Keywords
[1,2,4]triazolo[4,3-a]pyrimidinones
Antibacterial activity
heterocyclization
hybrid molecules
MRSA inhibitors
pyrimidinones
Smiles-type reaction
thieno[2,3-b]pyridines
VRE inhibitors
Abstract
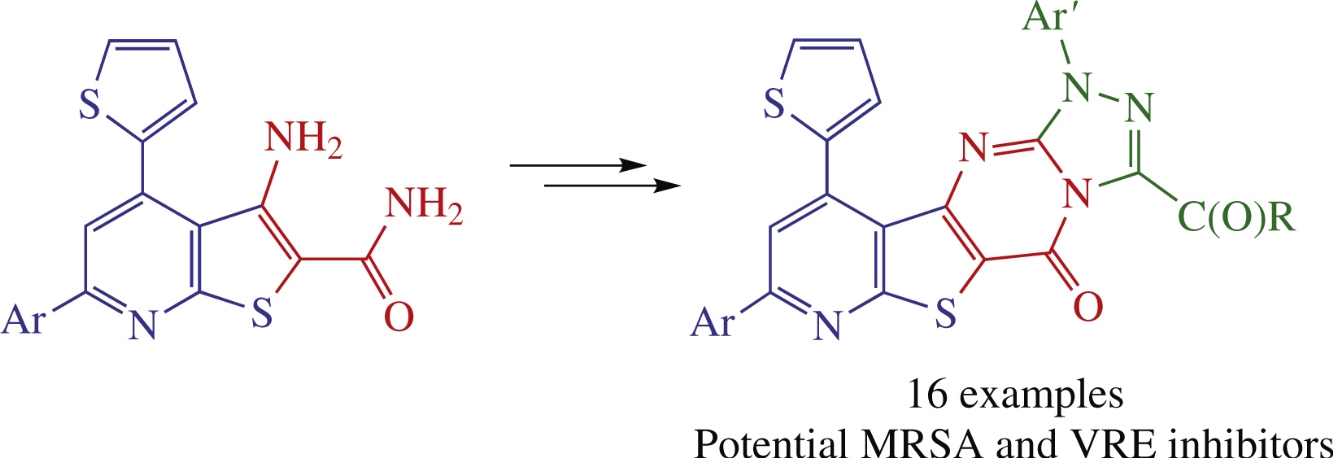
New thieno[2,3-b]pyridine-fused [1,2,4]triazolo[4,3-a]pyrimidinones were obtained by the [5+1] heterocyclization of the appropriate thieno[2,3-b]pyridines followed by the Smiles-type reaction protocol. In general, the triazoles tested exhibited better antibacterial activity against Gram-positive than against Gram-negative bacterial strains. Compoundscontaining 3-acetyl-1-(4-methoxyphenyl)[1,2,4]triazole unit demonstrated more potent inhibitory activities than the reference Linezolid.
References
1.

Gao F., Wang P., Yang H., Miao Q., Ma L., Lu G.
European Journal of Medicinal Chemistry,
2018
2.

Mekky A.E., Sanad S.M.
Bioorganic Chemistry,
2020
3.
Antibiotic Resistance: Key Facts, World Health Organization, https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance.
4.

Sanad S.M., Ahmed A.A., Mekky A.E.
Archiv der Pharmazie,
2020
5.

Rossiter S.E., Fletcher M.H., Wuest W.M.
Chemical Reviews,
2017
6.

Kaproń B., Łuszczki J.J., Płazińska A., Siwek A., Karcz T., Gryboś A., Nowak G., Makuch-Kocka A., Walczak K., Langner E., Szalast K., Marciniak S., Paczkowska M., Cielecka-Piontek J., Ciesla L.M., et. al.
European Journal of Pharmaceutical Sciences,
2019
7.
Paronikyan E.G., Dashyan S.S., Mamyan S.S.
Mendeleev Communications,
2020
8.
![Three-component green synthesis of 6-ethoxycarbonyl-5-methyl-7-(thien-2-yl)-4,7-dihydro[1,2,4]triazolo[1,5-a]pyrimidine, a promising antituberculosis drug](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Titova Y.A., Fedorova O.V., Rusinov G.L., Charushin V.N.
Russian Chemical Bulletin,
2019
9.

Identification of 1,2,4-triazoles as new thymidine phosphorylase inhibitors: Future anti-tumor drugs
Shahzad S.A., Yar M., Khan Z.A., Shahzadi L., Naqvi S.A., Mahmood A., Ullah S., Shaikh A.J., Sherazi T.A., Bale A.T., Kukułowicz J., Bajda M.
Bioorganic Chemistry,
2019
10.

El-Sherief H.A., Youssif B.G., Abbas Bukhari S.N., Abdelazeem A.H., Abdel-Aziz M., Abdel-Rahman H.M.
European Journal of Medicinal Chemistry,
2018
11.

Wittine K., Stipković Babić M., Makuc D., Plavec J., Kraljević Pavelić S., Sedić M., Pavelić K., Leyssen P., Neyts J., Balzarini J., Mintas M.
Bioorganic and Medicinal Chemistry,
2012
12.
![Synthesis of some novel thiourea derivatives obtained from 5-[(4-aminophenoxy)methyl]-4-alkyl/aryl-2,4-dihydro-3H-1,2,4-triazole-3-thiones and evaluation as antiviral/anti-HIV and anti-tuberculosis agents](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Küçükgüzel İ., Tatar E., Küçükgüzel Ş.G., Rollas S., De Clercq E.
European Journal of Medicinal Chemistry,
2008
13.

