Keywords
acylethynylpyrrole
aminoacrylonitrile
aminoenone
bipyrrole
Cyclization
pyridine
Pyrrole
pyrrolizine
pyrrolo[1,2-a]pyrazine
pyrrolyl-isoxazole
Abstract
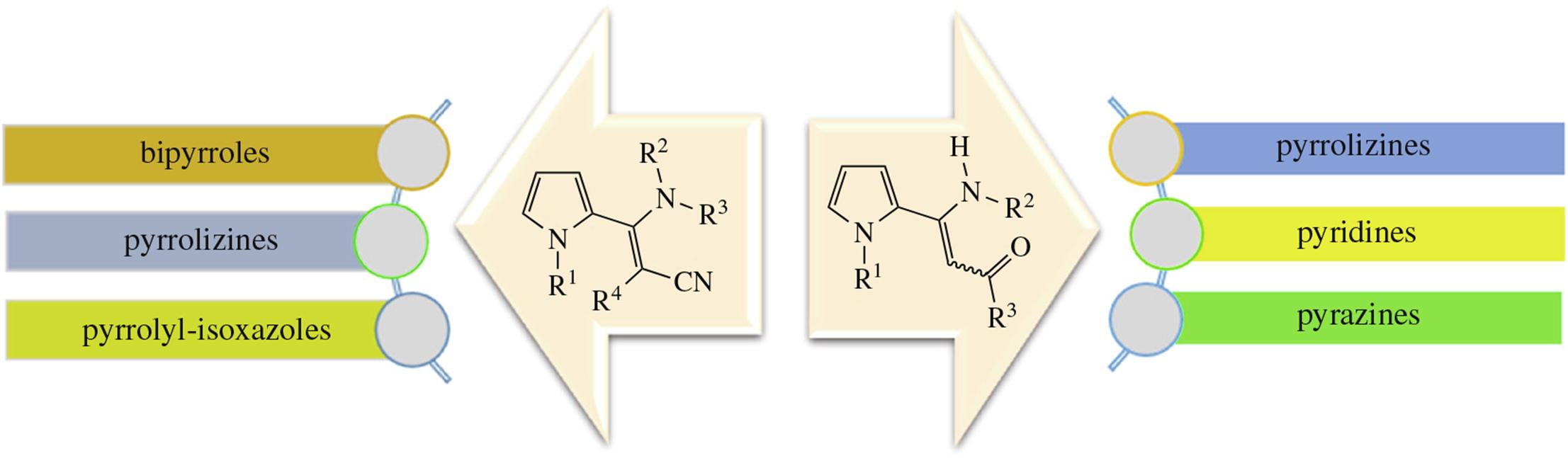
Methods for the preparation of two highly flexible synthetic building blocks, namely pyrrole/aminoenone and pyrrole/aminoacrylonitrile ensembles, on the basis of available starting materials such as 2-acylethynylpyrroles or pyrrole-2-carbodithioates, are summarized. The presence of several reactive centers in their molecules (pyrrole ring, enamine and carbonyl or nitrile moieties) ensures their multiple reactivity and application as versatile intermediates in the synthesis of heterocyclic ensembles such as pyrrolyl pyridines, bipyrroles, pyrrolyl-isoxazoles and condensed compounds, such as pyrrolo[3,2-a]pyrazines, pyrrolizines, which have high potential for use in medical chemistry and materials science.
References
1.

Clarke P.A., Santos S., Martin W.H.
Green Chemistry,
2007
2.
10.1016/j.mencom.2021.09.001_h0010
Zhang
Pot, Atom, and Step Economy (PASE) Synthesis,
2019
3.

Cheng Y., Huang Z., Wang M.
Current Organic Chemistry,
2004
4.

Dar'in D.V., Lobanov P.S.
Russian Chemical Reviews,
2015
5.

Smirnova Y.V., Krasnaya Z.A.
Russian Chemical Reviews,
2000
6.

Elassar A.A., El-Khair A.A.
Tetrahedron,
2003
7.

