Keywords
Catalysis engineering
Catalyst deactivation
dry reforming
heterogeneous catalysis
Methane activation
Methane halogenation
Methane upgrading
methanol
Selective oxidation
steam reforming
Abstract
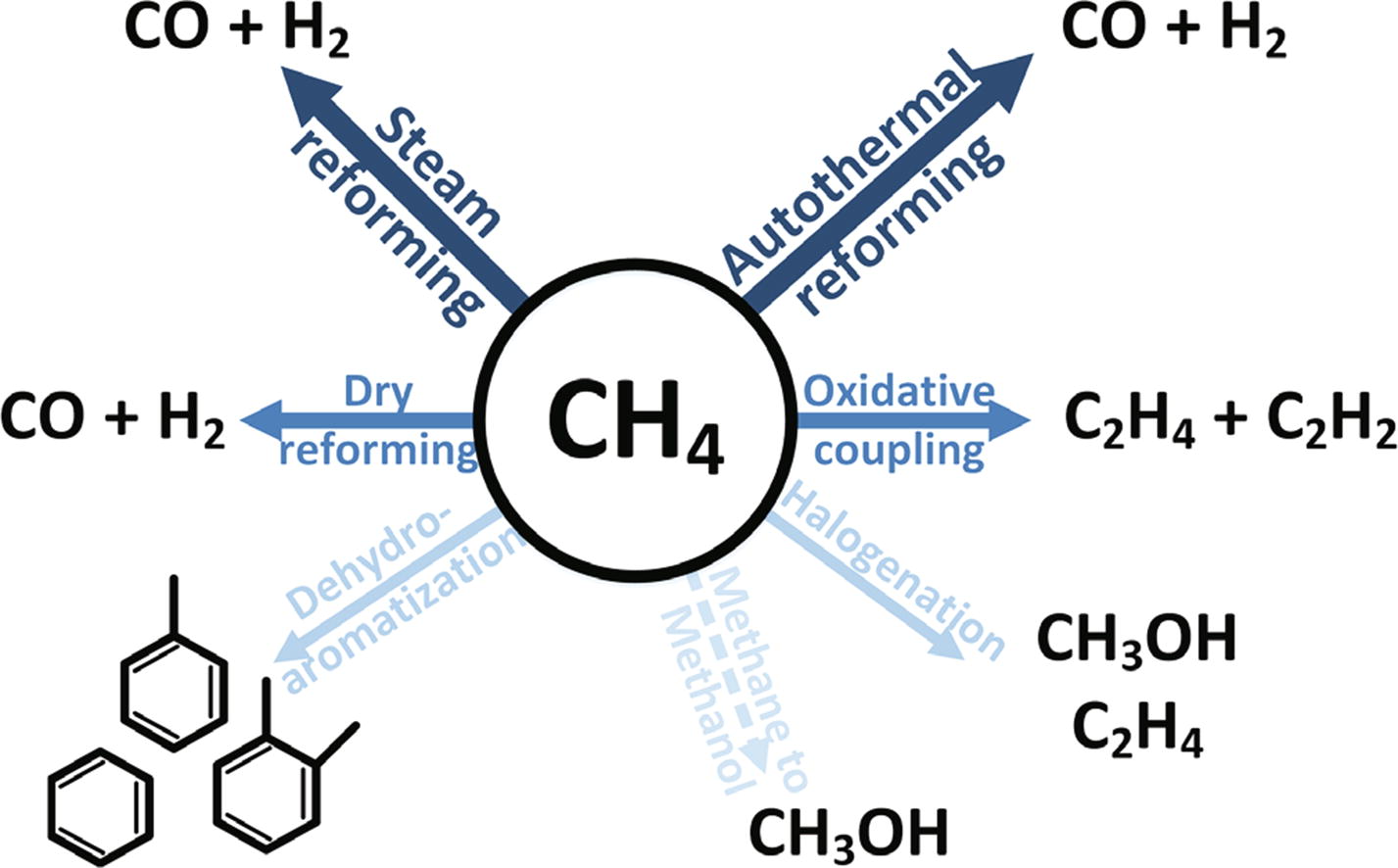
The abundance of methane has led to a strong interest to use methane as a feedstock in the chemical industry. One of the main challenges is the initial activation of the methane molecule. This has resulted in the development of several different approaches to utilize methane, some more developed than others. In this work the current status of the different approaches is discussed and the main issues for industrial utilization described. A special focus of this work is the status of catalyst development.
References
1.

Horn R., Schlögl R.
Catalysis Letters,
2014
2.
10.1016/j.mencom.2021.09.002_b0010
BP
Statistical Review of World Energy 2019,
2020
3.

Rasi S., Veijanen A., Rintala J.
Energy,
2007
4.

5.
Tracking Fuel Supply, IEA, https://www.iea.org/data-and-statistics/ charts/sources-of-greenhouse-gas-emissions-from-oil-and-gas-operations-in-2017, (accessed May 4, 2020).
6.
10.1016/j.mencom.2021.09.002_b0030
Rostrup-Nielsen
Handbook of Heterogeneous Catalysis,
2008
7.

Rostrup-Nielsen J.
Studies in Surface Science and Catalysis,
2004
8.
Murkin C., Brightling J.
Johnson Matthey Technology Review,
2016
9.

Andrew S.P.
Product R&D,
1969
10.

Rostrup-Nielsen J.R., Christensen T.S., Dybkjaer I.
Studies in Surface Science and Catalysis,
1998
11.
10.1016/j.mencom.2021.09.002_b0055
Rostrup-Nielsen
Academic Press,
2002
12.

Hammond C., Conrad S., Hermans I.
ChemSusChem,
2012
13.

Holmen A.
Catalysis Today,
2009
14.
10.1016/j.mencom.2021.09.002_b0070
Fuel Cells: Technologies for Fuel Processing,
2011
15.
10.1016/j.mencom.2021.09.002_b0075
Moulijn
Handbook of Heterogeneous Catalysis,
2008
16.

Argyle M., Bartholomew C.
Catalysts,
2015
17.

Iulianelli A., Liguori S., Wilcox J., Basile A.
Catalysis Reviews - Science and Engineering,
2016
18.

Bartholomew C.H.
Applied Catalysis A: General,
2001
19.

Wang C., Sun N., Zhao N., Wei W., Zhang J., Zhao T., Sun Y., Sun C., Liu H., Snape C.E.
ChemCatChem,
2014
20.
10.1016/j.mencom.2021.09.002_b0100
Franz
Sci. Technol.,
2020
21.

Rostrup-Nielsen J.R.
Catalysis Today,
1997
22.

Bengaard H.S., Nørskov J.K., Sehested J., Clausen B.S., Nielsen L.P., Molenbroek A.M., Rostrup-Nielsen J.R.
Journal of Catalysis,
2002
23.

Vogt C., Kranenborg J., Monai M., Weckhuysen B.M.
ACS Catalysis,
2019
24.

ROSTRUPNIELSEN J.
Journal of Catalysis,
1984
25.
10.1016/j.mencom.2021.09.002_b0125
Advances in Catalysis,
1982
26.
10.1016/j.mencom.2021.09.002_b0130
Roussière
PhD Thesis,
2013
27.

Trimm D.L.
Studies in Surface Science and Catalysis,
1991
28.

Moulijn J.A., van Diepen A.E., Kapteijn F.
Applied Catalysis A: General,
2001
29.
10.1016/j.mencom.2021.09.002_b0145
Deutschmann
in Ullmann’s Encyclopedia of Industrial Chemistry,
2011
30.
10.1016/j.mencom.2021.09.002_b0150
Hagen
Industrial Catalysis: A Practical Approach,
2015
31.

Peintinger M.F., Kratz M.J., Bredow T.
Journal of Materials Chemistry A,
2014
32.

Lamouri S., Hamidouche M., Bouaouadja N., Belhouchet H., Garnier V., Fantozzi G., Trelkat J.F.
Boletin de la Sociedad Espanola de Ceramica y Vidrio,
2017
33.
K. Wefers and C. Misra, Oxides and Hydroxides of Aluminium, Alcoa Technical Paper no. 19, Aluminium Company of America, Pittsburgh, PA, 1987.
34.
Technologies that Do More with Less, https://www.linde-engineering. com/en/about-linde-engineering/success-stories/technologies-more-with-less.html, 2019.
35.

Aramouni N.A., Touma J.G., Tarboush B.A., Zeaiter J., Ahmad M.N.
Renewable and Sustainable Energy Reviews,
2018
36.
10.1016/j.mencom.2021.09.002_b0180
Arora
RCS Adv.,
2016
37.

Vollmer I., Yarulina I., Kapteijn F., Gascon J.
ChemCatChem,
2018
38.

Spivey J.J., Hutchings G.
Chemical Society Reviews,
2014
39.