Zhang S., Xu Z., Gao C., Ren Q., Chang L., Lv Z., Feng L.
European Journal of Medicinal Chemistry,
2017
14.

Jin R., Zeng C., Liang X., Sun X., Liu Y., Wang Y., Zhou S.
Bioorganic Chemistry,
2018
15.

Synthesis and antimicrobial activities of novel quinoline derivatives carrying 1,2,4-triazole moiety
Eswaran S., Adhikari A.V., Shetty N.S.
European Journal of Medicinal Chemistry,
2009
16.

Fan Y., Ke X., Liu M.
Journal of Heterocyclic Chemistry,
2018
17.
![Synthesis and Characterization of New Heterocyclic Compounds Containing Thienylbenzo[h]Quinoline Moiety](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Al-Taifi E.A., Abbady M.S., Bakhite E.A.
Journal of Heterocyclic Chemistry,
2015
18.
![REACTIONS OF STYRYLTHIENYL KETONE, STYRYL FURYL KETONE WITH THIOCYANOACETAMIDE: SYNTHESIS OF SEVERAL NEW PYRIDINES, THIENO[2,3-b]PYRIDINES, PYRIDO [2′,3′:4,5]THIENO[3,2-c]PYRIDAZINES AND PYRIDO-[3′,2′:4,5]THIENO[3,2-d]PYRIMIDIN ONE DERIVATIVES](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Attaby F.A.
Phosphorus, Sulfur and Silicon and the Related Elements,
1998
19.

Purushothaman B., Arumugam P., Kulsi G., Song J.M.
European Journal of Medicinal Chemistry,
2018
20.

Gao F., Wang T., Xiao J., Huang G.
European Journal of Medicinal Chemistry,
2019
21.
![Synthesis, Characterization and Antibacterial Activity of Some New 1,2,4- Triazole Schiff bases and 1,2,4-triazolo[3,4-b][1,3,4]thiadiazines](/storage/images/resized/doUaFUZdxUEQjLi1TwZjGHi8HXYNWWSk04dSC6Xh_small_thumb.webp)
Sim K., Chan P., Boo X., Heng K., Lye K., Teo K.
Letters in Organic Chemistry,
2018
22.
![Structural Elucidation and Antimicrobial Evaluation of Novel [1,2,4]Triazolo[4,3-a]pyrimidines and Pyrido[2,3-d][1,2,4]triazolo[4,3-a]pyrimidinones](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Gomha S.M., Mohamed A.M., Zaki Y.H., Ewies M.M., Elroby S.A.
Journal of Heterocyclic Chemistry,
2018
23.
GOMHA S., ABDALLAH M., ABD EL-AZIZ M., SERAG N.
Turkish Journal of Chemistry,
2016
24.
![Synthesis and antitumor activity of pyrido [2,3-d]pyrimidine and pyrido[2,3-d] [1,2,4]triazolo[4,3-a]pyrimidine derivatives that induce apoptosis through G1 cell-cycle arrest](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fares M., Abou-Seri S.M., Abdel-Aziz H.A., Abbas S.E., Youssef M.M., Eladwy R.A.
European Journal of Medicinal Chemistry,
2014
25.
![Piperazine‐mediated tandem synthesis of bis(thieno[2,3‐ b ]pyridines): Versatile precursors for related fused [1,2,4]triazolo[4,3‐ a ]pyrimidines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Sanad S.M., Mekky A.E.
Journal of Heterocyclic Chemistry,
2020
26.

Sanad S.M., Ahmed A.A., Mekky A.E.
Journal of Heterocyclic Chemistry,
2019
27.
ABBAS I., GOMHA S., ELNEAIRY M., ELAASSER M., MABROUK B.
Turkish Journal of Chemistry,
2015
28.

Holden C.M., Greaney M.F.
Chemistry - A European Journal,
2017
29.
![Synthesis and in vitro activity of novel 1,2,4-triazolo[4,3-a]pyrimidine oxazolidinone antibacterial agents](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Khera M.K., Cliffe I.A., Mathur T., Prakash O.
Bioorganic and Medicinal Chemistry Letters,
2011
30.
![Synthesis, cytotoxicity, in-vitro antibacterial screening and in-silico study of novel thieno[2,3-b]pyridines as potential pim-1 inhibitors](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Mekky A.E., Sanad S.M., Said A.Y., Elneairy M.A.
Synthetic Communications,
2020
31.
![Utility of Pyridine‐2(1 H )‐thiones in the Synthesis of Novel Bis‐Thieno[2,3‐ b ]pyridines and Their Fused Azines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Sanad S.M., Abdel‐Fattah A.M., Attaby F.A., Elneairy M.A.
Journal of Heterocyclic Chemistry,
2019
32.
![Efficient Synthesis and Characterization of Novel Pyrido[3′,2′:4,5]thieno[3,2‐ d ]pyrimidines and Their Fused [1,2,4]triazole Derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Sanad S.M., Hawass M.A., Ahmed A.A., Elneairy M.A.
Journal of Heterocyclic Chemistry,
2018