Abstract
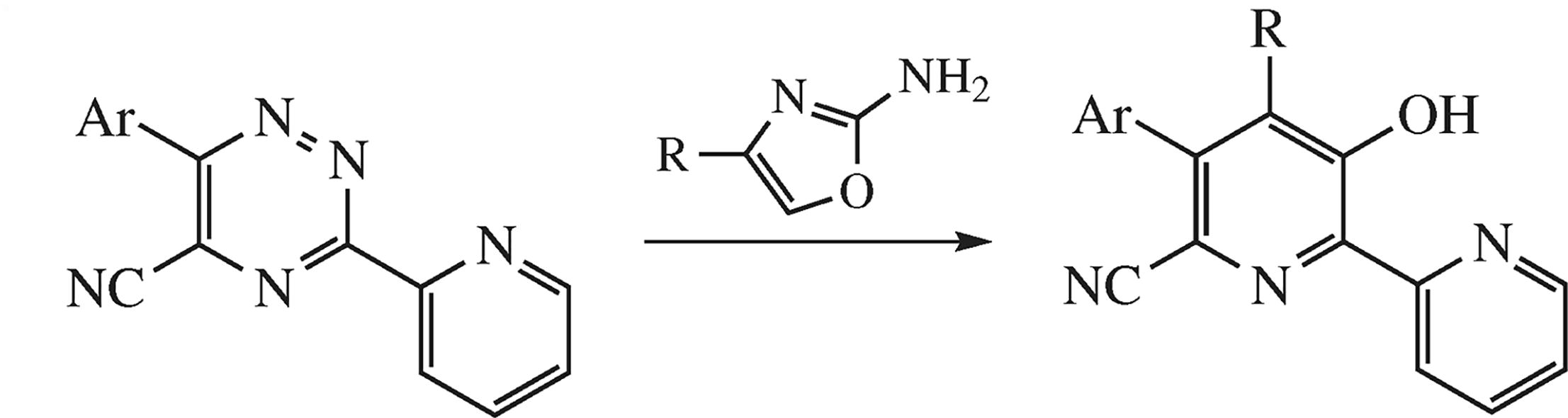
High temperature coupling of 6-aryl-5-cyano-3-(pyridin-2-yl)-1,2,4-triazines with 2-amino-4-aryloxazoles proceeds as the inverse demand Diels–Alder reaction between the oxazole moiety as dienophile and the 1,2,4-triazine moiety as diene to construct new 4,5-diaryl-6-cyano-3-hydroxypyridin-2-yl fragment. A reaction mechanism is proposed, and the structure of the key-product is proved by the X-ray diffraction analysis.
References
1.

Prokhorov A.M., Kozhevnikov D.N.
Chemistry of Heterocyclic Compounds,
2012
2.

Boger D.L., Panek J.S.
Journal of Organic Chemistry,
1981
3.

Kozhevnikov D.N., Kozhevnikov V.N., Prokhorov A.M., Ustinova M.M., Rusinov V.L., Chupakhin O.N., Aleksandrov G.G., König B.
Tetrahedron Letters,
2006
4.

Moseev T.D., Varaksin M.V., Gorlov D.A., Nikiforov E.A., Kopchuk D.S., Starnovskaya E.S., Khasanov A.F., Zyryanov G.V., Charushin V.N., Chupakhin O.N.
Journal of Fluorine Chemistry,
2019
5.

Kovalev I.S., Savchuk M.I., Kopchuk D.S., Zyryanov G.V., Pospelova T.A., Rusinov V.L., Chupakhin O.N.
Russian Journal of Organic Chemistry,
2019
6.
Fatykhov R.F., Savchuk M.I., Starnovskaya E.S., Bobkina M.V., Kopchuk D.S., Nosova E.V., Zyryanov G.V., Khalymbadzha I.A., Chupakhin O.N., Charushin V.N., Kartsev V.G.
Mendeleev Communications,
2019
7.

Kozhevnikov V.N., Kozhevnikov D.N., Nikitina T.V., Rusinov V.L., Chupakhin O.N., Zabel M., König B.
Journal of Organic Chemistry,
2003
8.
10.1016/j.mencom.2021.07.035_b0040
Catozzi
Synlett,
2007
9.
S. V. Shorshnev, S. E. Esipov, A. I. Chernyshev, A. F. Pozharskii, V. V. Kuz’menko and A. V. Gulevskaya, Chem. Heterocycl. Compd., 1990, 26, 191 (Khim. Geterotsikl. Soedin., 1990, 26, 224).
10.
![Cycloaddition Reactions with Azabenzenes, XVIII. Synthesis of [2]Pyrindines](/storage/images/resized/SovkqI6gX6to8o5XBnS9RNxEOYVicn7hatkMzrvl_small_thumb.webp)
Neunhoeffer H., Neuhoeffer H., Philipp B., Schildhauer B., Eckrich R., Krichbaum U.
Heterocycles,
1993
11.

Gonsalves A.M., Pinho e Melo T.M., Gilchrist T.L.
Tetrahedron,
1992
12.

Kopchuk D.S., Nikonov I.L., Zyryanov G.V., Kovalev I.S., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2014
13.

Kopchuk D.S., Nikonov I.L., Khasanov A.F., Gundala S., Krinochkin A.P., Slepukhin P.А., Zyryanov G.V., Venkatapuram P., Chupakhin O.N., Charushin V.N.
Chemistry of Heterocyclic Compounds,
2019
14.

