Keywords
catalysis
ferrocene
N-heterocyclic carbenes
redox active ligands
rhodium
Abstract
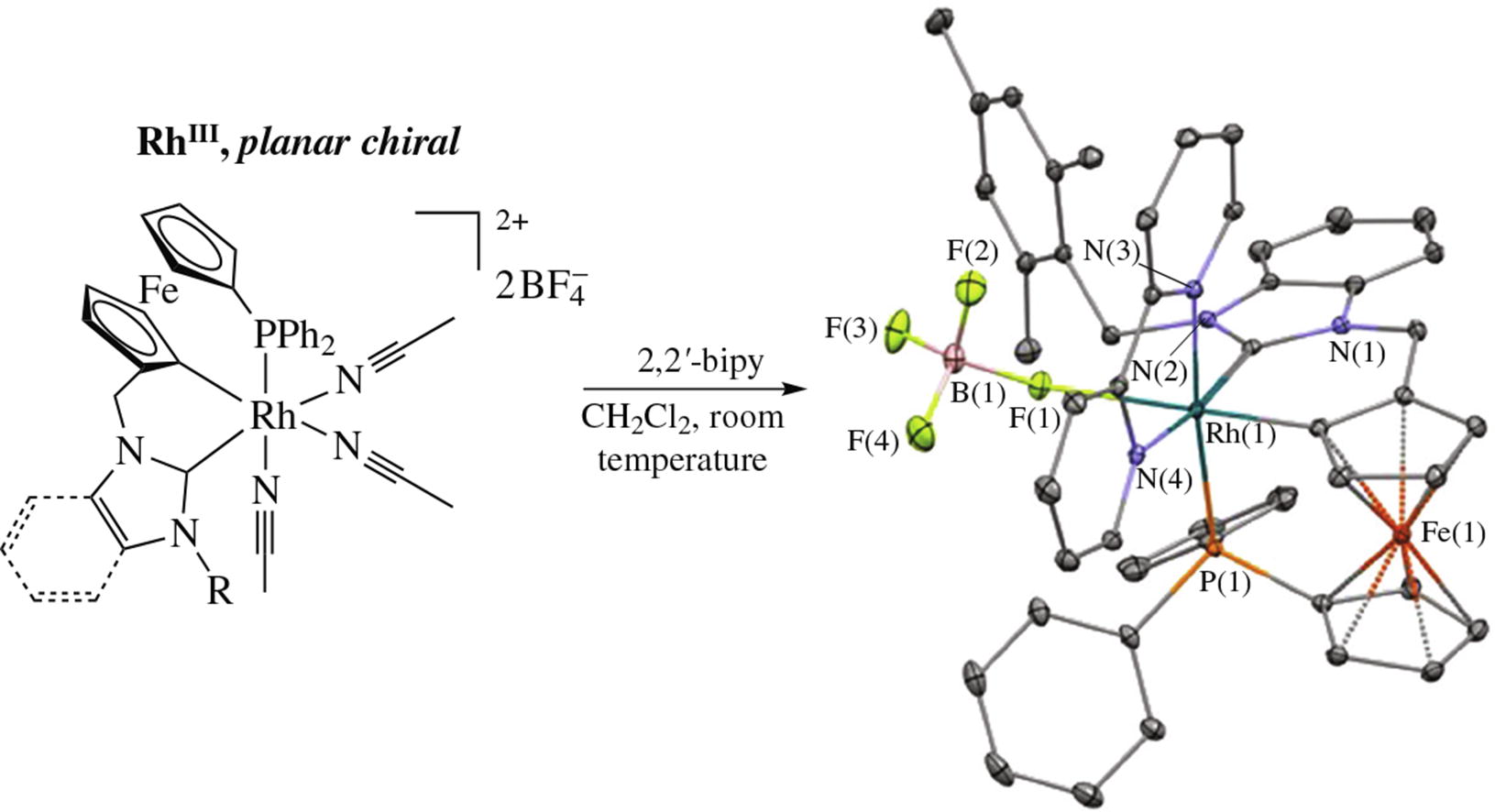
The chemical oxidation of rhodium(i) complexes [Rh(L)(COD)][BF4], where L is a ferrocenyl phosphine/N-heterocyclic carbene ligand, with 2 equiv. of a triaryl-aminium salt [(4-BrC6H4)3N][BF4] in acetonitrile gave planar chiral, air-stable [Rh(L–H)(MeCN)3][BF4]2 complexes where the ferrocene (C5H4CH2ImR or C5H4CH2BImCH2Mes) ring has been C–H activated at the position 2 in good to excellent yields. An important reactivity difference between our complexes and the ubiquitous [Cp*Rh(MeCN)3]X2 complex has been observed in the Grignard-type arylation of 4-nitrobenzaldehyde.
References
1.

