Keywords
5,11-dioxa-2,3a,4,6,6b,8,9a,10,12,12bdecaazadicyclopenta[e,l]pyrenes
aminals
aza-acetalization
Brønsted acids
cytotoxic activity
furazans
heterocyclization
Polycycles
Abstract
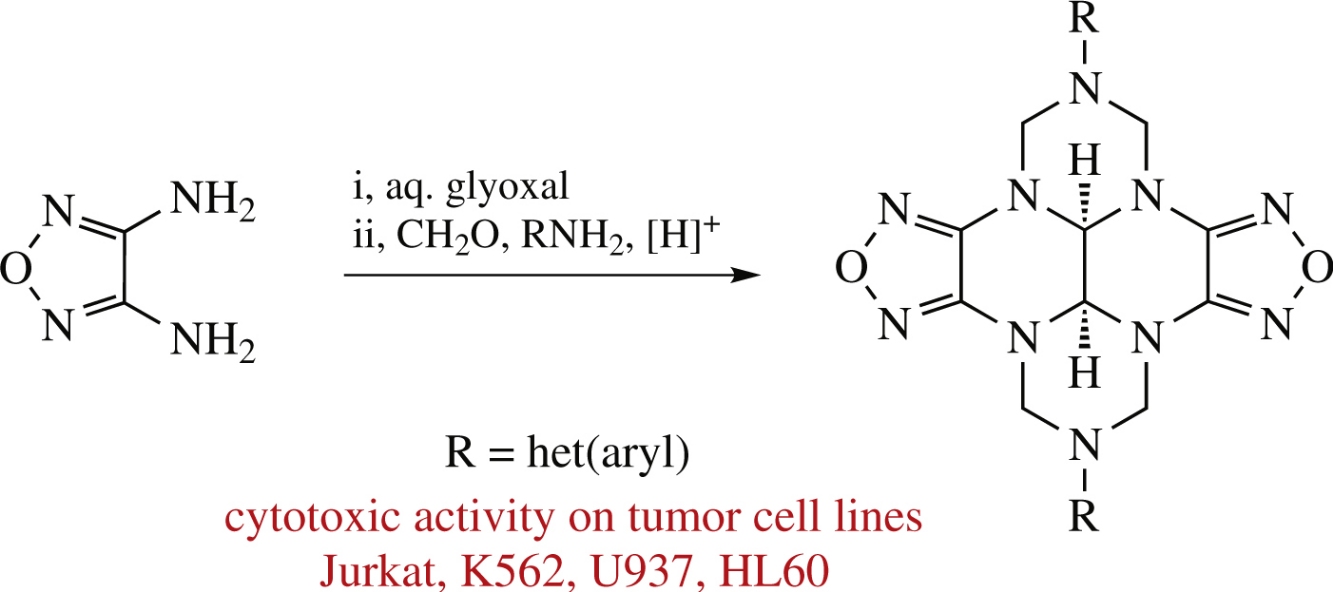
New 2,8-di(het)aryl-containing dioxadecaazadicyclopenta[e,l]pyrenes, furazan-annulated polyaza polycycles, have been synthesized by acid-catalyzed cyclocondensation of difurazanotetraazadecalin with formaldehyde and primary (het)arylamines. The cytotoxic activity of these compounds against tumor cell lines (Jurkat, K562, U937, HL60) has been estimated.
References
1.

Strizhenko K.V., Vasil’ev L.S., Suponitsky K.Y., Sheremetev A.B.
Chemistry of Heterocyclic Compounds,
2020
2.
10.1016/j.mencom.2021.04.026_bib0010
Roy
J. Mater. Sci.: Mater. Electron.,
2001
3.
Yu. S. Andreichenkov, D.D. Nekrasov, B.A. Bargteil and B. S. Zalesov, Patent RU 1042321, 1997.
4.
A. B. Sheremetev, V.L. Betin, I.L. Yudin, V.O. Kulagina, Yu. V. Khropov, T.V. Bulargina and A. Ya. Kots, Patent RU 2167161, 2001.
5.

Cabrera M., López G.V., Gómez L.E., Breijo M., Pintos C., Botti H., Raymondo S., Vettorazzi A., Ceráin A.L., Monge A., Rubbo H., González M., Cerecetto H.
Drug and Chemical Toxicology,
2011
6.

Cameron A., Read J., Tranter R., Winter V.J., Sessions R.B., Brady R.L., Vivas L., Easton A., Kendrick H., Croft S.L., Barros D., Lavandera J.L., Martin J.J., Risco F., García-Ochoa S., et. al.
Journal of Biological Chemistry,
2004
7.

Sravya G., Yamini G., Padmavathi V., Padmaja A.
European Journal of Medicinal Chemistry,
2016
8.
10.1016/j.mencom.2021.04.026_bib0040
Babu
ARKIVOC,
2006
9.

Schiefer I.T., VandeVrede L., Fa’ M., Arancio O., Thatcher G.R.
Journal of Medicinal Chemistry,
2012
10.
Sheremetev A.B., Dmitriev D.E., Lagutina N.K., Raihstat M.M., Kiselyov A.S., Semenova M.N., Ikizalp N.N., Semenov V.V.
Mendeleev Communications,
2010
11.

Meunier B.
Accounts of Chemical Research,
2007
12.

Rakhimova E.B., Kirsanov V.Y., Mescheryakova E.S., Khalilov L.M., Ibragimov A.G., Dzhemileva L.U., D’yakonov V.A., Dzhemilev U.M.
ACS Medicinal Chemistry Letters,
2019
13.

Chen J., Liang D., Xiao W.
Synthesis,
2020
14.

Rakhimova E.B., Ismagilov R.A., Meshcheryakova E.S., Khalilov L.M., Ibragimov A.G., Dzhemilev U.M.
Tetrahedron Letters,
2014
15.

Rakhimova E.B., Kirsanov V.Y., Meshcheryakova E.S., Khalilov L.M., Kutepov B.I., Ibragimov A.G., Dzhemilev U.M.
Tetrahedron,
2017
16.

Rakhimova E., Kirsanov V., Mescheryakova E., Khalilov L., Ibragimov A., Dzhemilev U.
Synlett,
2018
17.

Rakhimova E.B., Kirsanov V.Y., Tret'yakova E.V., Khalilov L.M., Ibragimov A.G., Dzhemileva L.U., D'yakonov V.A., Dzhemilev U.M.
RSC Advances,
2020
18.
![Synthesis and chemistry of some furazano- and furoxano[3,4-b]piperazines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Willer R.L., Moore D.W.
Journal of Organic Chemistry,
1985
19.

Rakhimova E.B., Kirsanov V.Y., Ibragimov A.G., Dzhemilev U.M.
Russian Journal of Organic Chemistry,
2018
20.
Rakhimova E.B., Kirsanov V.Y., Mescheryakova E.S., Ibragimov A.G., Dzhemilev U.M.
Mendeleev Communications,
2020
21.
CrysAlis PRO/2012, Agilent Ltd., Yarnton, Oxfordshire, England, 2012.
22.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
23.
10.1016/j.mencom.2021.04.026_bib0115
Sheldrick
Acta Crystallogr.,
2015
24.
10.1016/j.mencom.2021.04.026_bib0120
Sheldrick
Acta Crystallogr.,
2015
25.

Brown I.D.
Acta Crystallographica Section A,
1976