Stanovnik B., Svete J.
Chemical Reviews,
2004
8.
(d) K. M. Al-Zaydi and L. M. Nhari, Orient. J. Chem., 2006, 22, 38808
9.

Arshadi S., Vessally E., Edjlali L., Ghorbani-Kalhor E., Hosseinzadeh-Khanmiri R.
RSC Advances,
2017
10.

Bondock S., El-Gaber Tarhoni A., A. Fadda A.
Current Organic Chemistry,
2011
11.
![Chemistry of Enaminonitriles of Pyrano[2,3-c]pyrazole and Related Compounds](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Fadda A.A., El-Mekabaty A., Elattar K.M.
Synthetic Communications,
2013
12.
![The Reactivity of 8-Hydroxyquinoline and Its Derivatives Toward α-Cyanocinnamonitriles and Ethyl α-Cyanocinnamates: Synthesis, Reactions, and Applications of 4H-Pyrano[3,2-h]quinoline Derivatives](/storage/images/resized/SovkqI6gX6to8o5XBnS9RNxEOYVicn7hatkMzrvl_small_thumb.webp)
13.

Li X., Du Y., Liang Z., Li X., Pan Y., Zhao K.
Organic Letters,
2009
14.

Weng J., Chen Y., Yue B., Xu M., Jin H.
European Journal of Organic Chemistry,
2015
15.

Gao P., Wang J., Bai Z., Shen L., Yan Y., Yang D., Fan M., Guan Z.
Organic Letters,
2016
16.

Nguyen H.H., Kurth M.J.
Organic Letters,
2012
17.

Chen J., Properzi R., Uccello D.P., Young J.A., Dushin R.G., Starr J.T.
Organic Letters,
2014
18.
![One-Pot Synthesis of 2,4,5-Trisubstituted Imidazoles via [2 + 2 + 1] Cycloannulation of 1,3-Bishet(aryl)-monothio-1,3-diketones, α-Substituted Methylamines and Sodium Nitrite through α-Nitrosation of Enaminones](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Yugandar S., Konda S., Parameshwarappa G., Ila H.
Journal of Organic Chemistry,
2016
19.

Cheng G., Weng Y., Yang X., Cui X.
Organic Letters,
2015
20.

Fañanás F.J., Arto T., Mendoza A., Rodríguez F.
Organic Letters,
2011
21.

Li Y., Zhu Y., Xiang S., Fan W., Jin J., Huang D.
RSC Advances,
2020
22.

Alnajjar A., Abdelkhalik M.M., Al-Enezi A., Elnagdi M.H.
Molecules,
2008
23.

El Kharrat S., Laurent P., Blancou H.
Tetrahedron,
2014
24.
(a) H. M. F. Madkour, A. A. E. Afify, A. A. Abdalha, G. A. Elsayed and M. S. Salem, Phosphorus, Sulfur Silicon Relat. Elem., 2009, 184, 719; (b) M. A. Khalil, S. M. Sayed and M. A. Raslan, Am. J. Org. Chem., 2012, 2, 171; (c) A. A. Fadda and K. M. Elattar, J. Heterocycl. Chem., 2014, 51, 1697; (d) R. El-Sayed and I. Althagafi, J. Oleo Sci., 2016, 65, 177; (e) A. K. Elziaty, G. Bassioni, A. M. A. Hassan, H. A. Derbala and M. S. Abdel-Aziz, J. Chem., 2016, Article ID 5286462; (f) M. M. El-Shahawi and A. K. El-Ziaty, J. Chem., 2017, Article ID 5610707.
25.

Vitaku E., Smith D.T., Njardarson J.T.
Journal of Medicinal Chemistry,
2014
26.

Heravi M.M., Zadsirjan V.
RSC Advances,
2020
27.
10.1016/j.mencom.2021.09.001_h0115
Kerru
Molecules,
1909
28.

Benovsky P., Stille J.R.
Tetrahedron Letters,
1997
29.

