Keywords
1,3-dipolar cycloaddition
alkynes
iron nitrate
isoxazoles
isoxazolines
nitration
Abstract
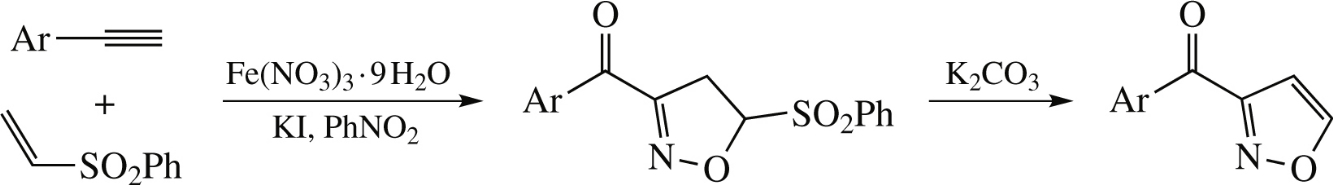
Iron(III) nitrate-assisted cycloaddition of (phenylsulfonyl)ethene to arylacetylenes in the presence of KI affords 5-phenylsulfonyl-3-aroylisoxazolines whose treatment with K2CO3 provides 4,5-unsubstituted 3-aroylisoxazoles. Both synthetic steps can be performed in a one-pot manner.
References
1.
![[(3-Chlorophenyl)piperazinylpropyl]pyridazinones and Analogues as Potent Antinociceptive Agents](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Giovannoni M.P., Vergelli C., Ghelardini C., Galeotti N., Bartolini A., Dal Piaz V.
Journal of Medicinal Chemistry,
2003
2.

Kaffy J., Pontikis R., Carrez D., Croisy A., Monneret C., Florent J.
Bioorganic and Medicinal Chemistry,
2006
3.

Kamal A., Surendranadha Reddy J., Janaki Ramaiah M., Dastagiri D., Vijaya Bharathi E., Ameruddin Azhar M., Sultana F., Pushpavalli S.N., Pal-Bhadra M., Juvekar A.
European Journal of Medicinal Chemistry,
2010
4.

Kamal A., Bharathi E.V., Reddy J.S., Ramaiah M.J., Dastagiri D., Reddy M.K., Viswanath A., Reddy T.L., Shaik T.B., Pushpavalli S.N., Bhadra M.P.
European Journal of Medicinal Chemistry,
2011
5.

Rück-Braun K., Freysoldt T.H., Wierschem F.
Chemical Society Reviews,
2005
6.
Tsyganov D.V., Semenova M.N., Konyushkin L.D., Ushkarov V.I., Raihstat M.M., Semenov V.V.
Mendeleev Communications,
2019
7.

Arai M.A., Kuraishi M., Arai T., Sasai H.
Journal of the American Chemical Society,
2001
8.

Fuller A.A., Chen B., Minter A.R., Mapp A.K.
Journal of the American Chemical Society,
2005
9.

Jiang D., Peng J., Chen Y.
Organic Letters,
2008
10.

Kozikowski A.P., Stein P.D.
Journal of the American Chemical Society,
1982
11.

Scott J.P., Oliver S.F., Brands K.M., Brewer S.E., Davies A.J., Gibb A.D., Hands D., Keen S.P., Sheen F.J., Reamer R.A., Wilson R.D., Dolling U.
Journal of Organic Chemistry,
2006
12.
13.

Han B., Yang X., Fang R., Yu W., Wang C., Duan X., Liu S.
Angewandte Chemie - International Edition,
2012
14.

Gangadhara Chary R., Rajeshwar Reddy G., Ganesh Y.S., Vara Prasad K., Raghunadh A., Krishna T., Mukherjee S., Pal M.
Advanced Synthesis and Catalysis,
2014
15.

Dai P., Tan X., Luo Q., Yu X., Zhang S., Liu F., Zhang W.
Organic Letters,
2019
16.

Guo X., Xu G., Zhou L., Yan H., Hao X., Wang Q.
Organic Chemistry Frontiers,
2020
17.

Zhang X., Hu W., Chen S., Hu X.
Organic Letters,
2018
18.

Wang X., Zhu L., Liu P., Wang X., Yuan H., Zhao Y.
Journal of Organic Chemistry,
2019
19.

Gasparrini F., Giovannoli M., Misiti D., Natile G., Palmieri G., Maresca L.
Journal of the American Chemical Society,
1993
20.

Rogachev V.O., Filimonov V.D., Yusubov M.S.
Russian Journal of Organic Chemistry,
2001
21.

Sau P., Santra S.K., Rakshit A., Patel B.K.
Journal of Organic Chemistry,
2017
22.

Zhang K., Jelier B., Passera A., Jeschke G., Katayev D.
Chemistry - A European Journal,
2019
23.

Li Y., Gao M., Liu B., Xu B.
Organic Chemistry Frontiers,
2017
24.

Lai Z., Li Z., Liu Y., Yang P., Fang X., Zhang W., Liu B., Chang H., Xu H., Xu Y.
Journal of Organic Chemistry,
2017
25.
![Copper Nitrate Mediated Regioselective [2+2+1] Cyclization of Alkynes with Alkenes: A Cascade Approach to Δ2-Isoxazolines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Gao M., Li Y., Gan Y., Xu B.
Angewandte Chemie - International Edition,
2015
26.

Bian Q., Wu C., Yuan J., Shi Z., Ding T., Huang Y., Xu H., Xu Y.
Journal of Organic Chemistry,
2020
27.

Kai H., Tomida M., Nakai T., Takase A.
Heterocycles,
2002