Keywords
1,2,3-dithiazoles
1,2,3-thiaselenazoles
1,2,5-chalcogenadiazoles
disulfur dichloride
exchange of chalcogen atoms
selenium dioxide
Abstract
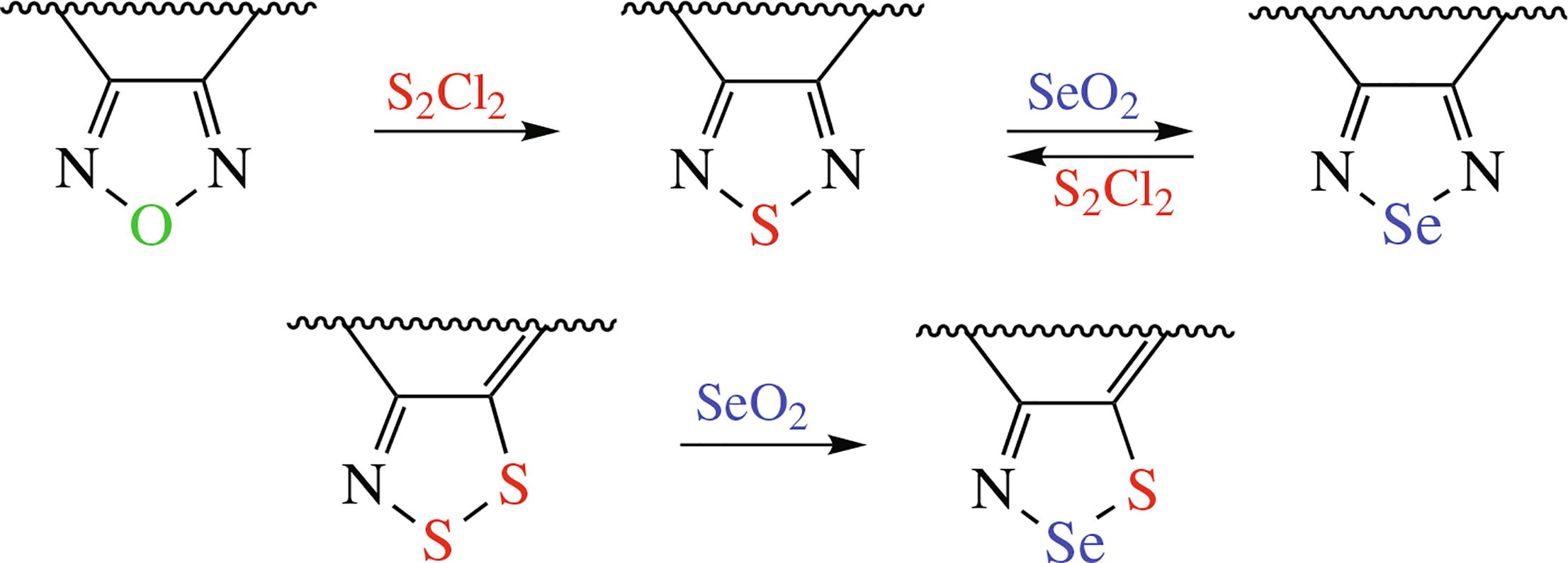
The procedures for synthesizing fused 1,2,5-chalcogenadi-azoles and 1,2,3-dithiazoles by direct exchange of one chalcogen atom for another, their scope and reaction pathways are discussed in this focus review.
References
1.
10.1016/j.mencom.2021.07.002_b0005
Yuryev
Zh. Obshch. Khim.,
1936
2.

Li Q., Xu Y., Liu C., Kim J.
Catalysis Letters,
2008
3.

Mashkina A.V.
Chemistry of Heterocyclic Compounds,
2010
4.
10.1016/j.mencom.2021.07.002_b0020
Zibarev
in Selenium and Tellurium Chemistry: From Small Molecules to Biomolecules and Materials, eds,
2011
5.