Tan P.L., Au C.T., Lai S.Y.
Catalysis Letters,
2006
40.

Weckhuysen B.M., Wang D., Rosynek M.P., Lunsford J.H.
Journal of Catalysis,
1998
41.

Schwach P., Pan X., Bao X.
Chemical Reviews,
2017
42.

Wong K.S., Thybaut J.W., Tangstad E., Stöcker M.W., Marin G.B.
Microporous and Mesoporous Materials,
2012
43.

Chen L.Y., Lin L.W., Xu Z.S., Li X.S., Zhang T.
Journal of Catalysis,
1995
44.

Kosinov N., Wijpkema A.S., Uslamin E., Rohling R., Coumans F.J., Mezari B., Parastaev A., Poryvaev A.S., Fedin M.V., Pidko E.A., Hensen E.J.
Angewandte Chemie - International Edition,
2017
45.

Ohnishi R., Liu S., Dong Q., Wang L., Ichikawa M.
Journal of Catalysis,
1999
46.

Tempelman C.H., Hensen E.J.
Applied Catalysis B: Environmental,
2015
47.
10.1016/j.mencom.2021.09.002_b0235
Portilla
Sci. Technol.,
2015
48.

Kosinov N., Coumans F.J., Uslamin E., Kapteijn F., Hensen E.J.
Angewandte Chemie - International Edition,
2016
49.

Xu Y., Lu J., Wang J., Suzuki Y., Zhang Z.
Chemical Engineering Journal,
2011
50.

Gimeno M.P., Soler J., Herguido J., Menéndez M.
Industrial & Engineering Chemistry Research,
2010
51.

Morejudo S.H., Zanón R., Escolástico S., Yuste-Tirados I., Malerød-Fjeld H., Vestre P.K., Coors W.G., Martínez A., Norby T., Serra J.M., Kjølseth C.
Science,
2016
52.
10.1016/j.mencom.2021.09.002_b0260
Natesakhawat
Sci. Technol.,
2015
53.

Guo X., Fang G., Li G., Ma H., Fan H., Yu L., Ma C., Wu X., Deng D., Wei M., Tan D., Si R., Zhang S., Li J., Sun L., et. al.
Science,
2014
54.

Sakbodin M., Wu Y., Oh S.C., Wachsman E.D., Liu D.
Angewandte Chemie - International Edition,
2016
55.

Oh S.C., Schulman E., Zhang J., Fan J., Pan Y., Meng J., Liu D.
Angewandte Chemie - International Edition,
2019
56.
Hydrogen and Syngas Production and Purification Technologies, eds. K. Liu, C. Song and V. Subramani, Wiley-AIChE, 2010.
57.
10.1016/j.mencom.2021.09.002_b0285
Reimert
Wiley-VCH,
2011
58.

York A.P.
Topics in Catalysis,
2003
59.

Schwiedernoch R., Tischer S., Correa C., Deutschmann O.
Chemical Engineering Science,
2003
60.
H. T. S/A, SynCOR™ – Autothermal Reformer (ATR), https://www. topsoe.com/products/equipment/syncortm-autothermal-reformer-atr, (accessed Mar. 19, 2020).
61.

Giroux T., Hwang S., Liu Y., Ruettinger W., Shore L.
Applied Catalysis B: Environmental,
2005
62.

Palma V., Ricca A., Ciambelli P.
Chemical Engineering Journal,
2012
63.
10.1016/j.mencom.2021.09.002_b0315
Hinsen
Chem.–Ztg.,
1983
64.

KELLER G.
Journal of Catalysis,
1982
65.
10.1016/j.mencom.2021.09.002_b0325
Zavyalova
ChemCatChem,
1935
66.

Hu L., Pinto D., Urakawa A.
Catalysis,
2020
67.
68.
C. A. Jones, J. J. Leonard and J. A. Sofranko, Patent US 4443644, 1984.
69.

Bostan A.I., Pyatnitskii Y.I., Raevskaya L.N., Pryanikova V.G., Nedil’ko S.A., Dzyaz’ko A.G., Zen’kovich E.G.
Theoretical and Experimental Chemistry,
2005
70.

Ito T., Wang J., Lin C.H., Lunsford J.H.
Journal of the American Chemical Society,
1985
71.

