Keywords
(1,2,3-triazol-1-yl)cyclohexane conformations
click reaction
cyclohexyl-1,2,3-triazol
triazolyl conformational energy
Abstract
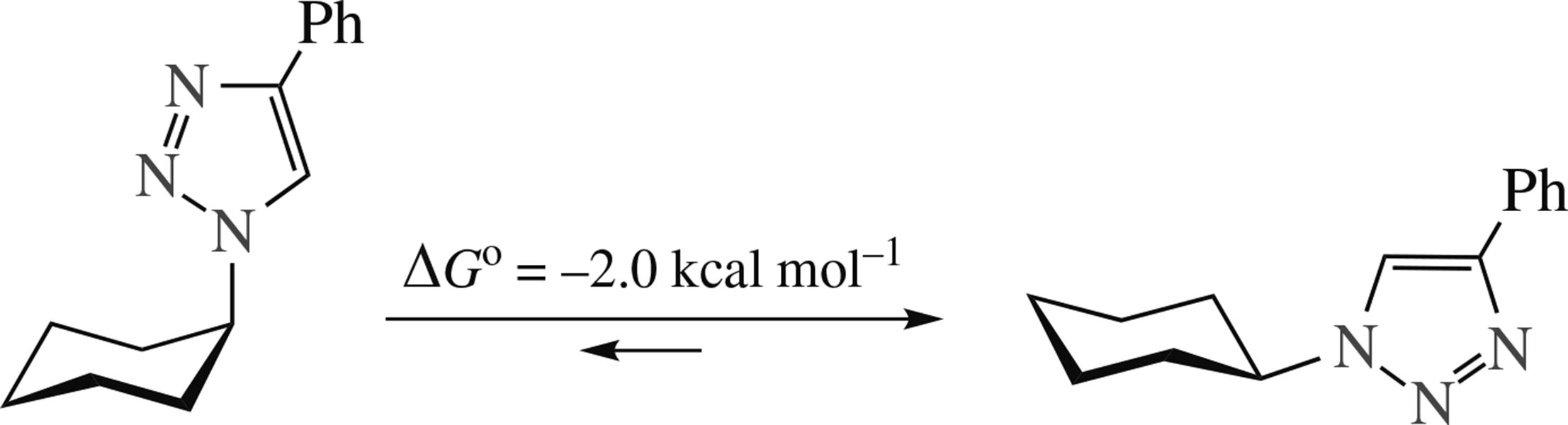
Conformational energy (A-value) of the 4-phenyl-1,2,3- triazolyl group was estimated as 2.0 kcal mol–1 (8.5 kJ mol–1) using 1H NMR-based conformational study for a set of (4-phenyl-1,2,3-triazol-1-yl)cyclohexanes. This provides a reasonable estimation also for the conformational energy of 1,2,3-triazolyl group itself and its substituted analogues.
References
1.

Lou J., Zhang X., Best M.D.
Chemistry - A European Journal,
2018
2.

Knipe P.C., Thompson S., Hamilton A.D.
Chemical Science,
2015
3.
Applications of Supramolecular Chemistry, ed. H.-J. Schneider, CRC Press, Boca Raton, 2012.
4.

Kay E., Leigh D., Zerbetto F.
Angewandte Chemie - International Edition,
2007
5.

Samoshin V.V.
Biomolecular Concepts,
2014
6.

Zheng Y., Liu X., Samoshina N.M., Samoshin V.V., Franz A.H., Guo X.
Chemistry and Physics of Lipids,
2018
7.

Samoshin V.V., Zheng Y., Liu X.
Journal of Physical Organic Chemistry,
2017
8.
Ruyonga M.R., Mendoza O., Samoshin V.V.
Mendeleev Communications,
2019
9.

Ruyonga M.R., Mendoza O., Browne M., Samoshin V.V.
Journal of Physical Organic Chemistry,
2020
10.

Tan A.
Journal of Molecular Structure,
2020
11.

Holmstrøm T., Raydan D., Pedersen C.M.
Beilstein Journal of Organic Chemistry,
2020
12.

Shiri P., Amani A.M., Mayer-Gall T.
Beilstein Journal of Organic Chemistry,
2021
13.
10.1016/j.mencom.2021.09.006_b0065
Samoshin
J. Undergrad. Chem. Res.,
2015
14.

Booth H.
Progress in Nuclear Magnetic Resonance Spectroscopy,
1969
15.
10.1016/j.mencom.2021.09.006_b0075
Eliel
Chem. Ind.,
1959
16.
10.1016/j.mencom.2021.09.006_b0080
Eliel
Stereochemistry of Organic Compounds,
1994
17.

Booth H., Thornburrow P.R.
Journal of the Chemical Society B Physical Organic,
1971