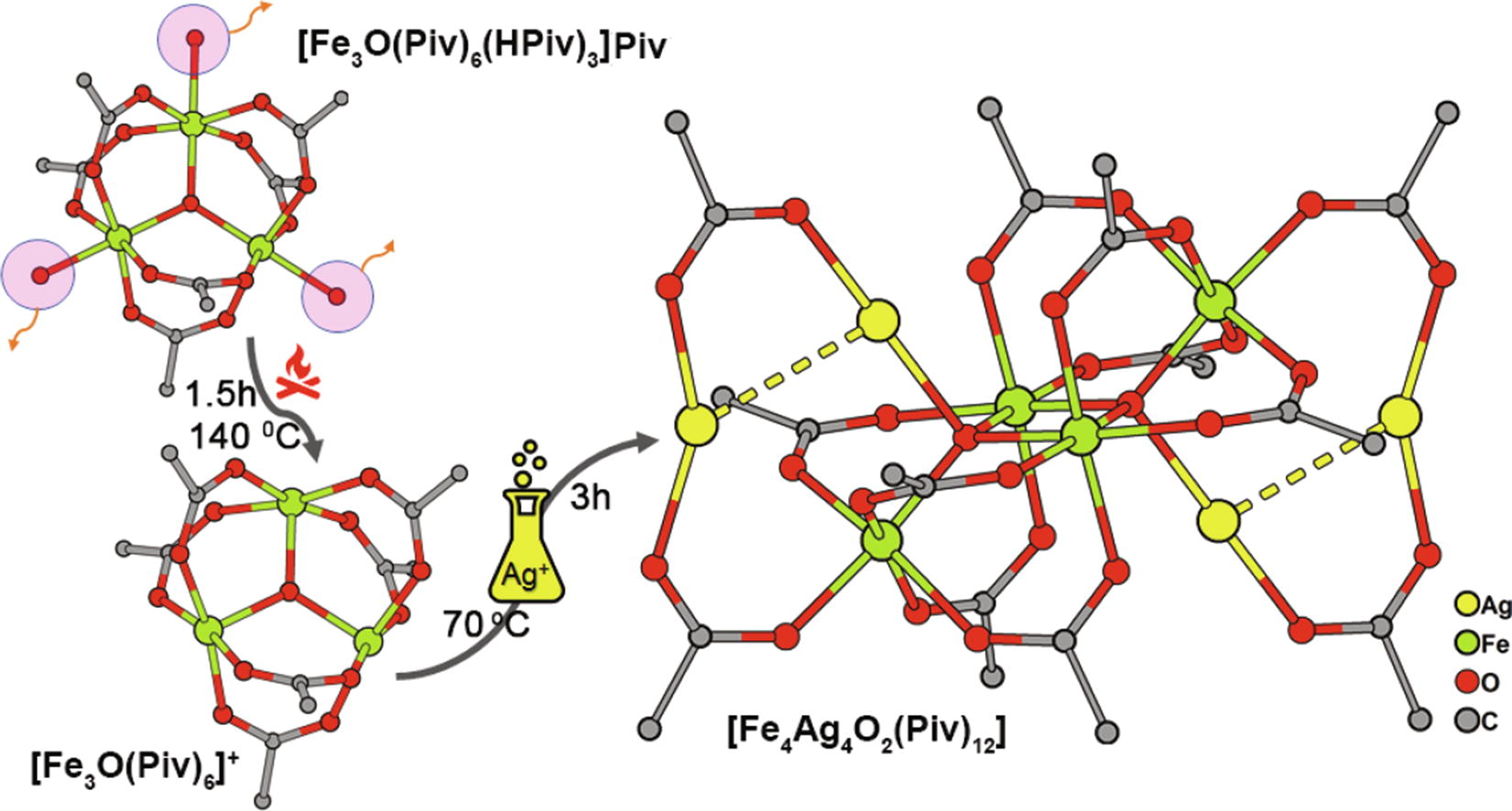
Solid phase thermolysis of pivalate complex [Fe3O(Piv)6(HPiv)3]Piv generates the [Fe3O(Piv)6]+ complex cation due to a deficiency of ligands in the coordination sphere of the metal ions. Crystallization of [Fe3O(Piv)6]+ from THF–EtOH leads to the heteroleptic complex [Fe3O(Piv)6(THF)(EtOH)(OH)] · 0.5 THF · 0.5 H2O in 69% yield, while the reaction of [Fe3O(Piv)6]+ with AgNO3 in toluene results in the complex [Fe4Ag4O2(Piv)12] · 2 PhMe with a rare combination of FeIII and AgI atoms. Crystal structures of the two new complexes have been established.
References
1.
Fu L., Yang Q., Li D., Lu J.Y.
Mendeleev Communications,
2020
2.
Rogovoy M.I., Tomilenko A.V., Samsonenko D.G., Nedolya N.A., Rakhmanova M.I., Artem’ev A.V.
Mendeleev Communications,
2020
3.

Sadovskii M.V.
Physics-Uspekhi,
2008
4.
![Iron-Based Layered Superconductor La[O1-xFx]FeAs (x = 0.05−0.12) with Tc = 26 K](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Kamihara Y., Watanabe T., Hirano M., Hosono H.
Journal of the American Chemical Society,
2008
5.

