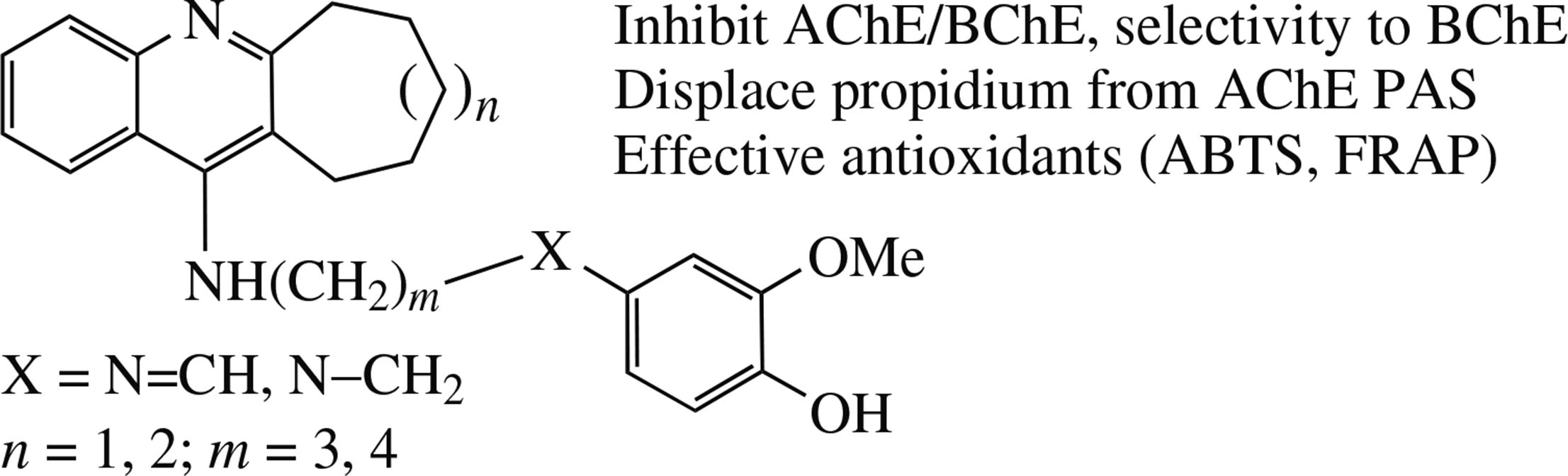
Abstract
A series of novel conjugates of 4-amino-2,3-polymethylene quinolines and phenolic antioxidant vanillin was synthesized by the condensation of aminoquinolines with vanillin followed by reduction of imines with sodium borohydride. The conjugates effectively inhibit AChE and BChE with preferable BChE inhibition and displace propidium from the PAS AChE. Compounds with aminoalkyl spacer have preferable esterase profile being more potent cholinesterases inhibitors with lower anti-CES activity and are the most potent antioxidants in ABTS and FRAP tests.
References
1.
World Alzheimer Report 2019: Attitudes to Dementia, Alzheimer’s Disease International, London, 2019, https://www.alz.co.uk/research/ world-report-2019.
2.

Bachurin S.O., Bovina E.V., Ustyugov A.A.
Medicinal Research Reviews,
2017
3.
10.1016/j.mencom.2021.09.005_b0015
Oset-Gasque
Neurosci.,
2018
4.
10.1016/j.mencom.2021.09.005_b0020
Martinez
Drugs,
2006
5.

Furukawa-Hibi Y., Alkam T., Nitta A., Matsuyama A., Mizoguchi H., Suzuki K., Moussaoui S., Yu Q., Greig N.H., Nagai T., Yamada K.
Behavioural Brain Research,
2011
6.
Nordberg A., Ballard C., Bullock R., Darreh-Shori T., Somogyi M.
The primary care companion for CNS disorders,
2013
7.

Lane R.M., Potkin S.G., Enz A.
International Journal of Neuropsychopharmacology,
2005
8.

BACHURIN S., TKACHENKO S., BASKIN I., LERMONTOVA N., MUKHINA T., PETROVA L., USTINOV A., PROSHIN A., GRIGORIEV V., LUKOYANOV N., PALYULIN V., ZEFIROV N.
Annals of the New York Academy of Sciences,
2006
9.