Gritsan N.P., Zibarev A.V.
Russian Chemical Bulletin,
2011
6.
10.1016/j.mencom.2021.07.002_b0030
Rakitin
J. Org. Chem.,
2018
7.
Chulanova E.A., Semenov N.A., Pushkarevsky N.A., Gritsan N.P., Zibarev A.V.
Mendeleev Communications,
2018
8.

Pushkarevsky N.A., Lonchakov A.V., Semenov N.A., Lork E., Buravov L.I., Konstantinova L.S., Silber G.T., Robertson N., Gritsan N.P., Rakitin O.A., Woollins J.D., Yagubskii E.B., Beckmann J., Zibarev A.V.
Synthetic Metals,
2012
9.

Konstantinova L.S., Knyazeva E.A., Obruchnikova N.V., Vasilieva N.V., Irtegova I.G., Nelyubina Y.V., Bagryanskaya I.Y., Shundrin L.A., Sosnovskaya Z.Y., Zibarev A.V., Rakitin O.A.
Tetrahedron,
2014
10.

Konstantinova L., Knyazeva E., Rakitin O.
Molecules,
2015
11.
Pedersen C.L., Lohse C., Poliakoff M., Matsutaka H., Matsuno T.
Acta Chemica Scandinavica,
1978
12.

Konstantinova L.S., Knyazeva E.A., Obruchnikova N.V., Gatilov Y.V., Zibarev A.V., Rakitin O.A.
Tetrahedron Letters,
2013
13.

Lonchakov A., Rakitin O., Gritsan N., Zibarev A.
Molecules,
2013
14.

Rakitin O.A., Konstantinova L.S.
Advances in Heterocyclic Chemistry,
2008
15.
Rakitin O.A.
Arkivoc,
2009
16.
Konstantinova L.S., Rakitin O.A.
Mendeleev Communications,
2009
17.

Konstantinova L.S., Rakitin O.A.
Russian Chemical Reviews,
2014
18.
Ul-Haq A., Donzello M.P., Stuzhin P.A.
Mendeleev Communications,
2007
19.

Konstantinova L.S., Knyazeva E.A., Rakitin O.A.
Organic Preparations and Procedures International,
2014
20.
10.1016/j.mencom.2021.07.002_b0100
Comprehensive Heterocyclic Chemistry III,
2008
21.

Rakitin O.A.
Tetrahedron Letters,
2020
22.
10.1016/j.mencom.2021.07.002_b0110
Comprehensive Heterocyclic Chemistry III,
2008
23.
10.1016/j.mencom.2021.07.002_b0115
Science of Synthesis,
2003
24.
10.1016/j.mencom.2021.07.002_b0120
Neto
Eur. J. Org. Chem.,
2013
25.

Rakitin O.А.
Chemistry of Heterocyclic Compounds,
2020
26.

Rakitin O.A.
Synthesis,
2019
27.

Suzuki T., Tsuji T., Okubo T., Okada A., Obana Y., Fukushima T., Miyashi T., Yamashita Y.
Journal of Organic Chemistry,
2001
28.
![Strong Charge-Transfer Chromophores from [2+2] Cycloadditions of TCNE and TCNQ to Peripheral Donor-Substituted Alkynes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Chen S., Li Y., Liu C., Yang W., Li Y.
European Journal of Organic Chemistry,
2011
29.

Tsubata Y., Suzuki T., Miyashi T., Yamashita Y.
Journal of Organic Chemistry,
1992
30.

Yang R., Tian R., Yan J., Zhang Y., Yang J., Hou Q., Yang W., Zhang C., Cao Y.
Macromolecules,
2004
31.
![Thieno[3,2-b]pyrrolo Donor Fused with Benzothiadiazolo, Benzoselenadiazolo and Quinoxalino Acceptors: Synthesis, Characterization, and Molecular Properties](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Cheng Y., Chen C., Ho Y., Chang S., Witek H.A., Hsu C.
Organic Letters,
2011
32.