Rashad M.M., Fouad O.A.
Materials Chemistry and Physics,
2005
6.

Karunananda M.K., Vázquez F.X., Alp E.E., Bi W., Chattopadhyay S., Shibata T., Mankad N.P.
Dalton Transactions,
2014
7.

Veith M., Haas M., Huch V.
Chemistry of Materials,
2004
8.

Godbole R.V., Rao P., Alegaonkar P.S., Bhagwat S.
Materials Chemistry and Physics,
2015
9.

Yeary L.W., Moon J., Rawn C.J., Love L.J., Rondinone A.J., Thompson J.R., Chakoumakos B.C., Phelps T.J.
Journal of Magnetism and Magnetic Materials,
2011
10.

Naseri M.G., Saion E.B., Hashim M., Shaari A.H., Ahangar H.A.
Solid State Communications,
2011
11.
![Electrochemical behaviour of mixed d metal-iron containing Wells-Dawson sandwich-type complexes: [(FeOH2)2M2(X2W15O56)2]− and [(MOH2)2Fe2(X2W15O56)2]− (M = CrIII, MnIII, MnII, CoII, NiII, CuII, ZnII, X = AsV or PV and n = 12 or 14)](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Doungmene F., Aparicio P.A., Ntienoue J., Mezui C.S., de Oliveira P., López X., Mbomekallé I.M.
Electrochimica Acta,
2014
12.
10.1016/j.mencom.2021.09.012_b0060
Long
The Chemistry of Nanostructured Materials,
2003
13.

Abdulwahab K.O., Malik M.A., O’Brien P., Timco G.A., Tuna F., Muryn C.A., Winpenny R.E., Pattrick R.A., Coker V.S., Arenholz E.
Chemistry of Materials,
2013
14.

Baca S.G., Speldrich M., Ellern A., Kögerler P.
Materials,
2011
15.

Baca S.G., Filippova I.G., Keene T.D., Botezat O., Malaestean I.L., Stoeckli‐Evans H., Kravtsov V.C., Chumacov I., Liu S., Decurtins S.
European Journal of Inorganic Chemistry,
2010
16.
![Structure and mixed-valence states of the trinuclear iron(II,III,III) pivalate complex-pivalic acid adduct, [Fe3O(O2CCMe3)6(Me3CCO2H)3] (Me = −CH3)](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Asamaki K., Nakamoto T., Kawata S., Sano H., Katada M., Endo K.
Inorganica Chimica Acta,
1995
17.

Lutsenko I.A., Kiskin M.A., Nelyubina Y.V., Efimov N.N., Maksimov Y.V., Imshennik V.K., Zueva E.M., Goloveshkin A.S., Khoroshilov A.V., Rentschler E., Sidorov A.A., Eremenko I.L.
Polyhedron,
2019
18.

Lutsenko I.A., Kiskin M.A., Imshennik V.K., Maksimov Y.V., Sidorov A.A., Eremenko I.L.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2017
19.

Lutsenko I.A., Kiskin M.A., Efimov N.N., Ugolkova E.A., Maksimov Y.V., Imshennik V.K., Goloveshkin A.S., Khoroshilov A.V., Lytvynenko A.S., Sidorov A.A., Eremenko I.L.
Polyhedron,
2017
20.

Lutsenko I.А., Kiskin М.А., Alexandrov G.G., Imshennik V.K., Maksimov Y.V., Khoroshilov A.V., Goloveshkin A.S., Sidorov А.А., Eremenko I.L.
Russian Chemical Bulletin,
2018
21.
Lutsenko I.A., Kiskin M.A., Nikolaevskii S.A., Nelyubina Y.V., Primakov P.V., Goloveshkin A.S., Imshennik V.K., Maksimov Y.V., Sidorov A.A., Eremenko I.L.
Mendeleev Communications,
2020
22.
![Crystal structures of ethylenediaminetetraacetato metal complexes. V. Structures containing the [Fe(C10H12N2O8)(H2O)]− anion](/storage/images/resized/xG3oC19920mmFVqkoVEGKGaMCeoalzyvWxPkVox5_small_thumb.webp)
Solans X., Font Altaba M., Garcia-Oricain J.
Acta Crystallographica Section C Crystal Structure Communications,
1984
23.

Do L.H., Lippard S.J.
Journal of the American Chemical Society,
2011
24.

Kiskin M.A., Fomina I.G., Sidorov A.A., Aleksandrov G.G., Proshenkina O.Y., Dobrokhotova Z.V., Ikorskii V.N., Shvedenkov Y.G., Novotortsev V.M., Eremenko I.L., Moiseev I.I.
Russian Chemical Bulletin,
2004
25.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
26.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
27.

Çelenligil-Çetin R., Staples R.J., Stavropoulos P.
Inorganic Chemistry,
2000
28.

Groom C.R., Bruno I.J., Lightfoot M.P., Ward S.C.
Acta Crystallographica Section B: Structural Science, Crystal Engineering and Materials,
2016