Abstract
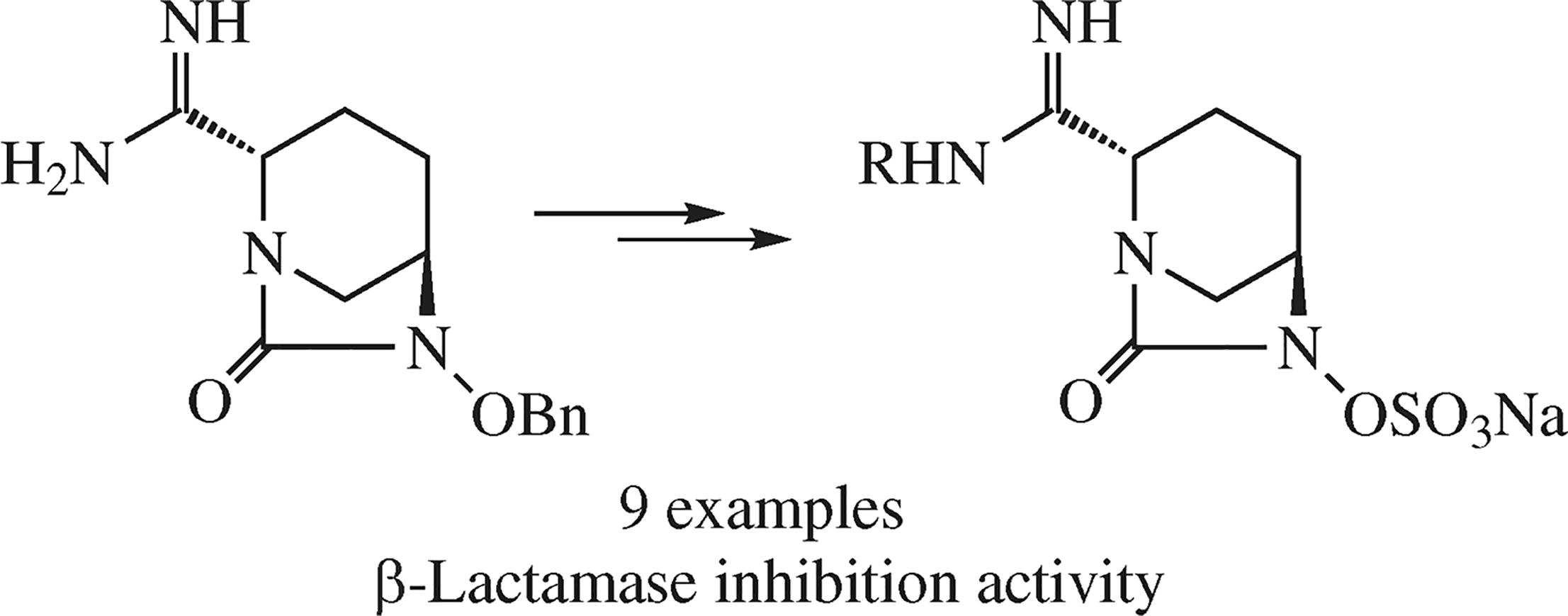
New avibactam derivatives in which amide moiety is replaced by substituted amidine groupings were obtained in several steps, the key step having involved the conversion of 2-positioned cyano group of the 1,6-diazabicyclo[3.2.1]-octane framework into the amidine one. Synergistic antibacterial activity of the new compounds in combination with meropenem infers the agonistic effect of these derivatives against β-lactamases.
References
1.

Palumbi S.R.
Science,
2001
2.

Davies J., Davies D.
Microbiology and Molecular Biology Reviews,
2010
3.

Walsh C.T., Wencewicz T.A.
Journal of Antibiotics,
2013
4.

Chellat M.F., Raguž L., Riedl R.
Angewandte Chemie - International Edition,
2016
5.

Eiamphungporn W., Schaduangrat N., Malik A., Nantasenamat C.
International Journal of Molecular Sciences,
2018
6.

Butler M.S., Paterson D.L.
Journal of Antibiotics,
2020
7.
10.1016/j.mencom.2021.07.020_b0035
Munita
Microbiol. Spectr.,
2016
8.

Peterson E., Kaur P.
Frontiers in Microbiology,
2018
9.

Bush K., Bradford P.A.
Cold Spring Harbor perspectives in medicine,
2016
10.

Morinaka A., Tsutsumi Y., Yamada M., Suzuki K., Watanabe T., Abe T., Furuuchi T., Inamura S., Sakamaki Y., Mitsuhashi N., Ida T., Livermore D.M.
Journal of Antimicrobial Chemotherapy,
2015
11.

Drawz S.M., Bonomo R.A.
Clinical Microbiology Reviews,
2010
12.

Shlaes D.M.
Annals of the New York Academy of Sciences,
2013
13.

Tehrani K.H., Martin N.I.
MedChemComm,
2018
14.

Gray D.A., Wenzel M.
ACS Infectious Diseases,
2020
15.
10.1016/j.mencom.2021.07.020_b0075
Uto
Clavulanic Acid,
2020
16.

Saudagar P.S., Survase S.A., Singhal R.S.
Biotechnology Advances,
2008
17.

Köck R., Cuny C.
Medizinische Klinik - Intensivmedizin und Notfallmedizin,
2018
18.
10.1016/j.mencom.2021.07.020_b0090
Nichols
Antimicrob. Agents Chemother.,
2018
19.

van Duin D., Bonomo R.A.
Clinical Infectious Diseases,
2016
20.

Rodriguez B.A., Girotto J.E., Nicolau D.P.
Current Pediatric Reviews,
2018
21.

22.

Gordon E.M., Duncton M.A., Gallop M.A.
Journal of Medicinal Chemistry,
2018
23.

Gao Y., Liu Y., Iqbal Z., Sun J., Ji J., Zhai L., Tang D., Ji J., He L., Mu Y., Yang H., Yang Z.
ChemistrySelect,
2021
24.
Z. Iqbal, L. Zhai, Y. Gao, D. Tang, X. Ma, J. Ji, J. Sun, J. Ji, Y. Liu, R. Jiang, Y. Mu, H. Yang and Z. Yang, Beilstein J. Org. Chem., Accepted.
25.

Yang H.Y., Tae J., Seo Y.W., Kim Y.J., Im H.Y., Choi G.D., Cho H., Park W., Kwon O.S., Cho Y.S., Ko M., Jang H., Lee J., Choi K., Kim C., et. al.
European Journal of Medicinal Chemistry,
2013
26.

Moss R.A., Ma W., Merrer D.C., Xue S.
Tetrahedron Letters,
1995
27.
10.1016/j.mencom.2021.07.020_b0135
Wikler
Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard – Eighth Edition, CLSI document M07–A8, Clinical and Laboratory Standards Institute,
2009