Keywords
antimicrobial activity
benzofuroxan-5-olates
hydrolysis
isothiuronium salts
nitroarenes
X-ray diffraction
Abstract
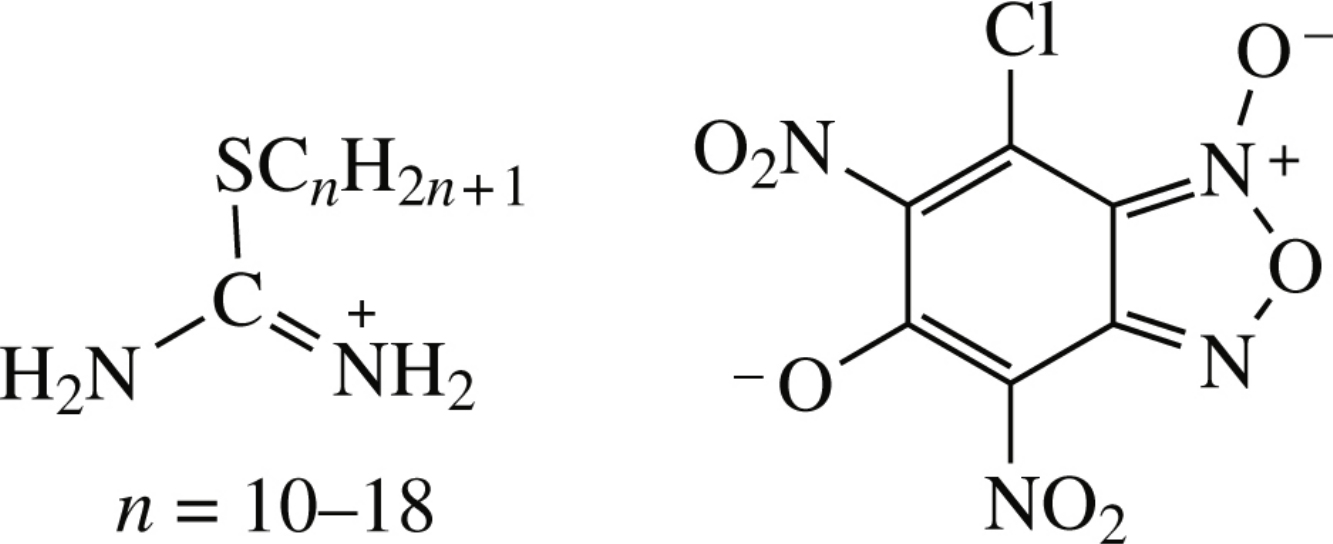
New thermally stable long-chained 2-alkylisothiuronium 7-chloro-4,6-dinitrobenzofuroxan-5-olates were obtained from the corresponding bromides and 5,7-dichloro-4,6-dinitrobenzofuroxan, the hydrolysis of the C(5)–Cl bond to produce phenolic function having occurred in the course of the process. The compound structure was determined by IR spectroscopy, elemental analysis and X-ray single crystal study. Salts with C14–C18 alkyl groups revealed moderate antibacterial and antifungal activities.
References
1.

MU L., FENG S., GO M.L.
Chemical and Pharmaceutical Bulletin,
2011
2.

Galkina I.V., Takhautdinova G.L., Ivshin K.A., Yusupova L.M., Krasnyuk I.I., Egorova S.N., Shulaeva M.P., Pozdeev O.K., Kataeva O.N., Galkin V.I.
Russian Journal of General Chemistry,
2017
3.

Galkina I.V., Tudriy E.V., Kataeva O.N., Yusupova L.M., Luftmann H., Galkin V.I.
Phosphorus, Sulfur and Silicon and the Related Elements,
2009
4.

Galkina I.V., Yusupova L.M., Gubaidullin A.T., Galkin V.I.
Russian Journal of Organic Chemistry,
2016
5.

