Keywords
3,7-diazabicyclo[3.3.1]nonane
conformation analysis
hydrogen peroxide
organoiodine compounds
peroxosolvates
X-ray diffraction study
α-halo carbonyl compounds
Abstract
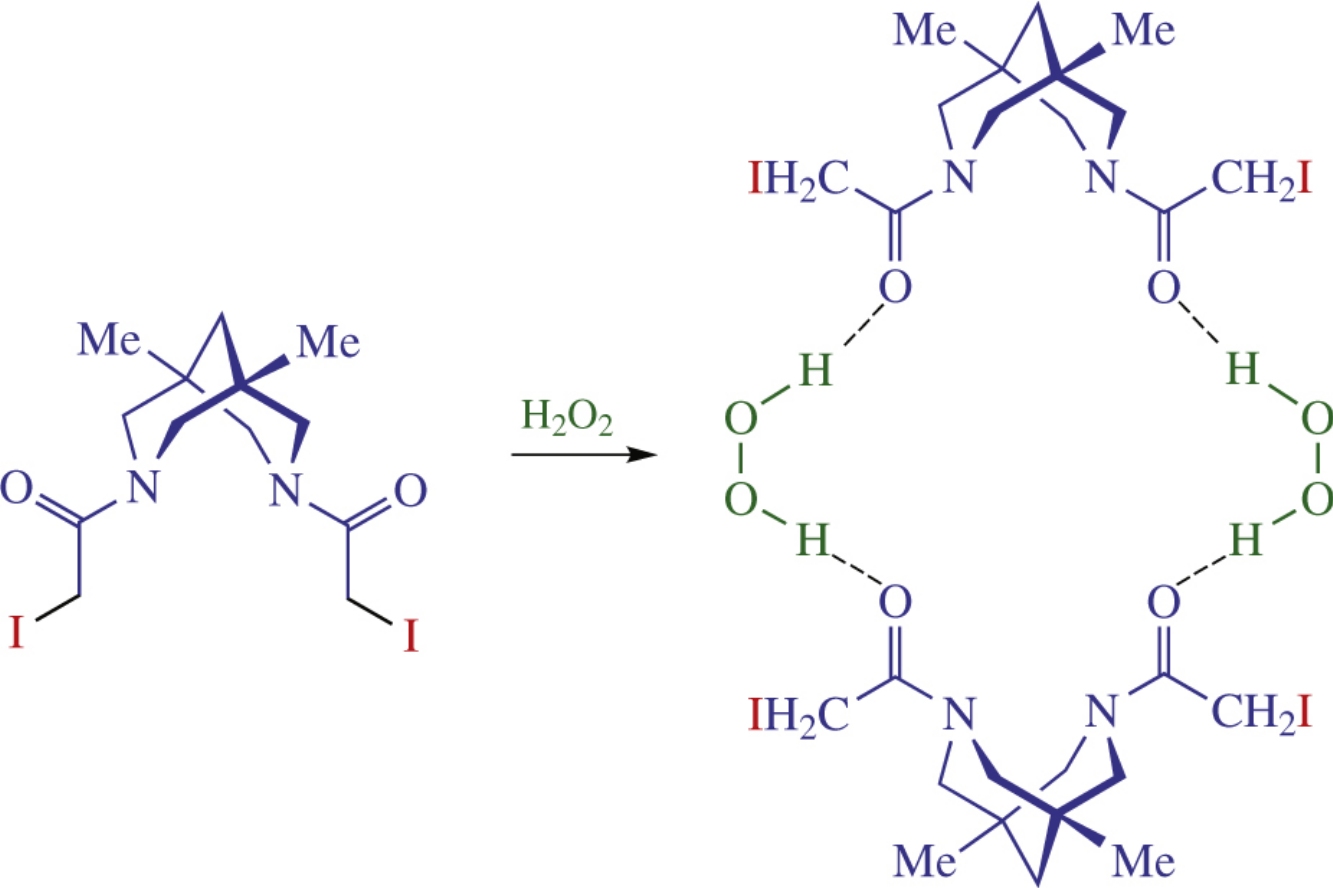
Six new bispidine-based bis-amides containing residues of haloacetic acids were synthesized and characterized by X-ray crystallography. The dissolution of iodine-containing compound in concentrated hydrogen peroxide afforded bispidine peroxosolvate, in which the peroxide molecules participated in two donor hydrogen bonds with carbonyl oxygen atoms of the distinct adjacent organic molecules thus forming cyclic motifs in crystals. This structure is a first example of peroxosolvate of iodine-containing organic molecule.
References
1.

