Abstract
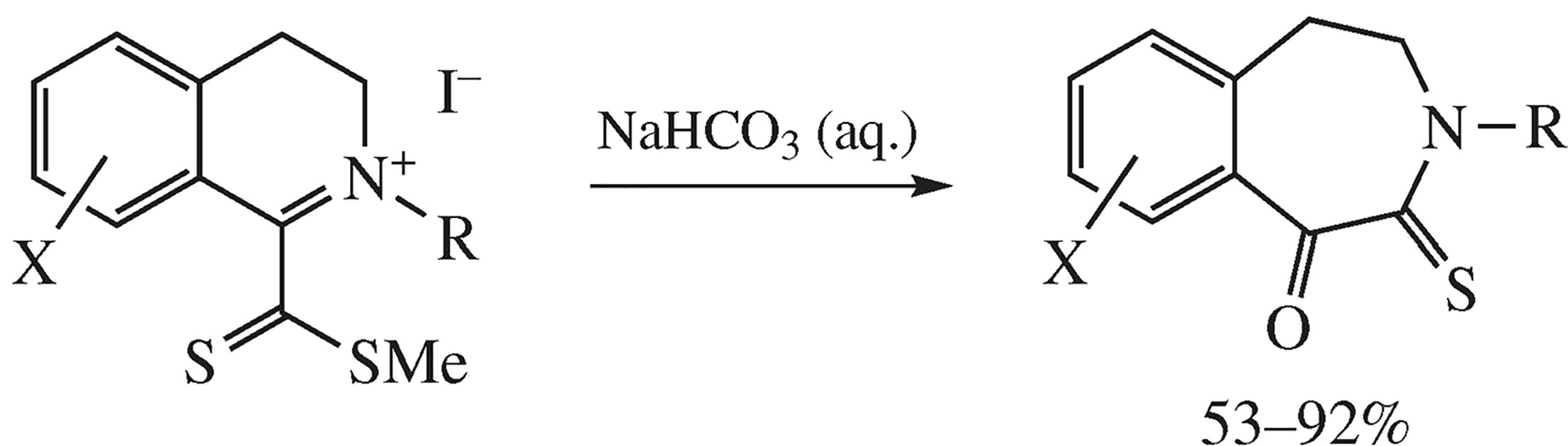
New 1-oxo-2-thioxo-1,2,4,5-tetrahydrobenz[d]azepine and 1-oxo-2-thioxo-1,2,4,5-tetrahydroazepino[4,5-b]indole derivatives were conveniently obtained by the novel recyclization reaction of (methylthio)carbonothioylsubstituted heterocyclic quaternary salts with expansion of the dihydropyridine ring.
References
1.

Danyliuk I.Y., Vas’kevich R.I., Vas’kevich A.I., Vovk M.V.
Chemistry of Heterocyclic Compounds,
2019
2.

Singh S., Goo J., Gajulapati V., Chang T., Lee K., Choi Y.
Anti-Cancer Agents in Medicinal Chemistry,
2015
3.
DrugBank, https://www.drugbank.com/.
4.
Chemistry of Heterocyclic Compounds, Vol. 56, Monocyclic Azepines: The Syntheses and Chemical Properties of the Monocyclic Azepines, eds. G. R. Proctor and J. Redpath, Wiley, New York, 1997.
5.
10.1016/j.mencom.2021.07.036_b0025
Surman
Sci. Synth.,
2003
6.
![[1,7]-Electrocyclization reactions in the synthesis of azepine derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Nedolya N.A., Trofimov B.A.
Chemistry of Heterocyclic Compounds,
2013
7.

Camara C.A., Pinto A.C., Vargas M.D., Zukerman-Schpector J.
Tetrahedron,
2002
8.

Joucla L., Putey A., Joseph B.
Tetrahedron Letters,
2005
9.
L. M. Potikha, V. V. Sypchenko and V. A. Kovtunenko, Chem. Heterocycl. Compd., 2020, 56, 1454 (Khim. Geterotsikl. Soedin., 2020, 56, 1454).
10.

Kita Y., Takada T., Gyoten M., Tohma H., Zenk M.H., Eichhorn J.
Journal of Organic Chemistry,
1996
11.

Kita Y., Arisawa M., Gyoten M., Nakajima M., Hamada R., Tohma H., Takada T.
Journal of Organic Chemistry,
1998
12.

Zubenko A.A., Kartsev V.G., Morkovnik A.S., Divaeva L.N., Suponitsky K.Y.
ChemistrySelect,
2016
13.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Kartsev V.G., Kuzmina L.G., Borodkin G.S., Klimenko A.I.
Mendeleev Communications,
2018
14.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Kartsev V.G., Suponitsky K.Y., Klimenko A.I.
Mendeleev Communications,
2018
15.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Demidov O.P., Sochnev V.S., Borodkina I.G., Drobin Y.D., Spasov A.A.
Mendeleev Communications,
2020
16.

Beaud R., Michelet B., Reviriot Y., Martin‐Mingot A., Rodriguez J., Bonne D., Thibaudeau S.
Angewandte Chemie - International Edition,
2019
17.

Morkovnik A., Zubenko A., Divaeva L., Demidov O., Gribanova T., Sochnev V., Klimenko A., Dobaeva N., Borodkin G., Drobin Y., Fetisov L., Kononenko K.
Tetrahedron Letters,
2020
18.

Tatto M.T.
Oxford Review of Education,
2015
19.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
20.
10.1016/j.mencom.2021.07.036_b0085
Sheldrick
Struct. Chem.,
2015
21.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
22.
10.1016/j.mencom.2021.07.036_b0095
Zhang
Bioorg. Chem.,
2020