Abstract
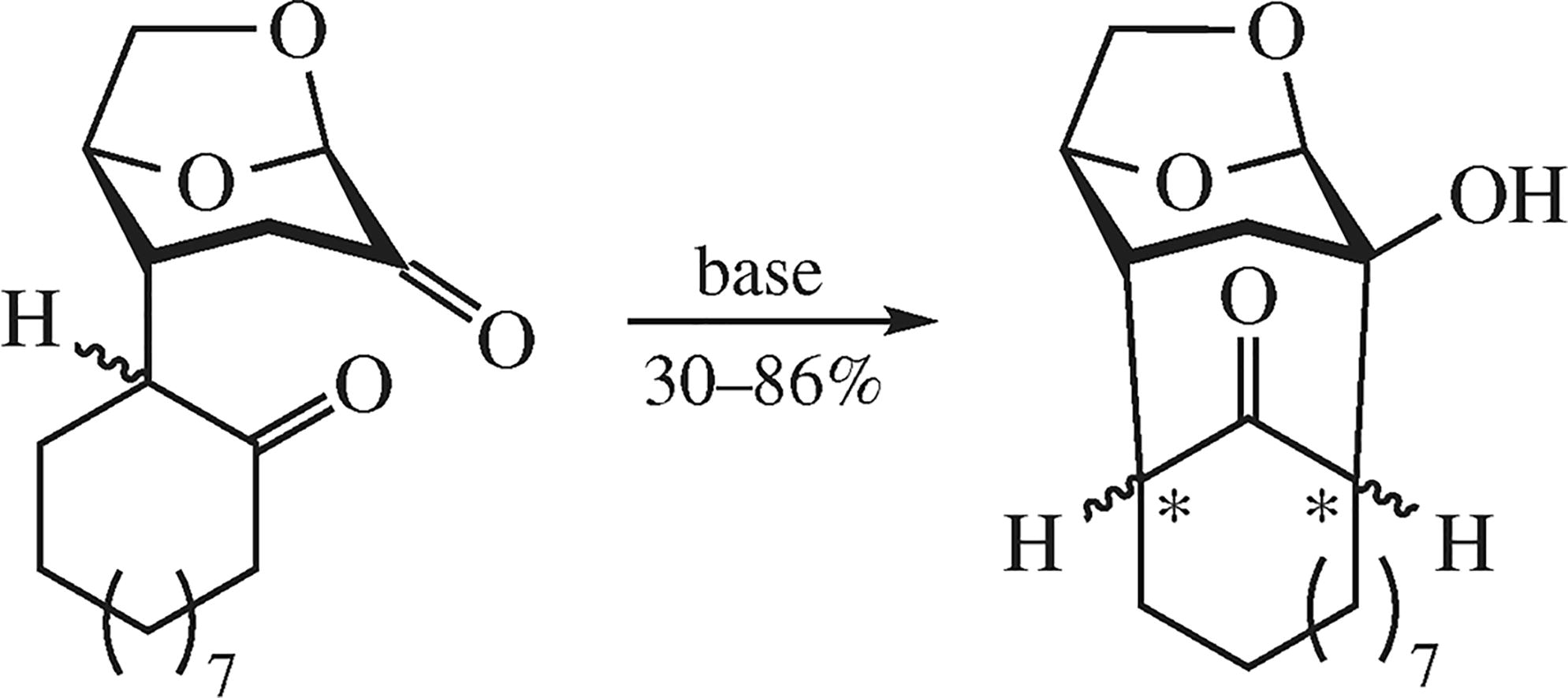
Base-promoted intramolecular aldol condensation of diastereomeric Michael adducts of levoglucosenone and cyclododecanone affords 12-hydroxy-14,20-dioxatetra-cyclo[9.6.1.112,17.113,16]icosan-18-one as a mixture of three diastereomers. The products thus obtained are promising for synthesizing 14-membered macrocyclic compounds, including cembranoids and their analogues.
References
1.

Cope A.C., Synerholm M.E.
Journal of the American Chemical Society,
1950
2.

Stork G., Landesman H.K.
Journal of the American Chemical Society,
1956
3.

Dauben W.G., McFarland J.W.
Journal of the American Chemical Society,
1960
4.

Frontier A.J., Danishefsky S.J., Koppel G.A., Meng D.
Tetrahedron,
1998
5.
![One-Pot Base-Promoted Tandem Michael Addition-Intramolecular Aldolization. Stereoselective Synthesis and Reactivity of 2-Hydroxybicyclo[3.2.1]octan-8-ones](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Filippini M., Faure R., Rodriguez J.
Journal of Organic Chemistry,
1995
6.
![Tandem Enamine Michael Additions to 4-(Mesyloxy)cyclopentenones: Bridged Tricyclic Skeletons via a Net [3 + 2] Construction1](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Gunawardena G.U., Arif A.M., West F.G.
Journal of the American Chemical Society,
1997
7.

Khalilova Y.A., Tagirov A.R., Doronina O.Y., Spirikhin L.V., Salikhov S.M., Valeev F.A.
Russian Journal of Organic Chemistry,
2014
8.

Faizullina L.K., Galimova Y.S., Salikhov S.M., Valeev F.A.
Chemistry of Heterocyclic Compounds,
2020
9.
Faizullina L.K., Galimova Y.S., Ovchinnikov M.Y., Salikhov S.M., Khursan S.L., Valeev F.A.
Mendeleev Communications,
2019
10.

Doronina O.Y., Khalilova Y.A., Sharipov B.T., Spirikhin L.V., Valeev F.A.
Russian Journal of Organic Chemistry,
2012
11.

Raldugin V.A., Shevtsov S.A.
Chemistry of Natural Compounds,
1987
12.

A. Paquette L., Bernardelli P.
Heterocycles,
1998