Bolla G., Nangia A.
Chemical Communications,
2016
2.
10.1016/j.mencom.2021.04.023_bib0010
Hilfiker
Polymorphism: in the Pharmaceutical Industry,
2006
3.

Kersten K.M., Breen M.E., Mapp A.K., Matzger A.J.
Chemical Communications,
2018
4.

Tomassoli I., Gündisch D.
Current Topics in Medicinal Chemistry,
2016
5.
Vatsadze S.Z., Medved’ko A.V., Bodunov A.A., Lyssenko K.A.
Mendeleev Communications,
2020
6.

Vatsadze S.Z., Krut’ko D.P., Blake A.J., Mountford P.
Macroheterocycles,
2017
7.
Lavrov M.I., Veremeeva P.N., Karlov D.S., Zamoyski V.L., Grigoriev V.V., Palyulin V.A.
Mendeleev Communications,
2019
8.

Montanari V., DesMarteau D.D., Pennington W.T.
Journal of Molecular Structure,
2000
9.
![[Hydroxy(sulfonyloxy)iodo]perfluoroalkanes - new hypervalent iodine species and promising reagents for organic synthesis](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Zhdankin V.V., Kuehl C.
Tetrahedron Letters,
1994
10.

Mukherjee T., Biswas S., Ehnbom A., Ghosh S.K., El-Zoghbi I., Bhuvanesh N., Bazzi H.S., Gladysz J.A.
Beilstein Journal of Organic Chemistry,
2017
11.

Kawashima T., Hoshiba K., Kano N.
Journal of the American Chemical Society,
2001
12.
![1-[Hydroxy(sulfonyloxy)iodo]-2,2,2-trifluoroethanes, CF3CH2I(OH)OSO2R: Stable, fluoroalkyl analogs of Koser's reagent](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Zhdankin V.V., Kuehl C.J., Simonsen A.J.
Tetrahedron Letters,
1995
13.
![1-[Hydroxy(sulfonyloxy)iodo]-1H,1H-perfluoroalkanes: Stable, Fluoroalkyl Analogs of Koser's Reagent](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Zhdankin V.V., Kuehl C.J., Simonsen A.J.
Journal of Organic Chemistry,
1996
14.
![A General and Convenient Preparation of [Bis(trifluoroacetoxy)iodo]perfluoroalkanes and [Bis(trifluoroacetoxy)iodo]arenes by Oxidation of Organic Iodides Using Oxone and Trifluoroacetic Acid](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Zagulyaeva A.A., Yusubov M.S., Zhdankin V.V.
Journal of Organic Chemistry,
2010
15.

Asensio G., Andreu C., Boix-Bernardini C., Mello R., González-Nuñez M.E.
Organic Letters,
1999
16.

Nicoletti D., Ghini A.A., Burton G.
Journal of Organic Chemistry,
1996
17.

Medved’ko A.V., Egorova B.V., Komarova A.A., Rakhimov R.D., Krut’ko D.P., Kalmykov S.N., Vatsadze S.Z.
ACS Omega,
2016
18.
![Synthesis of 3,7-diacyl-1,5-dimethyl-3,7-diazabicyclo[3.3.1]nonane derivatives as promising lipid bilayer modifiers](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Veremeeva P.N., Grishina I.V., Zaborova O.V., Averin A.D., Palyulin V.A.
Tetrahedron,
2019
19.

Wang Z., Islam M.J., Vukotic V.N., Revington M.J.
Journal of Organic Chemistry,
2016
20.
10.1016/j.mencom.2021.04.023_bib0100
Groom
Acta Crystallogr.,
2016
21.

Medved’ko A., Dalinger A., Nuriev V., Semashko V., Filatov A., Ezhov A., Churakov A., Howard J., Shiryaev A., Baranchikov A., Ivanov V., Vatsadze S.
Nanomaterials,
2019
22.

Grishanov D.A., Navasardyan M.A., Medvedev A.G., Lev O., Prikhodchenko P.V., Churakov A.V.
Angewandte Chemie - International Edition,
2017
23.

Churakov A.V., Grishanov D.A., Medvedev A.G., Mikhaylov A.A., Tripol'skaya T.A., Vener M.V., Navasardyan M.A., Lev O., Prikhodchenko P.V.
CrystEngComm,
2019
24.

Chernyshov I.Y., Vener M.V., Prikhodchenko P.V., Medvedev A.G., Lev O., Churakov A.V.
Crystal Growth and Design,
2016
25.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007