Diring S., Retailleau P., Ziessel R.
Journal of Organic Chemistry,
2007
15.

OHBA S., KONNO S., YAMANAKA H.
Chemical and Pharmaceutical Bulletin,
2011
16.

Kozhevnikov D.N., Kovalev I.S., Prokhorov A.M., Rusinov V.L., Chupakhin O.N.
Russian Chemical Bulletin,
2003
17.

Huang J.J.
Journal of Organic Chemistry,
1985
18.

Kozhevnikov D.N., Kozhevnikov V.N., Kovalev I.S., Rusinov V.L., Chupakhin O.N., Aleksandrov G.G.
Russian Journal of Organic Chemistry,
2002
19.
V. L. Rusinov, G. V. Zyryanov, I. N. Egorov, E. N. Ulomskii, G. G. Aleksandrov and O. N. Chupakhin, Russ. J. Org. Chem., 2004, 40, 85 (Zh. Org. Khim., 2044, 40, 94).
20.

Rammohan A., Reddy G.M., Krinochkin A.P., Kopchuk D.S., Savchuk M.I., Shtaitz Y.K., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Synthetic Communications,
2020
21.
D. S. Kopchuk, Doctoral Thesis, 2019.
22.

Krinochkin A.P., Kopchuk D.S., Kovalev I.S., Santra S., Zyryanov G.V., Majee A., Rusinov V.L., Chupakhin O.N.
ChemistrySelect,
2020
23.

Prokhorov A.M., Kozhevnikov D.N., Rusinov V.L., Chupakhin O.N., Glukhov I.V., Antipin M.Y., Kazheva O.N., Chekhlov A.N., Dyachenko O.A.
Organometallics,
2006
24.

Savchuk M.I., Khasanov A.F., Kopchuk D.S., Krinochkin A.P., Nikonov I.L., Starnovskaya E.S., Shtaitz Y.K., Kovalev I.S., Zyryanov G.V., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2019
25.
M. I. Savchuk, E. S. Starnovskaya, Y. K. Shtaitz, D. S. Kopchuk, E. V. Nosova, G. V. Zyryanov, V. L. Rusinov and O. N. Chupakhin, Russ. J. Gen. Chem., 2018, 88, 2213 (Zh. Obshch. Khim., 2018, 88, 1728).
26.

Kopchuk D.S., Chepchugov N.V., Kovalev I.S., Santra S., Rahman M., Giri K., Zyryanov G.V., Majee A., Charushin V.N., Chupakhin O.N.
RSC Advances,
2017
27.

Kopchuk D.S., Krinochkin A.P., Starnovskaya E.S., Shtaitz Y.K., Khasanov A.F., Taniya O.S., Santra S., Zyryanov G.V., Majee A., Rusinov V.L., Chupakhin O.N.
ChemistrySelect,
2018
28.
10.1016/j.mencom.2021.07.035_b0140
Starnovskaya
Proc.,
2019
29.
10.1016/j.mencom.2021.07.035_b0145
Kopchuk
Proc.,
2020
30.

Lintnerová L., Kováčiková L., Hanquet G., Boháč A.
Journal of Heterocyclic Chemistry,
2014
31.
CrysAlisPro, version 1.171.39.38a, Data Collection, Reduction and Correction Program, Rigaku Oxford Diffraction, 2017.
32.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
33.

Dondoni A., Fogagnolo M., Mastellari A., Pedrini P., Ugozzoli F.
Tetrahedron Letters,
1986
34.

Lindsey C.C., Pettus T.R.
Tetrahedron Letters,
2006
35.
![Thermally Controlled Decarboxylative [4 + 2] Cycloaddition between Alkoxyoxazoles and Acrylic Acid: Expedient Access to 3-Hydroxypyridines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Jouanno L., Tognetti V., Joubert L., Sabot C., Renard P.
Organic Letters,
2013
36.

Sharif S., Schagen D., Toney M.D., Limbach H.
Journal of the American Chemical Society,
2007
37.
10.1016/j.mencom.2021.07.035_h0185
Tolstikov
J. Org. Chem.,
1984
38.

Dean A., Ferlin M.G., Brun P., Castagliuolo I., Badocco D., Pastore P., Venzo A., Bombi G.G., Di Marco V.B.
Dalton Transactions,
2008
39.
10.1016/j.mencom.2021.07.035_b0190
Guda
Commun. Rev.,
2021
40.

Seitz G., Hoferichter R., Mohr R.
Archiv der Pharmazie,
1989
41.
10.1016/j.mencom.2021.07.035_b0200
Zhu
Patent US,
2019
42.
10.1016/j.mencom.2021.07.035_b0205
Weber
Patent WO,
2007
43.
T. Murata, S. Sakakibara, T. Yoshino, Y. Ikegami, T. Masuda, M. Shimada, T. Shintani, M. Shimazaki, T. B. Lowinger, K. B. Ziegelbauer, K. Fuchikami, M. Umeda, H. Komura and N. Yoshida, Patent WO 2002/44153 A1, 2002.
44.

Izuta S., Kosaka S., Kawai M., Miyano R., Matsuo H., Matsumoto A., Nonaka K., Takahashi Y., Ōmura S., Nakashima T.
Journal of Antibiotics,
2018
45.

Ding T., Yang L., Zhang W., Shen Y.
RSC Advances,
2019