Abstract
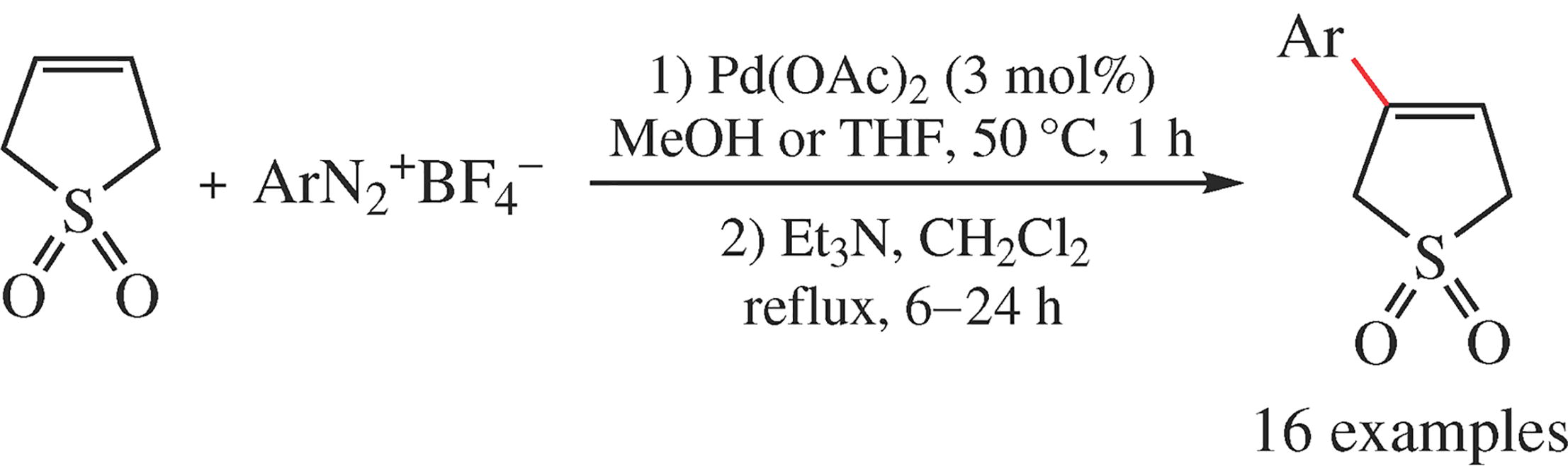
An efficient and practical method for a scalable synthesis of 3-arylbutadiene sulfones deals with the ligand-free Heck–Matsuda reaction of sulfolene with aryldiazonium tetrafluoroborates followed by triethylamine-promoted double bond shift.
References
1.

Chou T., Tso H.
Organic Preparations and Procedures International,
1989
2.

3.

Brant M.G., Wulff J.E.
Organic Letters,
2012
4.

Lim K.M., Hayashi T.
Journal of the American Chemical Society,
2015
5.

Zhou T., Peters B., Maldonado M.F., Govender T., Andersson P.G.
Journal of the American Chemical Society,
2012
6.

Kita Y., Hida S., Higashihara K., Jena H.S., Higashida K., Mashima K.
Angewandte Chemie - International Edition,
2016
7.

Meng K., Xia J., Wang Y., Zhang X., Yang G., Zhang W.
Organic Chemistry Frontiers,
2017
8.

Takale B.S., Thakore R.R., Gao E.S., Gallou F., Lipshutz B.H.
Green Chemistry,
2020
9.

Brant M.G., Bromba C.M., Wulff J.E.
Journal of Organic Chemistry,
2010
10.
![Rigid Alicyclic Molecules from Bicyclo[2.2.1]hept-2-enes (=8,9,10-Trinorbornenes) and 1,4-Dipyridin-2-ylphthalazines as Stereoselective Coupling Agents](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Margetić D., Murata Y., Komatsu K., Marinić Z.
Helvetica Chimica Acta,
2009
11.

Löber S., Tschammer N., Hübner H., Melis M.R., Argiolas A., Gmeiner P.
ChemMedChem,
2009
12.

Dang H.T., Nguyen V.D., Pham H.H., Arman H.D., Larionov O.V.
Tetrahedron,
2019
13.