Au C.T., Zhou X.P., Liu Y.W., Ji W.J., Ng C.F.
Journal of Catalysis,
1998
72.
10.1016/j.mencom.2021.09.002_b0360
Machida
J. Chem. Soc., Chem. Commun.,
1987
73.
E. C. Scher, F. R. Zurcher, J. M. Cizeron, W. P. Schammel, A. Tkachenko, J. Gamoras, D. Karshtedt and G. Nyce, Patent EP 2576046A2, 2013.
74.
Revisiting the oxidative coupling of methane to ethylene in the golden period of shale gas: A review
Galadima A., Muraza O.
Journal of Industrial and Engineering Chemistry,
2016
75.
10.1016/j.mencom.2021.09.002_b0375
Joe
Energy,
2019
76.
10.1016/j.mencom.2021.09.002_b0380
Butter
Patent US 3894107A,
1973
77.

Taylor C.E., Noceti R.P., Schehl R.R.
Studies in Surface Science and Catalysis,
1988
78.

SVELLE S., ARAVINTHAN S., BJORGEN M., LILLERUD K., KOLBOE S., DAHL I., OLSBYE U.
Journal of Catalysis,
2006
79.

Nilsen M.H., Svelle S., Aravinthan S., Olsbye U.
Applied Catalysis A: General,
2009
80.

Paunović V., Lin R., Scharfe M., Amrute A.P., Mitchell S., Hauert R., Pérez‐Ramírez J.
Angewandte Chemie - International Edition,
2017
81.

Paunović V., Zichittella G., Moser M., Amrute A.P., Pérez-Ramírez J.
Nature Chemistry,
2016
82.

Lange J.-., Tijm P.J.
Chemical Engineering Science,
1996
83.

Gesser H.D., Hunter N.R., Prakash C.B.
Chemical Reviews,
1985
84.

Foulds G.A., Gray B.F.
Fuel Processing Technology,
1995
85.

Feng W., Knopf F.C., Dooley K.M.
Energy & Fuels,
1994
86.

Olivos-Suarez A.I., Szécsényi À., Hensen E.J., Ruiz-Martinez J., Pidko E.A., Gascon J.
ACS Catalysis,
2016
87.

Barbaux Y., Elamrani A.R., Payen E., Gengembre L., Bonnelle J.P., Grzybowska B.
Applied Catalysis,
1988
88.

Barbero J.A., Alvarez M.C., Bañares M.A., Peña M.A., Fierro J.L.
Chemical Communications,
2002
89.

Chen S.Y., Willcox D.
Industrial & Engineering Chemistry Research,
1993
90.

SPENCER N.
Journal of Catalysis,
1988
91.

SPENCER N.
Journal of Catalysis,
1989
92.

Ravi M., Ranocchiari M., van Bokhoven J.A.
Angewandte Chemie - International Edition,
2017
93.

Friedle S., Reisner E., Lippard S.J.
Chemical Society Reviews,
2010
94.

A transient intermediate of the methane monooxygenase catalytic cycle containing an FeIVFeIV cluster
Lee S.K., Fox B.G., Froland W.A., Lipscomb J.D., Munck E.
Journal of the American Chemical Society,
1993
95.

Shteinman A.A.
FEBS Letters,
1995
96.

Cao L., Caldararu O., Rosenzweig A.C., Ryde U.
Angewandte Chemie - International Edition,
2017
97.

Dinh K.T., Sullivan M.M., Narsimhan K., Serna P., Meyer R.J., Dincă M., Román-Leshkov Y.
Journal of the American Chemical Society,
2019
98.

Groothaert M.H., Smeets P.J., Sels B.F., Jacobs P.A., Schoonheydt R.A.
Journal of the American Chemical Society,
2005
99.

Grundner S., Luo W., Sanchez-Sanchez M., Lercher J.A.
Chemical Communications,
2016
100.

Li G., Vassilev P., Sanchez-Sanchez M., Lercher J.A., Hensen E.J., Pidko E.A.
Journal of Catalysis,
2016
101.
10.1016/j.mencom.2021.09.002_b0505
Narsimhan
Sci.,
2016
102.

Tomkins P., Mansouri A., Bozbag S.E., Krumeich F., Park M.B., Alayon E.M., Ranocchiari M., van Bokhoven J.A.
Angewandte Chemie - International Edition,
2016
103.

Dubkov K.A., Ovanesyan N.S., Shteinman A.A., Starokon E.V., Panov G.I.
Journal of Catalysis,
2002
104.
10.1016/j.mencom.2021.09.002_b0520
Hammond
ACS Catal.,
1835
105.

