Keywords
2,2′:6′,2″-terpyridines
heterocyclization
organofluorine compounds
pyridines
regioisomerism
β-aminovinyl ketones
Abstract
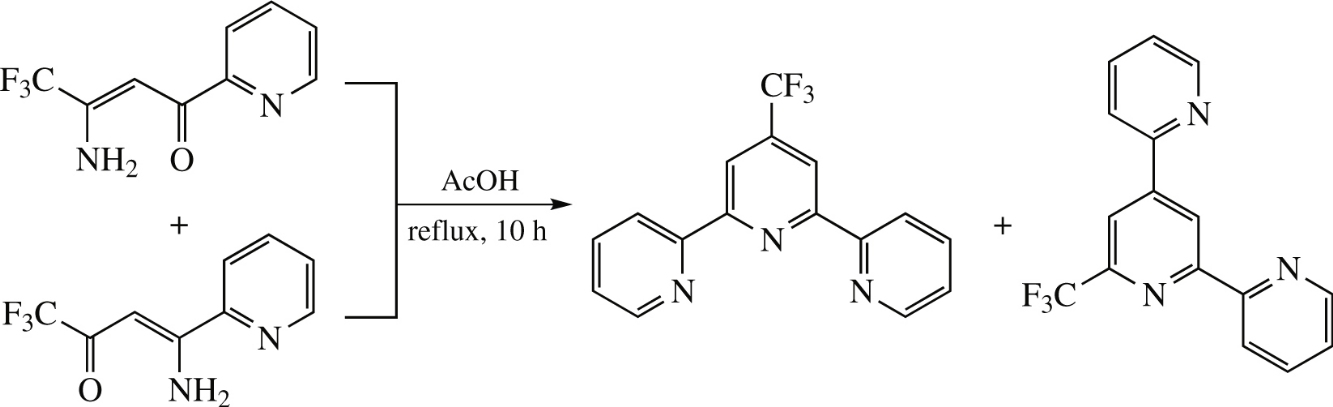
Reflux of two isomeric 3-amino-4,4,4-trifluoro-1-(2-pyridyl)but-2-en-1-one and 3-amino-1,1,1-trifluoro-4-(2-pyridyl)-but-3-en-2-one in acetic acid affords 4′-trifluoromethyl-2,2′:6′,2″-terpyridine (37%) with 1.4% admixture of 6′-trifluoromethyl-2,2′:4′,2″-terpyridine.
References
1.
10.1016/j.mencom.2021.04.035_bib0005
Schubert
Terpyridine-based Materials: For Catalytic, Optoelectronic and Life Science Applications,
2011
2.

Fallahpour R.
Synthesis,
2003
3.

Hofmeier H., Schubert U.S.
Chemical Society Reviews,
2004
4.

Winter A., Newkome G.R., Schubert U.S.
ChemCatChem,
2011
5.

Wild A., Winter A., Schlütter F., Schubert U.S.
Chemical Society Reviews,
2011
6.

Jiang T., Polizzi N.F., Rawson J., Therien M.J.
Journal of the American Chemical Society,
2017
7.
![Photomagnetic studies on spin-crossover solid solutions containing two different metal complexes, [Fe(1-bpp)2]x[M(terpy)2]1−x[BF4]2 (M = Ru or Co)](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Chastanet G., Tovee C.A., Hyett G., Halcrow M.A., Létard J.
Dalton Transactions,
2012
8.

Tokarev K.L., Kiskin M.A., Sidorov A.A., Aleksandrov G.G., Ikorskii V.N., Suzdalev I.P., Novotortsev V.M., Eremenko I.L.
Russian Chemical Bulletin,
2008
9.
10.1016/j.mencom.2021.04.035_bib0045
Sivasankari
J. Chem. Pharm. Res.,
2018
10.

Chiper M., Hoogenboom R., Schubert U.S.
Macromolecular Rapid Communications,
2009
11.

Shunmugam R., Gabriel G.J., Aamer K.A., Tew G.N.
Macromolecular Rapid Communications,
2010
12.

Husson J., Knorr M.
Journal of Heterocyclic Chemistry,
2012
13.

Zhao Y., Newton J.N., Liu J., Wei A.
Langmuir,
2009
14.

Joshi-Pangu A., Ganesh M., Biscoe M.R.
Organic Letters,
2011
15.
Filyakova V.I., Boltacheva N.S., Slepukhin P.A., Pervova M.G., Charushin V.N.
Mendeleev Communications,
2020
16.

Slepukhin P.A., Boltacheva N.S., Pervova M.G., Filyakova V.I., Charushin V.N.
Russian Chemical Bulletin,
2020
17.

Filyakova V.I., Pashkevich K.I., Postovskii I.Y.
Russian Chemical Bulletin,
1981
18.
10.1016/j.mencom.2021.04.035_bib0095
Filyakova
J. Org. Chem. USSR (Engl. Transl.),
1989
19.

Filyakova V.I., Yufit D.S., Struchkov Y.T., Pashkevich K.I.
Russian Chemical Bulletin,
1990