Wu X., Nie L., Fang H., Chen J., Cao W., Zhao G.
European Journal of Organic Chemistry,
2011
30.
![N-Heterocyclic carbene-catalyzed annulation of cyclic β-enamino esters with enals: access to functionalized indolo[2,3-a]quinolizidines](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Hu S., Wang B., Zhang Y., Tang W., Fang M., Lu T., Du D.
Organic and Biomolecular Chemistry,
2015
31.
![Construction of Pyrrolo[1,2-a]indoles via Cobalt(III)-Catalyzed Enaminylation of 1-(Pyrimidin-2-yl)-1H-indoles with Ketenimines and Subsequent Base-Promoted Cyclization](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Zhou X., Fan Z., Zhang Z., Lu P., Wang Y.
Organic Letters,
2016
32.

Xu Z., Wang Y., Zheng Y., Huang Z., Ackermann L., Ruan Z.
Organic Chemistry Frontiers,
2020
33.
10.1016/j.mencom.2021.09.001_h0145
Chakrabarti
Junjappa, Synlett,
2005
34.
Beliaev N.A., Beryozkina T.V., Lubec G., Bakulev V.A.
Mendeleev Communications,
2019
35.
Belyaeva K.V., Nikitina L.P., Mal’kina A.G., Afonin A.V., Trofimov B.A.
Mendeleev Communications,
2020
36.

Trofimov B.A., Stepanova Z.V., Sobenina L.N., Mikhaleva A.I., Ushakov I.A.
Tetrahedron Letters,
2004
37.
(b) B. A. Trofimov and L. N. Sobenina, in Targets in Heterocyclic Systems, eds. O. A. Attanasi and D. Spinelli, Italian Society of Chemistry, Rome, 2009, vol. 13, pp. 92-119
38.
Tomilin D.N., Soshnikov D.Y., Trofimov A.B., Gotsko M.D., Sobenina L.N., Ushakov I.A., Afonin A.V., Koldobsky A.B., Vitkovskaya N.M., Trofimov B.A.
Mendeleev Communications,
2016
39.
(d) E. F. Sagitova, D. N. Tomilin, O. V. Petrova, A. B. Budaev, L. N. Sobenina, B. A. Trofimov, G. Q. Yang and R. Hu, Mendeleev Commun., 2019, 29, 658
40.

Sobenina L.N., Trofimov B.A.
Molecules,
2020
41.

Sobenina L.N., Demenev A.P., Mikhaleva A.I., Trofimov B.A.
Russian Chemical Reviews,
2002
42.

Trofimov B.A., Sobenina L.N., Demenev A.P., Mikhaleva A.I.
Chemical Reviews,
2004
43.

Trofimov B.A., Tomilin D.N., Sobenina L.N., Ushakov I.A.
Synthesis,
2020
44.
10.1016/j.mencom.2021.09.001_b0135
Sobenina
Synthesis,
2010
45.

Sobenina L.N., Tomilin D.N., Sagitova E.F., Ushakov I.A., Trofimov B.A.
Organic Letters,
2017
46.
![Transition-Metal-Free Synthesis of Pyrrolo[1,2-a]pyrazines via Intramolecular Cyclization of N-Propargyl(pyrrolyl)enaminones](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Sobenina L., Sagitova E., Ushakov I., Trofimov B.
Synthesis,
2017
47.
![From Acylethynylpyrroles to Pyrrolo[1,2-a]pyrazines in One Step](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Sagitova E.F., Sobenina L.N., Trofimov B.A.
Russian Journal of Organic Chemistry,
2020
48.
10.1016/j.mencom.2021.09.001_b0155
Kaur
J. Pharm. Chem. Chem. Sci.,
2017
49.

Canjeevaram Balasubramanyam R.K., Kandjani A.E., Jones L.A., Periasamy S.R., Wong S., Narayan R., Bhargava S.K., Ippolito S.J., Basak P.
Advanced Electronic Materials,
2018
50.
![New class of antitubercular compounds: synthesis and anti-tubercular activity of 4-substituted pyrrolo[2,3-c]quinolines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Akula M., Sridevi J.P., Yogeeswari P., Sriram D., Bhattacharya A.
Monatshefte fur Chemie,
2014
51.

