Keywords
aryl bromides
arylation
cross-coupling
diaryl ethers
Pd-catalysis
phenols
solvent-free reactions
Abstract
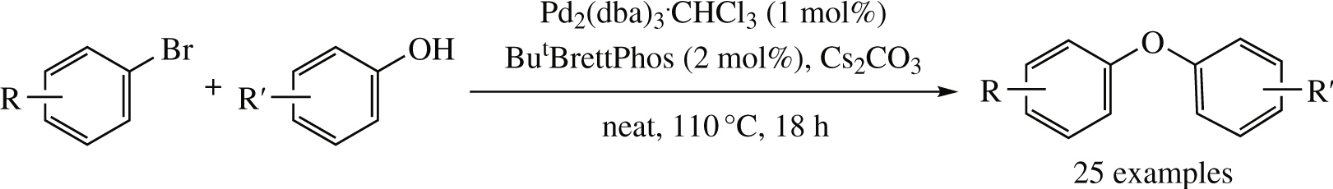
A new solvent-free procedure for C–O cross-coupling between phenols and aryl bromides comprising of Pd2(dba)3/ButBrettPhos catalytic system is efficient for substrates bearing donor or acceptor, as well as bulky substituents.
References
1.

2.

Jung M.E., Rohloff J.C.
Journal of Organic Chemistry,
1985
3.

Singh S.B., Pettit G.R.
Journal of Organic Chemistry,
1990
4.

Deshpande V.H., Gokhale N.J.
Tetrahedron Letters,
1992
5.

6.

Scott Sawyer J.
Tetrahedron,
2000
7.

ZENITANI S., TASHIRO S., SHINDO K., NAGAI K., SUZUKI K., IMOTO M.
Journal of Antibiotics,
2003
8.

Cristau P., Vors J., Zhu J.
Tetrahedron,
2003
9.

Wolfe J.P., Wagaw S., Marcoux J., Buchwald S.L.
Accounts of Chemical Research,
1998
10.

Hartwig J.F.
Angewandte Chemie - International Edition,
1998
11.

Yang B.H., Buchwald S.L.
Journal of Organometallic Chemistry,
1999
12.

Prim D., Campagne J., Joseph D., Andrioletti B.
Tetrahedron,
2002
13.
10.1016/j.mencom.2021.04.042_sbref0010e
Muci
Cross-Coupling Reactions: A Practical Guide,
2002
14.

Beletskaya I.P., Cheprakov A.V.
Coordination Chemistry Reviews,
2004
15.

Bedford R.B., Cazin C.S., Holder D.
Coordination Chemistry Reviews,
2004
16.

Aranyos A., Old D.W., Kiyomori A., Wolfe J.P., Sadighi J.P., Buchwald S.L.
Journal of the American Chemical Society,
1999
17.
10.1016/j.mencom.2021.04.042_sbref0020a
Akkoç
Synlett,
2008
18.

Gowrisankar S., Sergeev A.G., Anbarasan P., Spannenberg A., Neumann H., Beller M.
Journal of the American Chemical Society,
2010
19.

Anastas P., Eghbali N.
Chemical Society Reviews,
2010
20.

Porcheddu A., Colacino E., De Luca L., Delogu F.
ACS Catalysis,
2020
21.

Andrade M.A., Martins L.M.
Molecules,
2020
22.

Sherwood J., Clark J.H., Fairlamb I.J., Slattery J.M.
Green Chemistry,
2019
23.

Beletskaya I.P., Alonso F., Tyurin V.
Coordination Chemistry Reviews,
2019
24.
Artamkina G.A., Ermolina M.V., Beletskaya I.P.
Mendeleev Communications,
2003
25.

Gajare A.S., Toyota K., Yoshifuji M., Ozawa F.
Journal of Organic Chemistry,
2004
26.

Topchiy M.A., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2014
27.

Reilly S.W., Mach R.H.
Organic Letters,
2016
28.

Shao Q., Jiang Z., Su W.
Tetrahedron Letters,
2018
29.

Kubota K., Seo T., Koide K., Hasegawa Y., Ito H.
Nature Communications,
2019
30.

Cao Q., Nicholson W.I., Jones A.C., Browne D.L.
Organic and Biomolecular Chemistry,
2019
31.

Bernhardt F., Trotzki R., Szuppa T., Stolle A., Ondruschka B.
Beilstein Journal of Organic Chemistry,
2010
32.

Asachenko A.F., Sorochkina K.R., Dzhevakov P.B., Topchiy M.A., Nechaev M.S.
Advanced Synthesis and Catalysis,
2013
33.

Seo T., Ishiyama T., Kubota K., Ito H.
Chemical Science,
2019
34.

Pentsak E.O., Ananikov V.P.
European Journal of Organic Chemistry,
2019
35.

Dzhevakov P.B., Topchiy M.A., Zharkova D.A., Morozov O.S., Asachenko A.F., Nechaev M.S.
Advanced Synthesis and Catalysis,
2016
36.

Liang Y., Xie Y., Li J.
Journal of Organic Chemistry,
2005
37.

Díaz-Ortiz Á., Prieto P., Vázquez E.
Synlett,
1997
38.

39.

Gribanov P.S., Golenko Y.D., Topchiy M.A., Minaeva L.I., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2018
40.
Chesnokov G.A., Gribanov P.S., Topchiy M.A., Minaeva L.I., Asachenko A.F., Nechaev M.S., Bermesheva E.V., Bermeshev M.V.
Mendeleev Communications,
2017
41.

Chesnokov G.A., Topchiy M.A., Dzhevakov P.B., Gribanov P.S., Tukov A.A., Khrustalev V.N., Asachenko A.F., Nechaev M.S.
Dalton Transactions,
2017
42.
Gribanov P.S., Golenko Y.D., Topchiy M.A., Philippova A.N., Kirilenko N.Y., Krivoshchapov N.V., Sterligov G.K., Asachenko A.F., Bermeshev M.V., Nechaev M.S.
Mendeleev Communications,
2018
43.

Ageshina A.A., Sterligov G.K., Rzhevskiy S.A., Topchiy M.A., Chesnokov G.A., Gribanov P.S., Melnikova E.K., Nechaev M.S., Asachenko A.F., Bermeshev M.V.
Dalton Transactions,
2019
44.
Gribanov P.S., Chesnokov G.A., Dzhevakov P.B., Kirilenko N.Y., Rzhevskiy S.A., Ageshina A.A., Topchiy M.A., Bermeshev M.V., Asachenko A.F., Nechaev M.S.
Mendeleev Communications,
2019
45.

Rzhevskiy S.A., Ageshina A.A., Chesnokov G.A., Gribanov P.S., Topchiy M.A., Nechaev M.S., Asachenko A.F.
RSC Advances,
2019
46.

Burgos C.H., Barder T.E., Huang X., Buchwald S.L.
Angewandte Chemie - International Edition,
2006
47.

Topchiy M.A., Dzhevakov P.B., Rubina M.S., Morozov O.S., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2016
48.

Salvi L., Davis N.R., Ali S.Z., Buchwald S.L.
Organic Letters,
2011