Keywords
Cyclization
electrocatalysis
electrosynthesis
mediated electrolysis
spirobarbituric dihydrofurans
Abstract
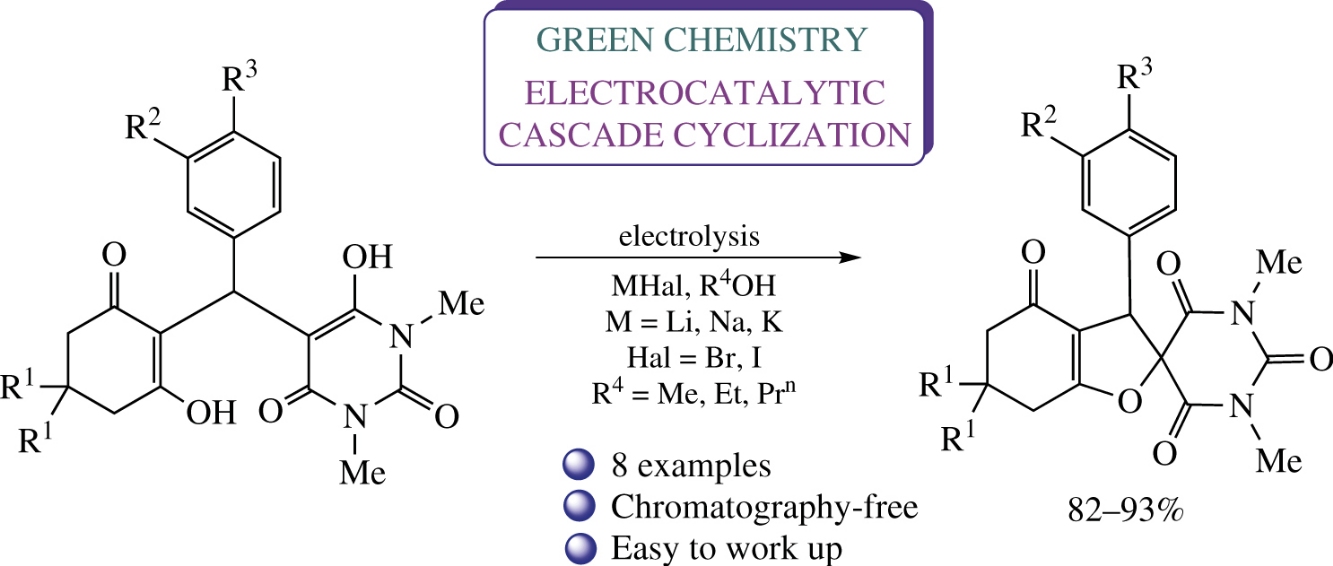
Electrocatalytic cyclization of 6-hydroxy-5-[(2-hydroxy-6-oxocyclohex-1-en-1-yl)(aryl)methyl]-1,3-dimethylpyrimidine-2,4-(1H,3H)-diones in alcohols in an undivided cell in the presence of sodium halides results in selective formation of substituted spirobarbituric dihydrofurans in 82–93% yields. Crystal structure of 3-(3-bromophenyl)-1′,3′,6,6-tetramethyl-3,5,6,7-tetrahydro-2′,4-dihydrospiro[benzofuran-2,5′-pyrimidine]-2′,4,4′,6′(1′H,3′H)-tetraone has been confirmed by X-ray diffraction data.
References
1.
10.1016/j.mencom.2021.04.021_sbref0005a
Organic Electrochemistry: Revised and Expanded,
2015
2.

Yan M., Kawamata Y., Baran P.S.
Angewandte Chemie - International Edition,
2017
3.
Nikishin G.I., Elinson M.N., Makhova I.V.
1988
4.

Francke R., Little R.D.
Chemical Society Reviews,
2014
5.

Ogibin Y.N., Elinson M.N., Nikishin G.I.
Russian Chemical Reviews,
2009
6.
10.1016/j.mencom.2021.04.021_sbref0020a
Arora
Int. J. Pharm. Sci. Res.,
2012
7.

Kachaeva M.V., Obernikhina N.V., Veligina E.S., Zhuravlova M.Y., Prostota Y.O., Kachkovsky O.D., Brovarets V.S.
Chemistry of Heterocyclic Compounds,
2019
8.

Gridina T.L., Fedchuk A.S., Basok S.S., Artemenko A.G., Ognichenko L.N., Shitikova L.I., Lutsyuk A.F., Gruzevskii A.A., Kuz’min V.E.
Chemistry of Heterocyclic Compounds,
2019
9.

Taylor A.P., Robinson R.P., Fobian Y.M., Blakemore D.C., Jones L.H., Fadeyi O.
Organic and Biomolecular Chemistry,
2016
10.

11.

Uhlmann C., Fröscher W.
CNS Neuroscience and Therapeutics,
2009
12.

Naguib F.N., Levesque D.L., Eng-Chi Wang, Panzica R.P., El Kouni M.H.
Biochemical Pharmacology,
1993
13.

Pyrimidine-2,4,6-Triones: A New Effective and Selective Class of Matrix Metalloproteinase Inhibitors
Grams F., Brandstetter H., DAlò S., Geppert D., Krell H., Leinert H., Livi V., Menta E., Oliva A., Zimmermann G.
Biological Chemistry,
2001
14.

Maquoi E., Sounni N.E., Devy L., Olivier F., Frankenne F., Krell H., Grams F., Foidart J., Noël A.
Clinical Cancer Research,
2004
15.

Murinov Y.I., Grabovskii S.A., Kabal’nova N.N.
Russian Chemical Bulletin,
2019
16.

Rios R.
Chemical Society Reviews,
2012
17.

Zheng Y., Tice C.M., Singh S.B.
Bioorganic and Medicinal Chemistry Letters,
2014
18.

King S.B., Stratford E.S., Craig C.R., Fifer E.K.
Pharmaceutical Research,
1995
19.

Prankerd R.J., McKeown R.H.
International Journal of Pharmaceutics,
1992
20.

Lomlim L., Einsiedel J., Heinemann F.W., Meyer K., Gmeiner P.
Journal of Organic Chemistry,
2008
21.

Galati E.M., Monforte M.T., Miceli N., