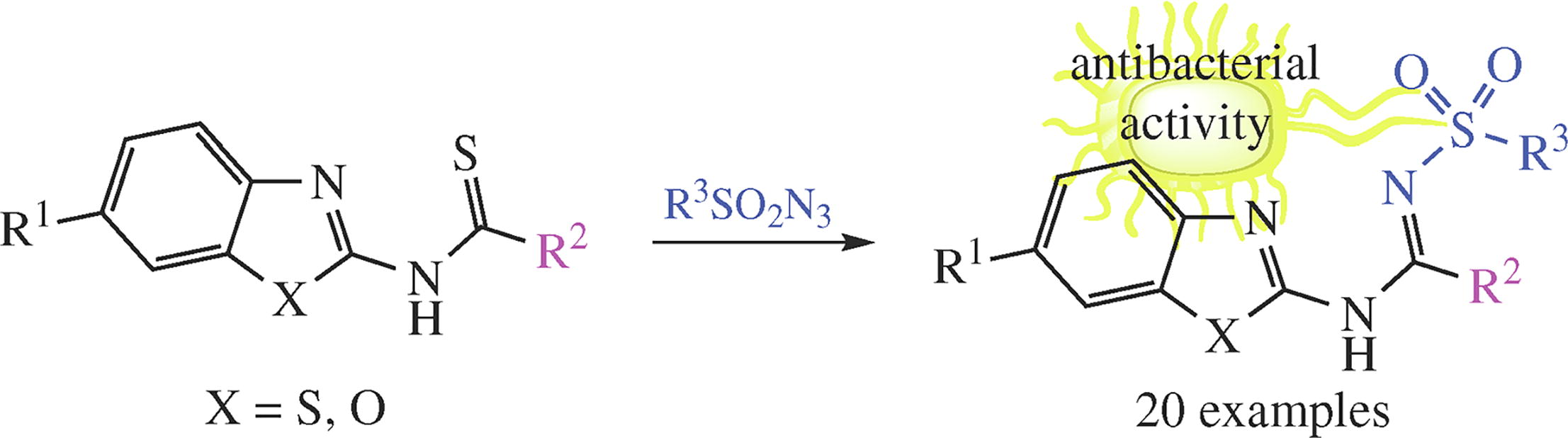Abstract
The reactions of N-(benz[d]oxazol-2-yl)- or N-(benzo[d]-thiazol-2-yl)-substituted carbothioamides with sulfonyl azides proceed as replacement of thioxo substituent by the sulfonylimino group to afford the corresponding N'-sulfonylated amidines. Acetic acid thioamides react smoothly upon boiling in ethanol, while for thioamides of trifluoroacetic and benzoic acids heating to 80–90 °C was required. Among hybrid molecules thus prepared, bacteriostatic-, bactericidal- and fungistatic-active against S. aureus and C. albicans representatives were found.
References
1.

Baud M.G., Bauer M.R., Verduci L., Dingler F.A., Patel K.J., Horil Roy D., Joerger A.C., Fersht A.R.
European Journal of Medicinal Chemistry,
2018
2.

Gardner C.R., Cheung B.B., Koach J., Black D.S., Marshall G.M., Kumar N.
Bioorganic and Medicinal Chemistry,
2012
3.

Li H., Wang X., Wang J., Shao T., Li Y., Mei Q., Lu S., Zhang S.
Bioorganic and Medicinal Chemistry,
2014
4.

Sharma P.C., Jain A., Yar M.S., Pahwa R., Singh J., Goel S.
Arabian Journal of Chemistry,
2015
5.

Tariq S., Kamboj P., Alam O., Amir M.
Bioorganic Chemistry,
2018
6.

Lee M.Y., Kim M.H., Kim J., Kim S.H., Kim B.T., Jeong I.H., Chang S., Kim S.H., Chang S.
Bioorganic and Medicinal Chemistry Letters,
2010
7.
10.1016/j.mencom.2021.07.019_b0025
Beryozkina
Synthesis,
2016
8.
Rupakova N.A., Bakulev V.A., Knippschild U., García-Reyes B., Eltsov O.S., Slesarev G.P., Beliaev N., Slepukhin P.A., Witt L., Peifer C., Beryozkina T.V.
Arkivoc,
2017
9.

Yavari I., Ahmadian S., Ghazanfarpur-Darjani M., Solgi Y.
Tetrahedron Letters,
2011
10.

He X., Shang Y., Hu J., Ju K., Jiang W., Wang S.
Science China Chemistry,
2011
11.

Yang T., Cui H., Zhang C., Zhang L., Su C.
Inorganic Chemistry,
2013
12.

Kim J., Stahl S.S.
Journal of Organic Chemistry,
2015
13.
Krstulović L., Ismaili H., Bajić M., Višnjevac A., Glavaš-Obrovac L., Žinić B.
Croatica Chemica Acta,
2012
14.

Fleury L.M., Wilson E.E., Vogt M., Fan T.J., Oliver A.G., Ashfeld B.L.
Angewandte Chemie - International Edition,
2013
15.

DeKorver K.A., Johnson W.L., Zhang Y., Hsung R.P., Dai H., Deng J., Lohse A.G., Zhang Y.
Journal of Organic Chemistry,
2011
16.

Chen S., Xu Y., Wan X.
Organic Letters,
2011
17.

Shafran Y.M., Silaichev P.S., Bakulev V.A.
Chemistry of Heterocyclic Compounds,
2019
18.

Chandna N., Chandak N., Kumar P., Kapoor J.K., Sharma P.K.
Green Chemistry,
2013
19.

Dianova L., Berseneva V., Beryozkina T., Efimov I., Kosterina M., Eltsov O., Dehaen W., Bakulev V.
European Journal of Organic Chemistry,
2015
20.

Ilkin V., Berseneva V., Beryozkina T., Glukhareva T., Dianova L., Dehaen W., Seliverstova E., Bakulev V.
Beilstein Journal of Organic Chemistry,
2020
21.
Beliaev N.A., Beryozkina T.V., Lubec G., Bakulev V.A.
Mendeleev Communications,
2019
22.

Efimov I., Beliaev N., Beryozkina T., Slepukhin P., Bakulev V.
Tetrahedron Letters,
2016
23.

Il’kin V.G., Berseneva V.S., Slepukhin P.А., Bakulev V.А.
Chemistry of Heterocyclic Compounds,
2018
24.

Hewitt R.J., Ong M.J., Lim Y.W., Burkett B.A.
European Journal of Organic Chemistry,
2015
25.

Augustin A.U., Sensse M., Jones P.G., Werz D.B.
Angewandte Chemie - International Edition,
2017
26.

Mlostoń G., Pipiak P., Linden A., Heimgartner H.
Helvetica Chimica Acta,
2015
27.
![Efficient synthesis of fluoroalkylated 1,4,2-oxathiazoles via regioselective [3 + 2]-cycloaddition of fluorinated nitrile oxides with thioketones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Mlostoń G., Kowalski M.K., Obijalska E., Heimgartner H.
Journal of Fluorine Chemistry,
2017
28.

Ullah Mughal E., Ayaz M., Hussain Z., Hasan A., Sadiq A., Riaz M., Malik A., Hussain S., Choudhary M.I.
Bioorganic and Medicinal Chemistry,
2006
29.
10.1016/j.mencom.2021.07.019_b0055
Padeyskaya
Infektsiya i Antimikrobnaya Terapiya,
2001
30.

Rex J.H., Walsh T.J., Sobel J.D., Filler S.G., Pappas P.G., Dismukes W.E., Edwards J.E.
Clinical Infectious Diseases,
2000