Keywords
acid salts
crystal structure
dihydrogen trinitrate
hydrogen bond
Ionic liquid
Abstract
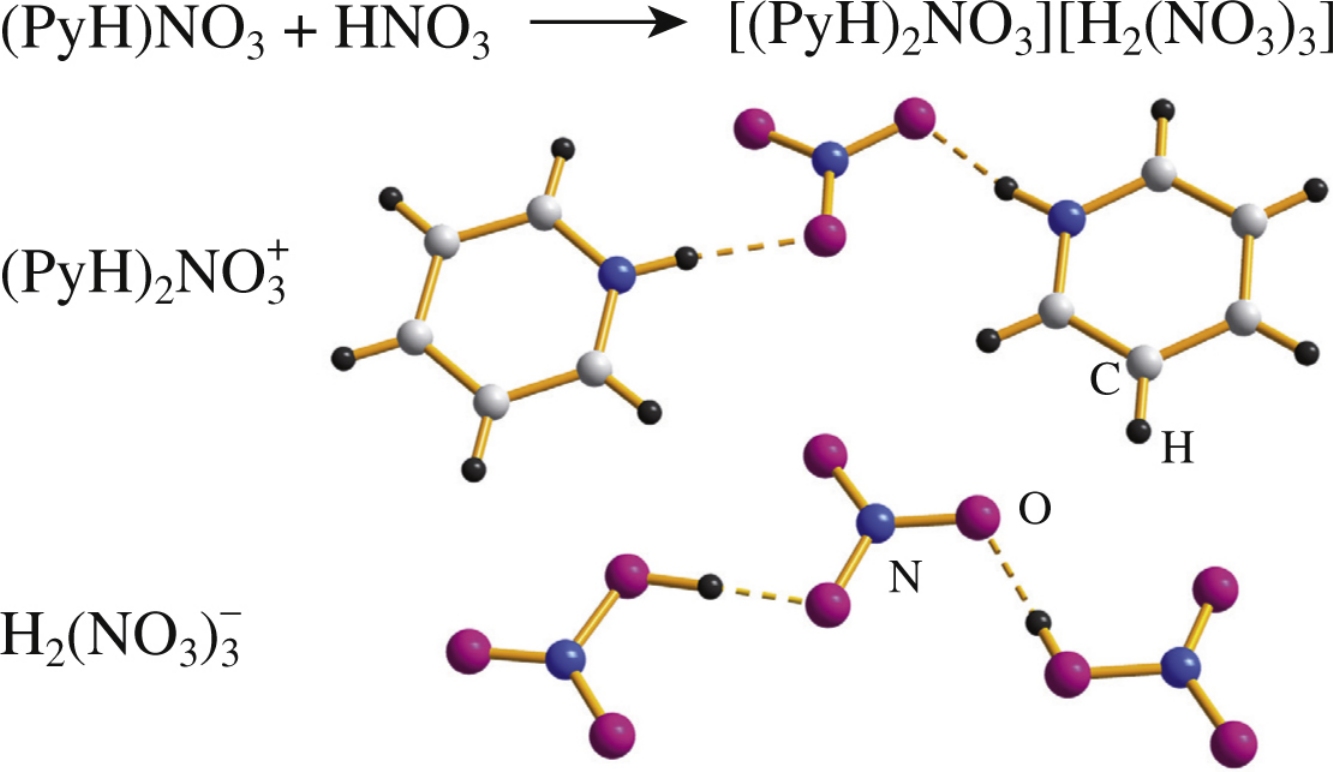
A new protic room-temperature ionic liquid (Tc=14 ± 1°C) – pyridinium hydrogen dinitrate – was obtained from a solution of pyridinium nitrate in anhydrous nitric acid in a desiccator with P2O5. According to single crystal X-ray diffraction data, the solid state structure of [(PyH)2(NO3)][H2(NO3)3] consists of hydrogen bonded cations [(PyH)2(NO3)]+ and anions [(NO3)(HNO3)2]− with a previously unknown configuration containing two HNO3 molecules linked via hydrogen bonds to the central NO3- anion by the sin–anti type.
References
1.

Glidewell C.
Journal of Molecular Structure THEOCHEM,
1981
2.
Morozov I.V., Kemnitz E., Troyanov S.I.
Mendeleev Communications,
2020
3.

Duke J.R., Llewellyn F.J.
Acta Crystallographica,
1950
4.

Diop L., Potier J.
Journal of Molecular Structure,
1977
5.

Roziere J., Roziere-Bories M.T., Williams J.M.
Inorganic Chemistry,
1976
6.

Faithful B.D., Wallwork S.C.
Chemical Communications (London),
1967
7.

Thevenet G., Rodier N.
Acta Crystallographica Section B,
1978
8.

Al-Zamil N.S., Evans E.H., Gillard R.D., James D.W., Jenkins T.E., Lancashire R.J., Williams P.A.
Polyhedron,
1982
9.
10.1016/j.mencom.2021.04.024_bib0045
Huta
Acta Crystallogr.,
2002
10.

Rozière J., Lehmann M.S., Potier J.
Acta Crystallographica Section B,
1979
11.

Junk P.C., Kepert C.J., Semenova L.I., Skelton B.W., White A.H.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2006
12.

Einstein F.W., Tuck D.G.
Acta Crystallographica Section B,
1970
13.
10.1016/j.mencom.2021.04.024_bib0065
Batsanov
Acta Crystallogr.,
2004
14.
10.1016/j.mencom.2021.04.024_bib0070
Handbuch der praparativen anorganischen Chemie,
1978
15.

Greaves T.L., Drummond C.J.
Chemical Reviews,
2007
16.

Greaves T.L., Drummond C.J.
Chemical Reviews,
2015
17.
![Synthesis and crystal structures of zirconium(IV) nitrate complexes (NO2)[Zr(NO3)3(H2O)3]2(NO3) 3, Cs[Zr(NO3)5], and (NH4)[Zr(NO3)5](HNO3)](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Morozov I.V., Fedorova A.A., Palamarchuk D.V., Troyanov S.I.
Russian Chemical Bulletin,
2005
18.
10.1016/j.mencom.2021.04.024_bib0090
Znamenkov
Russ. J. Inorg. Chem.,
2004
19.

20.

Cox A.P., Riveros J.M.
Journal of Chemical Physics,
1965
21.

Allan D.R., Marshall W.G., Francis D.J., Oswald I.D., Pulham C.R., Spanswick C.
Dalton Transactions,
2010
22.

Morozov I.V., Serezhkin V.N., Troyanov S.I.
Russian Chemical Bulletin,
2009