Makarov A.G., Selikhova N.Y., Makarov A.Y., Malkov V.S., Bagryanskaya I.Y., Gatilov Y.V., Knyazev A.S., Slizhov Y.G., Zibarev A.V.
Journal of Fluorine Chemistry,
2014
33.
![Cyclic aryleneazachalcogenenes. Part III [1] synthesis of polyfluorinated 2,1,3-benzothia-(selena) diazoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Zibarev A.V., Miller A.O.
Journal of Fluorine Chemistry,
1990
34.

Prima D.O., Makarov A.G., Bagryanskaya I.Y., Kolesnikov A.E., Zargarova L.V., Baev D.S., Eliseeva T.F., Politanskaya L.V., Makarov A.Y., Slizhov Y.G., Zibarev A.V.
ChemistrySelect,
2019
35.

Semenov N.A., Radiush E.A., Chulanova E.A., Slawin A.M., Woollins J.D., Kadilenko E.M., Bagryanskaya I.Y., Irtegova I.G., Bogomyakov A.S., Shundrin L.A., Gritsan N.P., Zibarev A.V.
New Journal of Chemistry,
2019
36.
![[1,2,5]Selenadiazolo[3,4-c][1,2,5]thiadiazole and [1,2,5]Selenadiazolo[3,4-c][1,2,5]thiadiazolidyl – A Synthetic, Structural, and Theoretical Study](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bagryanskaya I.Y., Gatilov Y.V., Gritsan N.P., Ikorskii V.N., Irtegova I.G., Lonchakov A.V., Lork E., Mews R., Ovcharenko V.I., Semenov N.A., Vasilieva N.V., Zibarev A.V.
European Journal of Inorganic Chemistry,
2007
37.

Konstantinova L.S., Knyazeva E.A., Nefyodov A.A., Camacho P.S., Ashbrook S.E., Woollins J.D., Zibarev A.V., Rakitin O.A.
Tetrahedron Letters,
2015
38.
![Selenadiazolo[3,4-b]pyrazines: Synthesis from 3,4-Diamino-1,2,5-selenadiazole and Generation of Persistent Radical Anions](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Konstantinova L.S., Bobkova I.E., Nelyubina Y.V., Chulanova E.A., Irtegova I.G., Vasilieva N.V., Camacho P.S., Ashbrook S.E., Hua G., Slawin A.M., Woollins J.D., Zibarev A.V., Rakitin O.A.
European Journal of Organic Chemistry,
2015
39.

Makarov A.Y., Zhivonitko V.V., Makarov A.G., Zikirin S.B., Bagryanskaya I.Y., Bagryansky V.A., Gatilov Y.V., Irtegova I.G., Shakirov M.M., Zibarev A.V.
Inorganic Chemistry,
2011
40.

Barclay T.M., Cordes A.W., Goddard J.D., Mawhinney R.C., Oakley R.T., Preuss K.E., Reed R.W.
Journal of the American Chemical Society,
1997
41.
![Benzo[2,1-c:3,4-c‘]bis(1,2,3-thiaselenazole) (BSe) and Its Charge Transfer Chemistry. Crystal and Electronic Structure of [BSe]3[ClO4]2](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Beer L., Britten J.F., Cordes A.W., Clements O.P., Oakley R.T., Pink M., Reed R.W.
Inorganic Chemistry,
2001
42.

Winter S.M., Cvrkalj K., Dube P.A., Robertson C.M., Probert M.R., Howard J.A., Oakley R.T.
Chemical Communications,
2009
43.

Robertson C.M., Myles D.J., Leitch A.A., Reed R.W., Dooley B.M., Frank N.L., Dube P.A., Thompson L.K., Oakley R.T.
Journal of the American Chemical Society,
2007
44.

The effect of selenium incorporation on the bandwidth and conductivity of neutral radical conductors
Beer L., Brusso J.L., Haddon R.C., Itkis M.E., Oakley R.T., Reed R.W., Richardson J.F., Secco R.A., Yu X.
Chemical Communications,
2005
45.