Tikhonova I.G., Baskin I.I., Palyulin V.A., Zefirov N.S., Bachurin S.O.
Journal of Medicinal Chemistry,
2002
10.

Cavalli A., Bolognesi M.L., Minarini A., Rosini M., Tumiatti V., Recanatini M., Melchiorre C.
Journal of Medicinal Chemistry,
2008
11.

Bolognesi M.L., Cavalli A.
ChemMedChem,
2016
12.

Zhou J., Jiang X., He S., Jiang H., Feng F., Liu W., Qu W., Sun H.
Journal of Medicinal Chemistry,
2019
13.

Ismaili L., Refouvelet B., Benchekroun M., Brogi S., Brindisi M., Gemma S., Campiani G., Filipic S., Agbaba D., Esteban G., Unzeta M., Nikolic K., Butini S., Marco-Contelles J.
Progress in Neurobiology,
2017
14.

Spilovska K., Korabecny J., Nepovimova E., Dolezal R., Mezeiova E., Soukup O., Kuca K.
Current Topics in Medicinal Chemistry,
2016
15.

Anti-cholinesterase hybrids as multi-target-directed ligands against Alzheimer’s disease (1998–2018)
Mishra P., Kumar A., Panda G.
Bioorganic and Medicinal Chemistry,
2019
16.

Makhaeva G.F., Kovaleva N.V., Boltneva N.P., Lushchekina S.V., Astakhova T.Y., Rudakova E.V., Proshin A.N., Serkov I.V., Radchenko E.V., Palyulin V.A., Bachurin S.O., Richardson R.J.
Molecules,
2020
17.

Mezeiova E., Soukup O., Korabecny J.
Russian Chemical Reviews,
2020
18.

Makhaeva G.F., Kovaleva N.V., Boltneva N.P., Lushchekina S.V., Rudakova E.V., Stupina T.S., Terentiev A.A., Serkov I.V., Proshin A.N., Radchenko E.V., Palyulin V.A., Bachurin S.O., Richardson R.J.
Bioorganic Chemistry,
2020
19.

Makhaeva G.F., Kovaleva N.V., Rudakova E.V., Boltneva N.P., Lushchekina S.V., Faingold I.I., Poletaeva D.A., Soldatova Y.V., Kotelnikova R.A., Serkov I.V., Ustinov A.K., Proshin A.N., Radchenko E.V., Palyulin V.A., Richardson R.J., et. al.
Molecules,
2020
20.

Tai A., Sawano T., Yazama F., Ito H.
Biochimica et Biophysica Acta - General Subjects,
2011
21.

Bezerra D.P., Soares A.K., de Sousa D.P.
Oxidative Medicine and Cellular Longevity,
2016
22.

Scipioni M., Kay G., Megson I., Kong Thoo Lin P.
European Journal of Medicinal Chemistry,
2018
23.

Makhaeva G.F., Rudakova E.V., Kovaleva N.V., Lushchekina S.V., Boltneva N.P., Proshin A.N., Shchegolkov E.V., Burgart Y.V., Saloutin V.I.
Russian Chemical Bulletin,
2019
24.

Chalupova K., Korabecny J., Bartolini M., Monti B., Lamba D., Caliandro R., Pesaresi A., Brazzolotto X., Gastellier A., Nachon F., Pejchal J., Jarosova M., Hepnarova V., Jun D., Hrabinova M., et. al.
European Journal of Medicinal Chemistry,
2019
25.

Casey Laizure S., Herring V., Hu Z., Witbrodt K., Parker R.B.
Pharmacotherapy,
2013
26.
10.1016/j.mencom.2021.09.005_b0130
Taylor
Biochemistry,
1989
27.

Bartolini M., Bertucci C., Cavrini V., Andrisano V.
Biochemical Pharmacology,
2003
28.

Arce M.P., Rodríguez-Franco M.I., González-Muñoz G.C., Pérez C., López B., Villarroya M., López M.G., García A.G., Conde S.
Journal of Medicinal Chemistry,
2009
29.
10.1016/j.mencom.2021.09.005_b0145
Re
Biol. Med.,
1999
30.