Lehuédé J., Fauconneau B., Barrier L., Ourakow M., Piriou A., Vierfond J.
European Journal of Medicinal Chemistry,
1999
52.
10.1016/j.mencom.2021.09.001_b0175
Raines
Patent US,
2016
53.

de Laszlo S.E., Visco D., Agarwal L., Chang L., Chin J., Croft G., Forsyth A., Fletcher D., Frantz B., Hacker C., Hanlon W., Harper C., Kostura M., Li B., Luell S., et. al.
Bioorganic and Medicinal Chemistry Letters,
1998
54.
10.1016/j.mencom.2021.09.001_b0185
Chang
Patent EP,
1996
55.

Belal A., El-Gendy B.E.
Bioorganic and Medicinal Chemistry,
2014
56.

Gouda A.M., Abdelazeem A.H., Arafa E.A., Abdellatif K.R.
Bioorganic Chemistry,
2014
57.

Argyropoulos N.G., Gkizis P., Coutouli-Argyropoulou E.
Tetrahedron,
2008
58.

Sutariya T.R., Labana B.M., Parmar N.J., Kant R., Gupta V.K., Plata G.B., Padrón J.M.
New Journal of Chemistry,
2015
59.

Gouda A.M., Abdelazeem A.H.
European Journal of Medicinal Chemistry,
2016
60.

Attalah K.M., Abdalla A.N., Aslam A., Ahmed M., Abourehab M.A., ElSawy N.A., Gouda A.M.
Bioorganic Chemistry,
2020
61.

Sparatore A., Basilico N., Casagrande M., Parapini S., Taramelli D., Brun R., Wittlin S., Sparatore F.
Bioorganic and Medicinal Chemistry Letters,
2008
62.
10.1016/j.mencom.2021.09.001_b0220
Barsoum
Boll. Chim. Farm.,
2003
63.

Sonnet P., Dallemagne P., Guillon J., Enguehard C., Stiebing S., Tanguy J., Bureau R., Rault S., Auvray P., Moslemi S., Sourdaine P., Séralini G.
Bioorganic and Medicinal Chemistry,
2000
64.

Smolka A.J., Goldenring J.R., Gupta S., Hammond C.E.
BMC Gastroenterology,
2004
65.

Liedtke A.J., Keck P.R., Lehmann F., Koeberle A., Werz O., Laufer S.A.
Journal of Medicinal Chemistry,
2009
66.
![Expansion of chemical space based on a pyrrolo[1,2-a]pyrazine core: Synthesis and its anticancer activity in prostate cancer and breast cancer cells](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Seo Y., Lee J.H., Park S., Namkung W., Kim I.
European Journal of Medicinal Chemistry,
2020
67.
10.1016/j.mencom.2021.09.001_b0245
Garzan
Dis.,
2017
68.
![Synthesis and In Vitro Anti-Inflammatory Activity of Pyrrolo[1,2-A]pyrazines via Pd-Catalyzed Intermolecular Cyclization Reaction](/storage/images/resized/lZMRu5MhhE0HtEBGlENti9uMMEivcrzFOV6rqpGN_small_thumb.webp)
Zhou Y.M., Wei Y., Yang J., Li H.H., Liu M.D., Huang N.Y.
Advanced Materials Research,
2013
69.

Reker D., Seet M., Pillong M., Koch C.P., Schneider P., Witschel M.C., Rottmann M., Freymond C., Brun R., Schweizer B., Illarionov B., Bacher A., Fischer M., Diederich F., Schneider G., et. al.
Angewandte Chemie - International Edition,
2014
70.
10.1016/j.mencom.2021.09.001_b0260
Kolli
Pharma Chem.,
2018
71.
![Novel fluorinated pyrrolo[1,2-a]pyrazine-2,6-dione derivatives: Synthesis and anticonvulsant evaluation in animal models of epilepsy](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Dawidowski M., Chońska J., Mika W., Turło J.
Bioorganic and Medicinal Chemistry,
2014
72.

