Keywords
ab initio computations
bicyclo[3.3.1]nonanes
conformational behaviour
conformational effects
Abstract
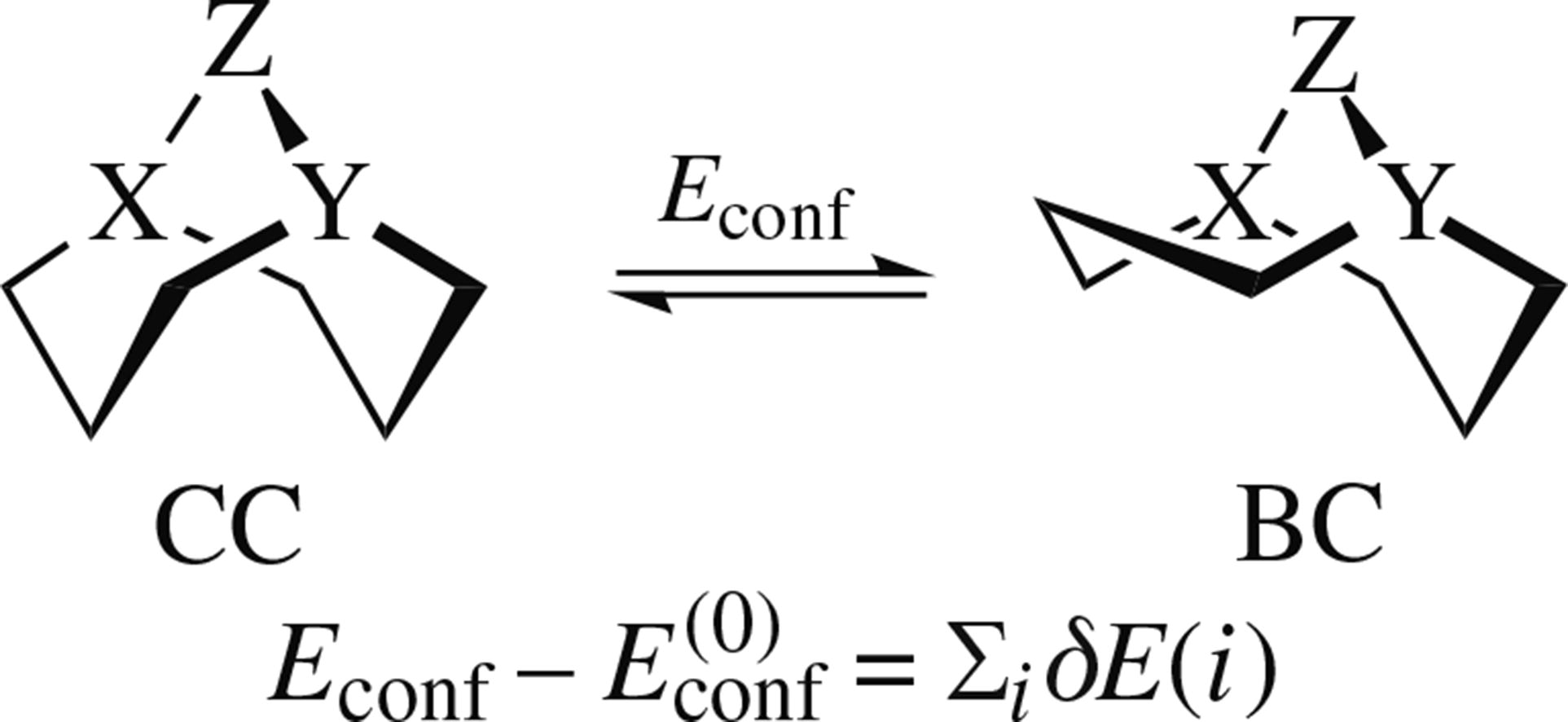
The high-level ab initio calculations on several derivatives of bicyclo[3.3.1]nonane, 1-aza- and 1,5-diazabicyclo[3.3.1]-nonanes show the ‘double chair’ (CC) conformer as optimal for all of them, dominating over the ‘boat–chair’ (BC) form. Conformational effects of several substitution types involving positions 1, 5 and 9 are quantified, and their values are found rather transferable.
References
1.
![Conformational Analysis of Bicyclo [3.3.1] Nonanes and their Hetero Analogs](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Zefirov N.S., Palyulin V.A.
Topics in Stereochemistry,
1991
2.
10.1016/j.mencom.2021.09.007_b0010
Eliel
Stereochemistry of Organic Compounds,
1994
3.
10.1016/j.mencom.2021.09.007_b0015
Eliel
Basic Organic Stereochemistry,
2001
4.
![The “Hockey Sticks” Effect Revisited: The Conformational and Electronic Properties of 3,7-Dithia-1,5-diazabicyclo[3.3.1]nonane from the QTAIM Perspective](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Bushmarinov I.S., Fedyanin I.V., Lyssenko K.A., Lapteva V.L., Pisarev S.A., Palyulin V.A., Zefirov N.S., Antipin M.Y.
Journal of Physical Chemistry A,
2011
5.

Wheeler S.E., Houk K.N., Schleyer P.V., Allen W.D.
Journal of the American Chemical Society,
2009
6.
10.1016/j.mencom.2021.09.007_b0030
Wheeler
Rev.: Comput. Mol. Sci.,
2012
7.
![Intramolecular strain and conformational energy of some symmetric heteroanalogues of bicyclo[3.3.1]nonane](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Pisarev S.A., Palyulin V.A., Zefirov N.S.
Doklady Chemistry,
2013
8.
![Lone pairs vs. covalent bonds: conformational effects in bicyclo[3.3.1]nonane derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Pisarev S.A., Shulga D.A., Palyulin V.A., Zefirov N.S.
Structural Chemistry,
2018
9.

Tajti A., Szalay P.G., Császár A.G., Kállay M., Gauss J., Valeev E.F., Flowers B.A., Vázquez J., Stanton J.F.
Journal of Chemical Physics,
2004
10.

Møller C., Plesset M.S.
Physical Review A,
1934
11.

Weigend F., Häser M.
Theoretical Chemistry Accounts,
1997
12.

Dunning T.H.
Journal of Chemical Physics,
1989
13.
10.1016/j.mencom.2021.09.007_b0065
Neese
Rev.: Comput. Mol. Sci.,
2018
14.

15.
![A skewed bicyclo[3.3.1]nonane. X-ray and neutron diffraction studies of 9-thiabicyclo[3.3.1]nonane-2,6-dione and molecular-mechanics calculations for bicyclo[3.3.1]nonane derivatives](/storage/images/resized/xG3oC19920mmFVqkoVEGKGaMCeoalzyvWxPkVox5_small_thumb.webp)
Bovill M.J., Cox P.J., Flitman H.P., Giu M.H., Hardy A.D., McCabe P.H., Macdonald M.A., Sim G.A., White D.N.
Acta Crystallographica Section B,
1979
16.
![The twin-chair conformation of bicyclo[3.3.1]nonane: X-ray diffraction study of 5-methyl-1-p-toluenesulphonyloxymethylbicyclo[3.3.1]nonan-9-ol](/storage/images/resized/xG3oC19920mmFVqkoVEGKGaMCeoalzyvWxPkVox5_small_thumb.webp)
Sim G.A.
Acta Crystallographica Section B,
1979