Keywords
bathochromic shift
cationic fragment
indolinium salts
merocyanine
molecular switch
photochromism
spiropyrans
Abstract
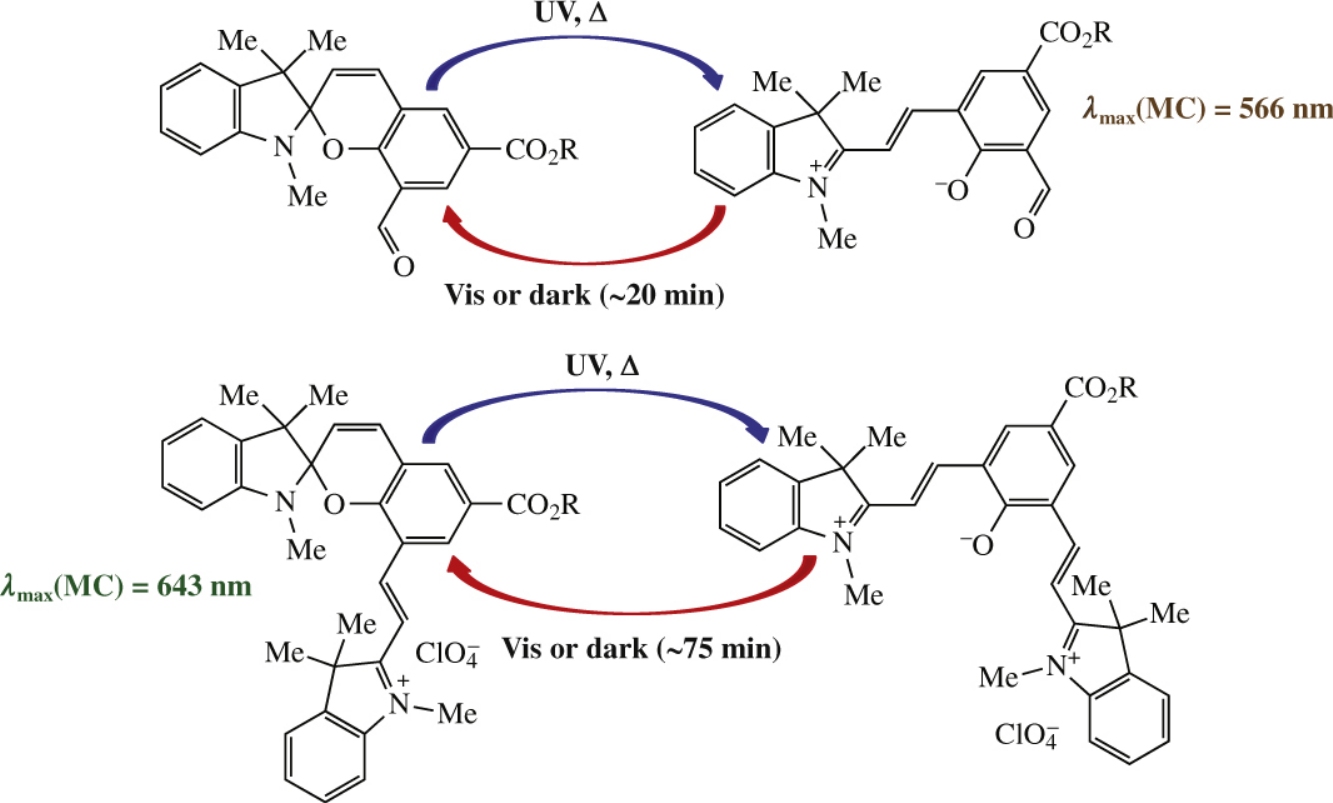
New spiropyrans with chromene and indoline cores bearing alkoxycarbonyl group and cationic π-acceptor in the chromene moiety were synthesized in one step from 1,2,3,3-tetramethylindolinium and isophthalic dialdehyde derivatives. The appending of a 8′-positioned conjugated cationic fragment causes a significant red-shift of the photoinduced isomers absorption maxima and provides them with extremely long lifetimes up to 75min. The obtained spiropyrans exhibit both positive and negative photochromism in solutions, demonstrating the properties of ‘photochromic balance’.
References
1.

2.

Nikolaeva O.G., Karlutova O.Y., Tikhomirova K.S., Revinskii Y.V., Dorogan I.V., Dubonosov A.D., Bren V.A., Metelitsa A.V., Minkin V.I.
Russian Chemical Bulletin,
2019
3.

Balmond E.I., Tautges B.K., Faulkner A.L., Or V.W., Hodur B.M., Shaw J.T., Louie A.Y.
Journal of Organic Chemistry,
2016
4.