Barresi E., Robello M., Costa B., Da Pozzo E., Baglini E., Salerno S., Da Settimo F., Martini C., Taliani S.
European Journal of Medicinal Chemistry,
2021
73.
![8-Hydroxy-3,4-dihydropyrrolo[1,2-a]pyrazine-1(2H)-one HIV-1 integrase inhibitors](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fisher T.E., Kim B., Staas D.D., Lyle T.A., Young S.D., Vacca J.P., Zrada M.M., Hazuda D.J., Felock P.J., Schleif W.A., Gabryelski L.J., Anari M.R., Kochansky C.J., Wai J.S.
Bioorganic and Medicinal Chemistry Letters,
2007
74.
![Synthesis and Mechanism Studies of 1,3-Benzoazolyl Substituted Pyrrolo[2,3-b]pyrazine Derivatives as Nonintercalative Topoisomerase II Catalytic Inhibitors](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Li P., Zeng P., Chen S., Yao P., Mai Y., Tan J., Ou T., Huang S., Li D., Gu L., Huang Z.
Journal of Medicinal Chemistry,
2015
75.
![Pyrrolo[1,2-a]pyrazine and pyrazolo[1,5-a]pyrazine: Novel, potent, and selective series of Vasopressin1b receptor antagonists](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Arban R., Bianchi F., Buson A., Cremonesi S., Fabio R.D., Gentile G., Micheli F., Pasquarello A., Pozzan A., Tarsi L., Terreni S., Tonelli F.
Bioorganic and Medicinal Chemistry Letters,
2010
76.

Padilla F., Bhagirath N., Chen S., Chiao E., Goldstein D.M., Hermann J.C., Hsu J., Kennedy-Smith J.J., Kuglstatter A., Liao C., Liu W., Lowrie L.E., Luk K.C., Lynch S.M., Menke J., et. al.
Journal of Medicinal Chemistry,
2013
77.

Crawford J.J., Johnson A.R., Misner D.L., Belmont L.D., Castanedo G., Choy R., Coraggio M., Dong L., Eigenbrot C., Erickson R., Ghilardi N., Hau J., Katewa A., Kohli P.B., Lee W., et. al.
Journal of Medicinal Chemistry,
2018
78.

Burdick D.J., Wang S., Heise C., Pan B., Drummond J., Yin J., Goeser L., Magnuson S., Blaney J., Moffat J., Wang W., Chen H.
Bioorganic and Medicinal Chemistry Letters,
2015
79.
10.1016/j.mencom.2021.09.001_b0295
Zhao
Patent WO,
2019
80.
![Novel, Highly Potent Aldose Reductase Inhibitors: (R)-(−)-2-(4-Bromo-2-fluorobenzyl)-1,2,3,4-tetrahydropyrrolo[1,2-a]pyrazine- 4-spiro-3‘-pyrrolidine-1,2‘,3,5‘-tetrone (AS-3201) and Its Congeners](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Negoro T., Murata M., Ueda S., Fujitani B., Ono Y., Kuromiya A., Komiya M., Suzuki K., Matsumoto J.
Journal of Medicinal Chemistry,
1998
81.
10.1016/j.mencom.2021.09.001_b0305
Micheli
Bioorg. Med. Chem. Lett.,
1804
82.
10.1016/j.mencom.2021.09.001_b0310
Cheng
Patent WO,
2005
83.
10.1016/j.mencom.2021.09.001_b0315
Prunier
J. Med. Chem.,
1808
84.

Sobenina L.N., Sagitova E.F., Markova M.V., Ushakov I.A., Ivanov A.V., Trofimov B.A.
Tetrahedron Letters,
2018
85.
Sagitova E.F., Sobenina L.N., Tomilin D.N., Markova M.V., Ushakov I.A., Trofimov B.A.
Mendeleev Communications,
2019
86.

Trofimov B.A., Sobenina L.N., Mikhaleva A.I., Petrova O.V., Drichkov V.N., Ushakov I.A., Tarasova O.A., Toryashinova D.D., Rusakov Y.Y., Krivdin L.B.
Journal of Heterocyclic Chemistry,
2007
87.