Tse J.S., Leitch A.A., Yu X., Bao X., Zhang S., Liu Q., Jin C., Secco R.A., Desgreniers S., Ohishi Y., Oakley R.T.
Journal of the American Chemical Society,
2010
46.

Robertson C.M., Leitch A.A., Cvrkalj K., Myles D.J., Reed R.W., Dube P.A., Oakley R.T.
Journal of the American Chemical Society,
2008
47.

Brusso J.L., Cvrkalj K., Leitch A.A., Oakley R.T., Reed R.W., Robertson C.M.
Journal of the American Chemical Society,
2006
48.

Makarov A.Y., Blockhuys F., Bagryanskaya I.Y., Gatilov Y.V., Shakirov M.M., Zibarev A.V.
Inorganic Chemistry,
2013
49.

Thirunavukkuarasu K., Winter S.M., Beedle C.C., Kovalev A.E., Oakley R.T., Hill S.
Physical Review B,
2015
50.

Winter S.M., Hill S., Oakley R.T.
Journal of the American Chemical Society,
2015
51.

Winter S.M., Oakley R.T., Kovalev A.E., Hill S.
Physical Review B,
2012
52.

Winter S.M., Datta S., Hill S., Oakley R.T.
Journal of the American Chemical Society,
2011
53.

Mito M., Komorida Y., Tsuruda H., Tse J.S., Desgreniers S., Ohishi Y., Leitch A.A., Cvrkalj K., Robertson C.M., Oakley R.T.
Journal of the American Chemical Society,
2009
54.

Leitch A.A., Brusso J.L., Cvrkalj K., Reed R.W., Robertson C.M., Dube P.A., Oakley R.T.
Chemical Communications,
2007
55.
Volkova Y.M., Makarov A.Y., Pritchina E.A., Gritsan N.P., Zibarev A.V.
Mendeleev Communications,
2020
56.

Beer L., Brusso J.L., Haddon R.C., Itkis M.E., Kleinke H., Leitch A.A., Oakley R.T., Reed R.W., Richardson J.F., Secco R.A., Yu X.
Journal of the American Chemical Society,
2005
57.

�fros L.S., Strelets B.K., Akulin Y.I.
Chemistry of Heterocyclic Compounds,
1976
58.

Konstantinova L.S., Baranovsky I.V., Pritchina E.A., Mikhailov M.S., Bagryanskaya I.Y., Semenov N.A., Irtegova I.G., Salnikov G.E., Lyssenko K.A., Gritsan N.P., Zibarev A.V., Rakitin O.A.
Chemistry - A European Journal,
2017
59.
10.1016/j.mencom.2021.07.002_b0295
Greenwood
Chemistry of the Elements,
1997
60.
10.1016/j.mencom.2021.07.002_b0300
Baranovsky
Russian Academy of Sciences, Moscow,
2019
61.
![Selenium for sulfur substitution in a thiazyl ring: identification of the phenyl-selenathiadiazolylium cation, [PhCNSeSN]+](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Less R.J., Rawson J.M., Jones M.
Polyhedron,
2001
62.

Taponen A.I., Wong J.W., Lekin K., Assoud A., Robertson C.M., Lahtinen M., Clérac R., Tuononen H.M., Mailman A., Oakley R.T.
Inorganic Chemistry,
2018
63.

Structural Studies of Perfluoroaryldiselenadiazolyl Radicals: Insights into Dithiadiazolyl Chemistry
Melen R.L., Less R.J., Pask C.M., Rawson J.M.
Inorganic Chemistry,
2016
64.
![Prototypal 1,2,3,5-dithia- and -diselenadiazolyl [HCN2E2].bul. (E = sulfur, selenium): molecular and electronic structures of the radicals and their dimers, by theory and experiment](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Cordes A.W., Bryan C.D., Davis W.M., de Laat R.H., Glarum S.H., Goddard J.D., Haddon R.C., Hicks R.G., Kennepohl D.K.
Journal of the American Chemical Society,
1993