Dorogan I.V., Minkin V.I.
Chemistry of Heterocyclic Compounds,
2016
5.

Liu F., Morokuma K.
Journal of the American Chemical Society,
2013
6.

Wang P., Bai F., Zhang Z., Wang Y., Wang J., Zhang H.
Organic Electronics,
2017
7.

Bénard S., Rivière E., Yu P., Nakatani K., Delouis J.F.
Chemistry of Materials,
2000
8.

Aldoshin S.M., Sanina N.A.
Russian Chemical Reviews,
2016
9.

Yoshida J., Kida N., Okazawa A., Kojima N.
Polyhedron,
2013
10.

Compain J., Deniard P., Dessapt R., Dolbecq A., Oms O., Sécheresse F., Marrot J., Mialane P.
Chemical Communications,
2010
11.

Chai X., Han H., Sedgwick A.C., Li N., Zang Y., James T.D., Zhang J., Hu X., Yu Y., Li Y., Wang Y., Li J., He X., Tian H.
Journal of the American Chemical Society,
2020
12.

Gao M., Shen B., Zhou J., Kapre R., Louie A.Y., Shaw J.T.
ACS Omega,
2020
13.

Velema W.A., Szymanski W., Feringa B.L.
Journal of the American Chemical Society,
2014
14.

Frolova L.A., Rezvanova A.A., Lukyanov B.S., Sanina N.A., Troshin P.A., Aldoshin S.M.
Journal of Materials Chemistry C,
2015
15.

Minkin V.I.
Russian Chemical Bulletin,
2008
16.
Tuktarov A.R., Salikhov R.B., Khuzin A.A., Safargalin I.N., Mullagaliev I.N., Venidiktova O.V., Valova T.M., Barachevsky V.A., Dzhemilev U.M.
Mendeleev Communications,
2019
17.
V. I. Minkin, M.B. Lukyanova, B.S. Lukyanov, I.V. Ozhogin, A.D. Pugachev, O.A. Komissarova and E. L. Mukhanov, Patent RU 2627358 C1, 2017.
18.

Tkachev V.V., Lukyanova M.B., Lukyanov B.S., Pugachev A.D., Aldoshin S.M., Minkin V.I.
Journal of Structural Chemistry,
2016
19.

Pugachev A.D., Lukyanova M.B., Lukyanov B.S., Ozhogin I.V., Kozlenko A.S., Rostovtseva I.A., Makarova N.I., Tkachev V.V., Aksenov N.A.
Journal of Molecular Structure,
2019
20.

Pugachev A.D., Ozhogin I.V., Lukyanova M.B., Lukyanov B.S., Rostovtseva I.A., Dorogan I.V., Makarova N.I., Tkachev V.V., Metelitsa A.V., Aldoshin S.M.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2020
21.
![Interaction of 6′-methoxycarbonyl-1,3,3-trimethyl-indolino-2,2′-[2H]chromene with hydrazine hydrate](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Lukyanova M.V., Kogan V.A., Lukyanov B.S., Alekseenko Y.S.
Chemistry of Heterocyclic Compounds,
2007
22.

Laptev A.V., Lukin A.Y., Belikov N.E., Zvezdin K.V., Demina O.V., Barachevsky V.A., Varfolomeev S.D., Khodonov A.A., Shvets V.I.
Russian Chemical Bulletin,
2014
23.

Aiken S., Edgar R.J., Gabbutt C.D., Heron B.M., Hobson P.A.
Dyes and Pigments,
2018
24.

Hobley J., Malatesta V., Millini R., Montanari L., O Neil Parker, Jr W.
Physical Chemistry Chemical Physics,
1999
25.

Hobley J., Pfeifer-Fukumura U., Bletz M., Asahi T., Masuhara H., Fukumura H.
Journal of Physical Chemistry A,
2002
26.

Schulz-Senft M., Gates P.J., Sönnichsen F.D., Staubitz A.
Dyes and Pigments,
2017
27.

Abdullah A., Nevell T.G., Sammes P.G., Roxburgh C.J.
Dyes and Pigments,
2015
28.

Minami M., Taguchi N.
Chemistry Letters,
1996
29.

Toppet S., Quintens W., Smets G.
Tetrahedron,
1975
30.
![Synthesis and structure of indoline spiropyrans based on benzo[f]coumarin](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Nikolaeva O.G., Karlutova O.Y., Guseva A.A., Gaeva E.B., Starikov A.G., Dubonosov A.D., Bren V.A., Metelitsa A.V., Minkin V.I.
Russian Chemical Bulletin,
2020
31.

Metelitsa A., Chernyshev A., Voloshin N., Solov'eva E., Rostovtseva I., Dorogan I., Gaeva E., Guseva A.
Dyes and Pigments,
2021