Galkina I.V., Takhautdinova G.L., Tudrii E.V., Yusupova L.M., Falyakhov I.F., Pozdeev O.K., Shulaeva M.P., Kipenskaya L.V., Sakhibullina V.G., Krivolapov D.B., Litvinov I.A., Galkin V.I., Cherkasov R.A.
Russian Journal of Organic Chemistry,
2013
6.

Bohn H., Brendel J., Martorana P.A., Schönafinger K.
British Journal of Pharmacology,
1995
7.

Feelisch M., Schönafingeri K., Noack H.
Biochemical Pharmacology,
1992
8.

Macphee D.G., Robert G.P., Ternai B., Ghosh P., Stephens R.
Chemico-Biological Interactions,
1977
9.

Cerecetto H., Di Maio R., González M., Risso M., Saenz P., Seoane G., Denicola A., Peluffo G., Quijano C., Olea-Azar C.
Journal of Medicinal Chemistry,
1999
10.

Zarranz B., Jaso A., Aldana I., Monge A.
Bioorganic and Medicinal Chemistry,
2003
11.

Chugunova E., Akylbekov N., Bulatova A., Gavrilov N., Voloshina A., Kulik N., Zobov V., Dobrynin A., Syakaev V., Burilov A.
European Journal of Medicinal Chemistry,
2016
12.

Panov A.A., Lakatosh S.A., Kubbutat M.H., Dezhenkova L.G., Totzke F., Schechtel K.
Chemistry of Heterocyclic Compounds,
2019
13.

Chugunova E.A., Gazizov A.S., Burilov A.R., Yusupova L.M., Pudovik M.A., Sinyashin O.G.
Russian Chemical Bulletin,
2019
14.

Bandelin F.J., Tuschhoff J.V.
Journal of the American Chemical Society,
1952
15.

Brooks J.D., Charlton P.T., Macey P.E., Peak D.A., Short W.F.
Journal of the Chemical Society (Resumed),
1950
16.

Bella M.D., Tait A., Parenti C., Bondi M., Quaglio G.
Archiv der Pharmazie,
1986
17.
10.1016/j.mencom.2021.04.027_bib0085
Keera
J. Sci. Ind. Res.,
2002
18.

Cohen S., Laitman I., Lublin Tennenbaum T., Natan M., Banin E., Margel S.
Polymers for Advanced Technologies,
2016
19.

Hefni H.H., Negm N.A.
Journal of Surfactants and Detergents,
2013
20.

Badawi A.M., Azzam E.M., Morsy S.M.
Bioorganic and Medicinal Chemistry,
2006
21.

Negm N.A., Mohamed A.S., Ahmed S.M., El-Raouf M.A.
Journal of Surfactants and Detergents,
2015
22.

Ferreira M., Assunção L.S., Silva A.H., Filippin-Monteiro F.B., Creczynski-Pasa T.B., Sá M.M.
European Journal of Medicinal Chemistry,
2017
23.
Alexeev A.A., Nurieva E.V., Trofimova T.P., Chesnakova E.A., Grishin Y.K., Lyssenko K.A., Filimonova M.V., Zefirova O.N.
Mendeleev Communications,
2019
24.

Valeeva F.G., Karimova T.R., Pavlov R.V., Bakhtiyarov D.I., Sapunova A.S., Ivshin K.A., Kataeva O.N., Gaynanova G.A., Syakaev V.V., Voloshina A.D., Galkina I.V., Latypov S.K., Zakharova L.Y.
Journal of Molecular Liquids,
2021
25.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
26.
10.1016/j.mencom.2021.04.027_bib0130
Sheldrick
Acta Crystallogr.,
2015
27.
10.1016/j.mencom.2021.04.027_bib0135
Sheldrick
Acta Crystallogr.,
2007
28.

Macrae C.F., Edgington P.R., McCabe P., Pidcock E., Shields G.P., Taylor R., Towler M., van de Streek J.
Journal of Applied Crystallography,
2006