Keywords
antiproliferative activity
copper catalysis
iodoalkenes
MTT assay
nitrogen heterocycles
steroids
Abstract
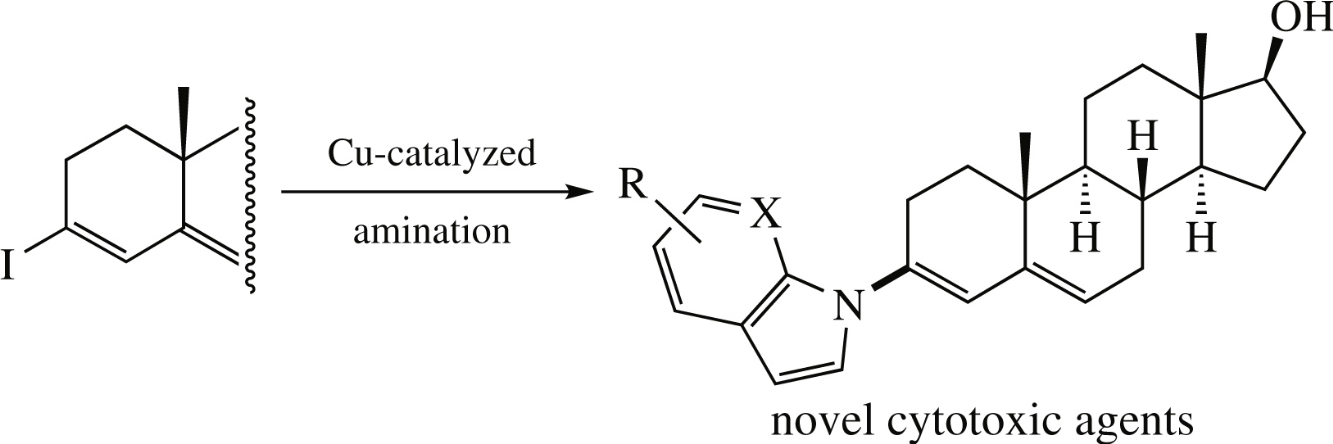
A series of novel 3-azolylandrosta-3,5-dienes was prepared via Cu-catalyzed Ullmann C–N coupling between a readily available steroidal vinyliodide and a variety of NH-heterocycles. The cytotoxic activity of the target compounds was evaluated against selected cancer cell lines (MCF-7, SKOV-3, DU-145, PC-3). Compound bearing gramine fragment showed the highest antiproliferative effect with IC50 values in the range of 2.0–10.1μm.
References
1.

Khan M.O., Lee H.J.
Chemical Reviews,
2008
2.

Ke S.
Mini-Reviews in Medicinal Chemistry,
2017
3.

Shagufta, Ahmad I., Panda G.
European Journal of Medicinal Chemistry,
2017
4.

Blanco M., La D., Coughlin Q., Newman C.A., Griffin A.M., Harrison B.L., Salituro F.G.
Bioorganic and Medicinal Chemistry Letters,
2018
5.

Heasley B.
Chemistry - A European Journal,
2012
6.

Hanson J.R.
Natural Product Reports,
2010
7.

Bansal R., Acharya P.C.
Chemical Reviews,
2014
8.

Gupta A., Sathish Kumar B., Negi A.S.
Journal of Steroid Biochemistry and Molecular Biology,
2013
9.

Salvador J.A., Carvalho J.F., Neves M.A., Silvestre S.M., Leitão A.J., Silva M.M., Sá e Melo M.L.
Natural Product Reports,
2013
10.

Tantawy M.A., Nafie M.S., Elmegeed G.A., Ali I.A.
Bioorganic Chemistry,
2017
11.

Kovács D., Wölfling J., Szabó N., Szécsi M., Kovács I., Zupkó I., Frank É.
European Journal of Medicinal Chemistry,
2013
12.

Banday A.H., Mir B.P., Lone I.H., Suri K.A., Kumar H.M.
Steroids,
2010
13.
![Synthesis of new steroidal imidazo [1,2-a] pyridines: DNA binding studies, cleavage activity and in vitro cytotoxicity](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Dar A.M., Shamsuzzaman, Gatoo M.A.
Steroids,
2015
14.