Petrova O.V., Sagitova E.F., Ushakov I.A., Sobenina L.N., Mikhaleva A.I., Trofimov B.A.
Journal of Sulfur Chemistry,
2014
88.

Petrova O.V., Sagitova E.F., Sobenina L.N., Ushakov I.A., Borodina T.N., Smirnov V.I., Trofimov B.A.
Tetrahedron Letters,
2016
89.

Trofimov B.A., Sagitova E.F., Petrova O.V., Sobenina L.N., Ushakov I.A., Vashchenko A.V.
Tetrahedron Letters,
2017
90.

Lauria A., Bruno M., Diana P., Barraja P., Montalbano A., Cirrincione G., Dattolo G., Almerico A.M.
Bioorganic and Medicinal Chemistry,
2005
91.
![Adenosine Kinase Inhibitors. 6. Synthesis, Water Solubility, and Antinociceptive Activity of 5-Phenyl-7-(5-deoxy-β-d-ribofuranosyl)pyrrolo[2,3-d]pyrimidines Substituted at C4 with Glycinamides and Related Compounds](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Bookser B.C., Ugarkar B.G., Matelich M.C., Lemus R.H., Allan M., Tsuchiya M., Nakane M., Nagahisa A., Wiesner J.B., Erion M.D.
Journal of Medicinal Chemistry,
2005
92.
![Synthesis and biological evaluation of some thio containing pyrrolo [2,3-d]Pyrimidine derivatives for their anti-inflammatory and anti-microbial activities](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Mohamed M.S., Kamel R., Fatahala S.S.
European Journal of Medicinal Chemistry,
2010
93.

Sobenina L.N., Drichkov V.N., Mikhaleva A.I., Petrova O.V., Ushakov I.A., Trofimov B.A.
Tetrahedron,
2005
94.

Sechi M., Sannia L., Carta F., Palomba M., Dallocchio R., Dessì A., Derudas M., Zawahir Z., Neamati N.
Antiviral Chemistry and Chemotherapy,
2005
95.

Padmavathi V., Radha Lakshmi T., Mahesh K., Padmaja A.
Chemical and Pharmaceutical Bulletin,
2009
96.

Padmavathi V., Lakshmi T.R., Venkatesh B.C., Mahesh K.
International Journal of Organic Chemistry,
2011
97.

Barbachyn M.R., Cleek G.J., Dolak L.A., Garmon S.A., Morris J., Seest E.P., Thomas R.C., Toops D.S., Watt W., Wishka D.G., Ford C.W., Zurenko G.E., Hamel J.C., Schaadt R.D., Stapert D., et. al.
Journal of Medicinal Chemistry,
2002
98.

Joshi S.D., Dixit S.R., Kirankumar M.N., Aminabhavi T.M., Raju K.V., Narayan R., Lherbet C., Yang K.S.
European Journal of Medicinal Chemistry,
2016
99.

Meijer F.A., Doveston R.G., de Vries R.M., Vos G.M., Vos A.A., Leysen S., Scheepstra M., Ottmann C., Milroy L., Brunsveld L.
Journal of Medicinal Chemistry,
2019
100.
![Identification and profiling of 3,5-dimethyl-isoxazole-4-carboxylic acid [2-methyl-4-((2S,3′S)-2-methyl-[1,3′]bipyrrolidinyl-1′-yl)phenyl] amide as histamine H3 receptor antagonist for the treatment of depression](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Gao Z., Hurst W.J., Czechtizky W., Hall D., Moindrot N., Nagorny R., Pichat P., Stefany D., Hendrix J.A., George P.G.
Bioorganic and Medicinal Chemistry Letters,
2013
101.

Ionescu S., Chiorescu I., Hillebrand M.
Journal of Molecular Structure THEOCHEM,
2003
102.

Yang Z., Zhang K., Gong F., Li S., Chen J., Ma J.S., Sobenina L.N., Mikhaleva A.I., Yang G., Trofimov B.A.
Beilstein Journal of Organic Chemistry,
2011
103.

Jin R., Sun W., Tang S.
International Journal of Molecular Sciences,
2012