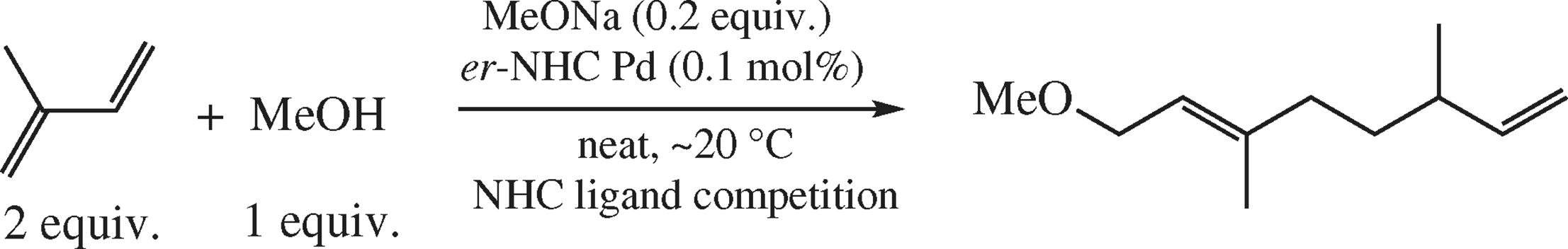Abstract
A comparative study of N-heterocyclic carbene Pd complexes in the head-to-head isoprene telomerization with methanol revealed significant impact of ligand structure as well as axial group structure on the catalyst activity. Some N,N'-diaryl substituted imidazol-2-ylidene, imidazolidin-2-ylidene and expanded-ring tetrahydropyrimidin-2-ylidene and tetrahydrodiazepin-2-ylidene based ligands were tested to explore the fundamental correlations between structure (ring carbene size along with the substituent sterical and electronic properties) and catalytic activity.
References
1.

Behr A., Becker M., Beckmann T., Johnen L., Leschinski J., Reyer S.
Angewandte Chemie - International Edition,
2009
2.

Clement N., Routaboul L., Grotevendt A., Jackstell R., Beller M.
Chemistry - A European Journal,
2008
3.

Cornils B.
Journal of Molecular Catalysis A Chemical,
1999
4.

Smutny E.J.
Journal of the American Chemical Society,
1967
5.

Takahashi S., Shibano T., Hagihara N.
Tetrahedron Letters,
1967
6.

Zapf A., Beller M.
Topics in Catalysis,
2002
7.

Messerle L.
Journal of the American Chemical Society,
1998
8.

van Leeuwen P.W., Clément N.D., Tschan M.J.
Coordination Chemistry Reviews,
2011
9.
Asghar U., Masoom A., Javed A., Abbas A.
American Journal of Chemical Engineering,
2020
10.
10.1016/j.mencom.2021.07.013_h0025
Zakharkin
Dokl. Akad. Nauk,
2001
11.
![Intermediates in the palladium-catalyzed reactions of 1,3-dienes. 1. (.eta.3,.eta.3-Dodecatrienediyl)palladium [Pd(.eta.3,.eta.3-C12H18)]](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Benn R., Jolly P.W., Mynott R., Schenker G.
Organometallics,
1985
12.

Jackstell R., Grotevendt A., Michalik D., El Firdoussi L., Beller M.
Journal of Organometallic Chemistry,
2007
13.
(a) E. A. Petrushkina, D. V. Khomishin, N. E. Mysova, A. V. Orlinkov, V. N. Kalinin, A. B. Ponomarev, I. A. Godovikov, Ya. V. Zubavichus and A. A. Veligzhanin, Moscow Univ. Chem. Bull., 2007, 62, 276 (Vestn. Mosk. Univ. Khim., 2007, 337)
14.
(b) E. A. Petrushkina and D. V. Khomishin, in Pheromones: Theories, Types and Uses, ed. I. M. Gregory, Nova Science Publishers, New York, 2010, pp. 125-164
15.

Maluenda I., Chen M., Guest D., Mark Roe S., Turner M.L., Navarro O.
Catalysis Science and Technology,
2015
16.

Faßbach T.A., Vorholt A.J., Leitner W.
ChemCatChem,
2019
17.

Maddock S.M., Finn M.G.
Organometallics,
2000
18.

Suslov D.S., Bykov M.V., Belova M.V., Abramov P.A., Tkach V.S.
Journal of Organometallic Chemistry,
2014
19.

Astakhov A.V., Soliev S.B., Chernyshev V.M.
Russian Chemical Bulletin,
2020
20.
10.1016/j.mencom.2021.07.013_h0050
N-Heterocyclic Carbenes in Transition Metal Catalysis and Organocatalysis,
2011
21.
10.1016/j.mencom.2021.07.013_h0055
N-Heterocyclic Carbenes: From Laboratory Curiosities to Efficient Synthetic Tools,
2010
22.
10.1016/j.mencom.2021.07.013_h0060
N-Heterocyclic Carbenes: Effective Tools for Organometallic Synthesis,
2014
23.

Kolychev E.L., Asachenko A.F., Dzhevakov P.B., Bush A.A., Shuntikov V.V., Khrustalev V.N., Nechaev M.S.
Dalton Transactions,
2013
24.

Morozov O.S., Lunchev A.V., Bush A.A., Tukov A.A., Asachenko A.F., Khrustalev V.N., Zalesskiy S.S., Ananikov V.P., Nechaev M.S.
Chemistry - A European Journal,
2014
25.

Azide-Alkyne Cycloaddition (CuAAC) in Alkane Solvents Catalyzed by Fluorinated NHC Copper(I) Complex
Topchiy M.A., Ageshina A.A., Gribanov P.S., Masoud S.M., Akmalov T.R., Nefedov S.E., Osipov S.N., Nechaev M.S., Asachenko A.F.
European Journal of Organic Chemistry,
2018
26.

Gribanov P.S., Chesnokov G.A., Topchiy M.A., Asachenko A.F., Nechaev M.S.
Organic and Biomolecular Chemistry,
2017
27.
Chesnokov G.A., Gribanov P.S., Topchiy M.A., Minaeva L.I., Asachenko A.F., Nechaev M.S., Bermesheva E.V., Bermeshev M.V.
Mendeleev Communications,
2017
28.
Gribanov P.S., Chesnokov G.A., Dzhevakov P.B., Kirilenko N.Y., Rzhevskiy S.A., Ageshina A.A., Topchiy M.A., Bermeshev M.V., Asachenko A.F., Nechaev M.S.
Mendeleev Communications,
2019