Scherbakov A.M., Komkov A.V., Komendantova A.S., Yastrebova M.A., Andreeva O.E., Shirinian V.Z., Hajra A., Zavarzin I.V., Volkova Y.A.
Frontiers in Pharmacology,
2018
15.

Volkova Y.A., Antonov Y.S., Komkov A.V., Scherbakov A.M., Shashkov A.S., Menchikov L.G., Chernoburova E.I., Zavarzin I.V.
RSC Advances,
2016
16.

Samanta S., Ghosh A.K., Ghosh S., Ilina A.A., Volkova Y.A., Zavarzin I.V., Scherbakov A.M., Salnikova D.I., Dzichenka Y.U., Sachenko A.B., Shirinian V.Z., Hajra A.
Organic and Biomolecular Chemistry,
2020
17.
![A novel [1,2,4] triazolo [1,5-a] pyrimidine-based phenyl-linked steroid dimer: Synthesis and its cytotoxic activity](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Yu B., Shi X., Zheng Y., Fang Y., Zhang E., Yu D., Liu H.
European Journal of Medicinal Chemistry,
2013
18.

O'Donnell A., Judson I., Dowsett M., Raynaud F., Dearnaley D., Mason M., Harland S., Robbins A., Halbert G., Nutley B., Jarman M.
British Journal of Cancer,
2004
19.

Logothetis C.J., Efstathiou E., Manuguid F., Kirkpatrick P.
Nature Reviews Drug Discovery,
2011
20.

Rehman Y., Rehman Y.
Drug Design, Development and Therapy,
2012
21.

Bryce A., Ryan C.J.
Clinical Pharmacology and Therapeutics,
2011
22.

Skoda-Földes R., Kollár L.
Chemical Reviews,
2003
23.

Kotora M., Hessler F., Eignerová B.
European Journal of Organic Chemistry,
2011
24.

Czajkowska-Szczykowska D., Morzycki J.W., Wojtkielewicz A.
Steroids,
2015
25.
Ibrahim-Ouali M., Dumur F.
Arkivoc,
2017
26.

Lukashev N.V., Latyshev G.V., Donez P.A., Skryabin G.A., Beletskaya I.P.
Synthesis,
2005
27.

Lukashev N.V., Latyshev G.V., Donez P.A., Skryabin G.A., Beletskaya I.P.
Synthesis,
2006
28.

Latyshev G., Kotovshchikov Y., Beletskaya I., Lukashev N.
Synthesis,
2018
29.

Latyshev G.V., Lukashev N.V., Beletskaya I.P.
Russian Journal of Organic Chemistry,
2008
30.

Averin A.D., Ranyuk E.R., Lukashev N.V., Beletskaya I.P.
Chemistry - A European Journal,
2005
31.

Ranyuk E.R., Averin A.D., Lukashev N.V., Buryak A.K., Beletskaya I.P.
Russian Journal of Organic Chemistry,
2009
32.

Averin A.D., Abel A.S., Grigorova O.K., Latyshev G.V., Kotovshchikov Y.N., Mitrofanov A.Y., Bessmertnykh-Lemeune A., Beletskaya I.P.
Pure and Applied Chemistry,
2020
33.

Kotovshchikov Y.N., Latyshev G.V., Lukashev N.V., Beletskaya I.P.
European Journal of Organic Chemistry,
2013
34.

Kotovshchikov Y.N., Latyshev G.V., Lukashev N.V., Beletskaya I.P.
Organic and Biomolecular Chemistry,
2014
35.

Kotovshchikov Y.N., Latyshev G.V., Lukashev N.V., Beletskaya I.P.
Organic and Biomolecular Chemistry,
2015
36.

Erzunov D.A., Latyshev G.V., Averin A.D., Beletskaya I.P., Lukashev N.V.
European Journal of Organic Chemistry,
2015
37.
10.1016/j.mencom.2021.04.025_sbref0040f
Latyshev
Synthesis,
2009
38.

Beletskaya I.P., Cheprakov A.V.
Coordination Chemistry Reviews,
2004
39.

Evano G., Blanchard N., Toumi M.
Chemical Reviews,
2008
40.

Monnier F., Taillefer M.
Angewandte Chemie - International Edition,
2009
41.

Beletskaya I.P., Cheprakov A.V.
Organometallics,
2012