Wu X., Ye H., Dai H., Yang B., Wang Y., Chen S., Hu L.
Organic Chemistry Frontiers,
2020
14.

Bloch R., Benecou C., Guibé-Jampel E.
Tetrahedron Letters,
1985
15.

Bloch R., Hassan D., Mandard X.
Tetrahedron Letters,
1983
16.

Dang H.T., Nguyen V.D., Haug G.C., Vuong N.T., Arman H.D., Larionov O.V.
ACS Catalysis,
2021
17.
![General methods for alkaloid synthesis via intramolecular [4 + 2] cycloaddition reactions of enamides. A new approach to the synthesis of Aspidosperma alkaloids](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Martin S.F., Desai S.R., Philips G.W., Miller A.C.
Journal of the American Chemical Society,
1980
18.

Takayama H., Nomoto T.
Heterocycles,
1985
19.
10.1016/j.mencom.2021.07.037_h0070
Bruyère
Synlett,
2005
20.

Tao Y.T., Liu C.L., Lee S.J., Chou S.S.
Journal of Organic Chemistry,
1986
21.
10.1016/j.mencom.2021.07.037_h0080
Pasfield
J. Org. Chem.,
2013
22.
10.1016/j.mencom.2021.07.037_h0085
Yamada
J. Chem. Soc., Chem. Commun.,
1987
23.

Köse A., Ünal A., Şahin E., Bozkaya U., Kara Y.
Beilstein Journal of Organic Chemistry,
2019
24.

Bischoff L., Martial L.
Synlett,
2015
25.

Chou T.S., Hung S.C., Tso H.H.
Journal of Organic Chemistry,
1987
26.

Bew S., Sweeney J.
Synlett,
1997
27.

Harrington P.J., DiFiore K.A.
Tetrahedron Letters,
1987
28.

Sengupta S., Bhattacharyya S.
Synthetic Communications,
1996
29.
(a) D. A. Alentiev, S. A. Korchagina, E. S. Finkel'shtein, M. S. Nechaev, A. F. Asachenko, M. A. Topchiy, P. S. Gribanov and M. V. Bermeshev, Russ. Chem. Bull., Int. Ed., 2018, 67, 121 (Izv. Akad. Nauk, Ser. Khim., 2018, 121)
30.
Gribanov P.S., Golenko Y.D., Topchiy M.A., Philippova A.N., Kirilenko N.Y., Krivoshchapov N.V., Sterligov G.K., Asachenko A.F., Bermeshev M.V., Nechaev M.S.
Mendeleev Communications,
2018
31.
Gribanov P.S., Chesnokov G.A., Dzhevakov P.B., Kirilenko N.Y., Rzhevskiy S.A., Ageshina A.A., Topchiy M.A., Bermeshev M.V., Asachenko A.F., Nechaev M.S.
Mendeleev Communications,
2019
32.

Dzhevakov P.B., Topchiy M.A., Ageshina A.A., Minaeva L.I., Rzhevskiy S.A., Nechaev M.S., Osipov S.N., Asachenko A.F.
Russian Chemical Bulletin,
2020
33.
Rzhevskiy S.A., Topchiy M.A., Golenko Y.D., Gribanov P.S., Sterligov G.K., Kirilenko N.Y., Ageshina A.A., Bermeshev M.V., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2020
34.
Topchiy M.A., Dzhevakov P.B., Kirilenko N.Y., Rzhevskiy S.A., Ageshina A.A., Khrustalev V.N., Paraschuk D.Y., Bermeshev M.V., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2019
35.
Topchiy M.A., Rzhevskiy S.A., Ageshina A.A., Kirilenko N.Y., Sterligov G.K., Mladentsev D.Y., Paraschuk D.Y., Osipov S.N., Nechaev M.S., Asachenko A.F.
Mendeleev Communications,
2020
36.

Chesnokov G.A., Ageshina A.A., Maryanova A.V., Rzhevskiy S.A., Gribanov P.S., Topchiy M.A., Nechaev M.S., Asachenko A.F.
Russian Chemical Bulletin,
2020
37.
Gribanov P.S., Lypenko D.A., Dmitriev A.V., Pozin S.I., Topchiy M.A., Asachenko A.F., Loginov D.A., Osipov S.N.
Mendeleev Communications,
2021