Keywords
arylboronic acids
biaryls
Buchwald ligands
cross-coupling
polychloroarenes
Abstract
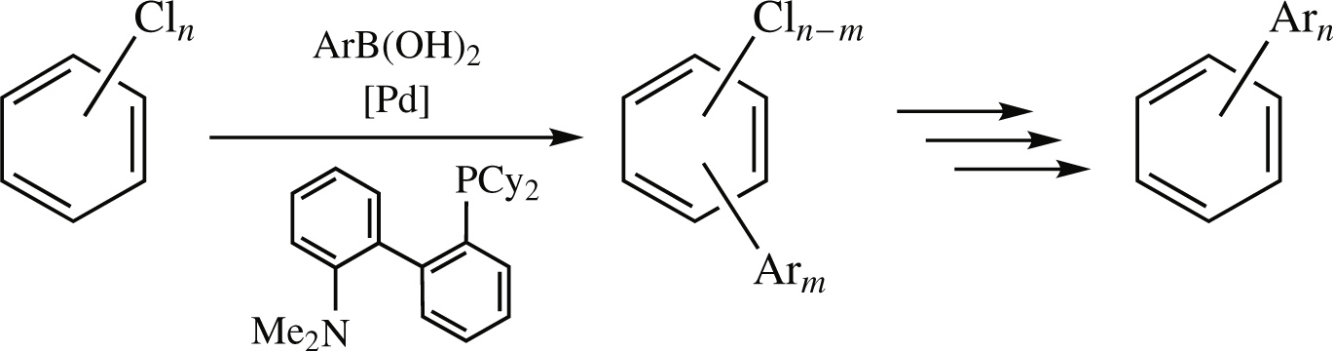
Screening of four Buchwald ligands for the cross-coupling of isomeric di-, tri- and tetrachlorobenzenes with arylboronic acids revealed that good yields of exhaustive substitution can be best provided by 2-dicyclohexylphosphino-2′-(dimethylamino) biphenyl (DavePHOS).
References
1.

Hoke J.B., Gramiccioni G.A., Balko E.N.
Applied Catalysis B: Environmental,
1992
2.

Murena F.
Applied Catalysis B: Environmental,
2000
3.

Ukisu Y., Iimura S., Uchida R.
Chemosphere,
1996
4.

5.
10.1016/j.mencom.2021.04.039_sbref0010a
Polychloroaromatic Compounds,
1974
6.

Gorbunova T.I., Saloutin V.I., Chupakhin O.N.
Russian Chemical Reviews,
2010
7.

Vasil'ev A.A., Burukin A.S., Zlotin S.G.
Russian Chemical Reviews,
2007
8.

Burukin A.S., Vasil'ev A.A., Chizhov A.O., Zlotin S.G.
Russian Chemical Bulletin,
2005
9.
![Cross-coupling of polychlorobenzenes with phenylboronic acid in the presence of [Pd]-imidazolium salts as catalytic systems](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Burukin A.S., Vasil’ev A.A., Merkulova N.L., Chizhov A.O., Mistryukov E.A., Zlotin S.G.
Russian Chemical Bulletin,
2007
10.

Burukin A.S., Vasil’ev A.A., Merkulova N.L., Struchkova M.I., Zlotin S.G.
Russian Chemical Bulletin,
2006
11.
A. S. Burukin, PhD Thesis, 2006.
12.

Wolfe J.P., Singer R.A., Yang B.H., Buchwald S.L.
Journal of the American Chemical Society,
1999
13.

Wolfe J.P., Tomori H., Sadighi J.P., Yin J., Buchwald S.L.
Journal of Organic Chemistry,
2000
14.

Milne J.E., Buchwald S.L.
Journal of the American Chemical Society,
2004
15.

Catalysts for Suzuki−Miyaura Coupling Processes: Scope and Studies of the Effect of Ligand Structure
Barder T.E., Walker S.D., Martinelli J.R., Buchwald S.L.
Journal of the American Chemical Society,
2005
16.

Martin R., Buchwald S.L.
Accounts of Chemical Research,
2008
17.

Lee D., Jin M.
Organic Letters,
2010
18.

Tu T., Sun Z., Fang W., Xu M., Zhou Y.
Organic Letters,
2012
19.

Reimann S., Ehlers P., Sharif M., Spannenberg A., Langer P.
Tetrahedron,
2016
20.
Kuchurov I.V., Vasil’ev A.A., Zlotin S.G.
Mendeleev Communications,
2010
21.
Gribanov P.S., Chesnokov G.A., Dzhevakov P.B., Kirilenko N.Y., Rzhevskiy S.A., Ageshina A.A., Topchiy M.A., Bermeshev M.V., Asachenko A.F., Nechaev M.S.
Mendeleev Communications,
2019
22.

Soliev S.B., Astakhov A.V., Pasyukov D.V., Chernyshev V.M.
Russian Chemical Bulletin,
2020
23.
Verbitskiy E.V., Dinastiya E.M., Eltsov O.S., Zhilina E.F., Schepochkin A.V., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Mendeleev Communications,
2020
24.
Matyugina E.S., Khandazhinskaya A.L., Kochetkov S.N., Seley-Radtke K.L.
Mendeleev Communications,
2020