Kondratenko E.V., Pérez-Ramírez J.
Catalysis Today,
2007
106.

Ovanesyan N.S., Dubkov K.A., Pyalling A.A., Shteinman A.A.
Journal of Radioanalytical and Nuclear Chemistry,
2000
107.

Sobolev V.I., Dubkov K.A., Panna O.V., Panov G.I.
Catalysis Today,
1995
108.

Shan J., Huang W., Nguyen L., Yu Y., Zhang S., Li Y., Frenkel A.I., Tao F.(.
Langmuir,
2014
109.

Beznis N.V., van Laak A.N., Weckhuysen B.M., Bitter J.H.
Microporous and Mesoporous Materials,
2011
110.

Beznis N.V., Weckhuysen B.M., Bitter J.H.
Catalysis Letters,
2009
111.

Kung M.C., Lin S.S., Kung H.H.
Topics in Catalysis,
2012
112.

Baek J., Rungtaweevoranit B., Pei X., Park M., Fakra S.C., Liu Y., Matheu R., Alshmimri S.A., Alshehri S., Trickett C.A., Somorjai G.A., Yaghi O.M.
Journal of the American Chemical Society,
2018
113.

Ikuno T., Zheng J., Vjunov A., Sanchez-Sanchez M., Ortuño M.A., Pahls D.R., Fulton J.L., Camaioni D.M., Li Z., Ray D., Mehdi B.L., Browning N.D., Farha O.K., Hupp J.T., Cramer C.J., et. al.
Journal of the American Chemical Society,
2017
114.

Osadchii D.Y., Olivos-Suarez A.I., Szécsényi Á., Li G., Nasalevich M.A., Dugulan I.A., Crespo P.S., Hensen E.J., Veber S.L., Fedin M.V., Sankar G., Pidko E.A., Gascon J.
ACS Catalysis,
2018
115.

Zheng J., Ye J., Ortuño M.A., Fulton J.L., Gutiérrez O.Y., Camaioni D.M., Motkuri R.K., Li Z., Webber T.E., Mehdi B.L., Browning N.D., Penn R.L., Farha O.K., Hupp J.T., Truhlar D.G., et. al.
Journal of the American Chemical Society,
2019
116.

Tomkins P., Ranocchiari M., van Bokhoven J.A.
Accounts of Chemical Research,
2017
117.

Grundner S., Markovits M.A., Li G., Tromp M., Pidko E.A., Hensen E.J., Jentys A., Sanchez-Sanchez M., Lercher J.A.
Nature Communications,
2015
118.

Wulfers M.J., Teketel S., Ipek B., Lobo R.F.
Chemical Communications,
2015
119.

Markovits M.A., Jentys A., Tromp M., Sanchez-Sanchez M., Lercher J.A.
Topics in Catalysis,
2016
120.

Ipek B., Lobo R.F.
Chemical Communications,
2016
121.

Parfenov M.V., Starokon E.V., Pirutko L.V., Panov G.I.
Journal of Catalysis,
2014
122.

Dinh K.T., Sullivan M.M., Serna P., Meyer R.J., Dincă M., Román-Leshkov Y.
ACS Catalysis,
2018
123.

Lee S.J., McCormick M.S., Lippard S.J., Cho U.
Nature,
2013
124.

Shilov A.E., Shul'pin G.B.
Russian Chemical Reviews,
1987
125.

Periana R.A., Taube D.J., Gamble S., Taube H., Satoh T., Fujii H.
Science,
1998
126.

Ahlquist M., Nielsen R.J., Periana R.A., Goddard III W.A.
Journal of the American Chemical Society,
2009
127.
10.1016/j.mencom.2021.09.002_b0635
Vargaftik
J. Chem. Soc., Chem. Commun.,
1990
128.

Low-temperature, palladium(II)-catalyzed, solution-phase oxidation of methane to methanol derivative
Kao L.C., Hutson A.C., Sen A.
Journal of the American Chemical Society,
1991
129.

Ravi M., Sushkevich V.L., Knorpp A.J., Newton M.A., Palagin D., Pinar A.B., Ranocchiari M., van Bokhoven J.A.
Nature Catalysis,
2019
130.

Bavykina A.V., Olivos-Suarez A.I., Osadchii D., Valecha R., Franz R., Makkee M., Kapteijn F., Gascon J.
ACS applied materials & interfaces,
2017
131.

Palkovits R., Antonietti M., Kuhn P., Thomas A., Schüth F.
Angewandte Chemie - International Edition,
2009