Colby D.A., Bergman R.G., Ellman J.A.
Chemical Reviews,
2009
2.

Colby D.A., Tsai A.S., Bergman R.G., Ellman J.A.
Accounts of Chemical Research,
2011
3.

Wencel-Delord J., Dröge T., Liu F., Glorius F.
Chemical Society Reviews,
2011
4.

Kuhl N., Schröder N., Glorius F.
Advanced Synthesis and Catalysis,
2014
5.

Song G., Wang F., Li X.
Chemical Society Reviews,
2012
6.

Song G., Li X.
Accounts of Chemical Research,
2015
7.

Gensch T., James M.J., Dalton T., Glorius F.
Angewandte Chemie - International Edition,
2018
8.

Rej S., Chatani N.
Angewandte Chemie - International Edition,
2019
9.

Wang C., Chen F., Qian P., Cheng J.
Organic and Biomolecular Chemistry,
2021
10.
Kolos A.V., Perekalin D.S.
Mendeleev Communications,
2021
11.

Grushin V.V., Marshall W.J., Thorn D.L.
Advanced Synthesis and Catalysis,
2001
12.

Hyster T.K., Knörr L., Ward T.R., Rovis T.
Science,
2012
13.

Ye B., Cramer N.
Science,
2012
14.

Zheng J., Cui W., Zheng C., You S.
Journal of the American Chemical Society,
2016
15.

Jia Z., Merten C., Gontla R., Daniliuc C.G., Antonchick A.P., Waldmann H.
Angewandte Chemie - International Edition,
2017
16.

Trifonova E.A., Ankudinov N.M., Mikhaylov A.A., Chusov D.A., Nelyubina Y.V., Perekalin D.S.
Angewandte Chemie - International Edition,
2018
17.

Wencel-Delord J., Colobert F.
Chemistry - A European Journal,
2013
18.

Motevalli S., Sokeirik Y., Ghanem A.
European Journal of Organic Chemistry,
2016
19.
10.1016/j.mencom.2021.09.010_b0095
Rhodium Catalysis in Organic Synthesis,
2019
20.

Yoshino T., Satake S., Matsunaga S.
Chemistry - A European Journal,
2020
21.

Labande A., Debono N., Sournia-Saquet A., Daran J., Poli R.
Dalton Transactions,
2013
22.

Debono N., Daran J., Poli R., Labande A.
Polyhedron,
2015
23.

Connelly N.G., Geiger W.E.
Chemical Reviews,
1996
24.
10.1016/j.mencom.2021.09.010_b0120
Gülcemal
Eur. J. Inorg. Chem.,
1806
25.

Labande A., Daran J., Manoury E., Poli R.
European Journal of Inorganic Chemistry,
2007
26.

Mas-Marzá E., Poyatos M., Sanaú M., Peris E.
Organometallics,
2003
27.

Poyatos M., Sanaú M., Peris E.
Inorganic Chemistry,
2003
28.

Feller M., Ben-Ari E., Gupta T., Shimon L.J., Leitus G., Diskin-Posner Y., Weiner L., Milstein D.
Inorganic Chemistry,
2007
29.

Rybtchinski B., Oevers S., Montag M., Vigalok A., Rozenberg H., Martin J.M., Milstein D.
Journal of the American Chemical Society,
2001
30.

Salem H., Ben-David Y., Shimon L.J., Milstein D.
Organometallics,
2006
31.
![Aliphatic and aromatic C–H activation of benzo[h]quinolines by Rh(I). Unique precursor dependent formation of mono-, di- and trinuclear complexes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Salem H., Leitus G., Shimon L.J., Diskin-Posner Y., Milstein D.
Inorganica Chimica Acta,
2011
32.

Wei S., Pedroni J., Meißner A., Lumbroso A., Drexler H., Heller D., Breit B.
Chemistry - A European Journal,
2013
33.

Yang L., Correia C.A., Li C.
Advanced Synthesis and Catalysis,
2011
34.

Chernyshev V.M., Denisova E.A., Eremin D.B., Ananikov V.P.
Chemical Science,
2020
35.

Guimond N., Gorelsky S.I., Fagnou K.
Journal of the American Chemical Society,
2011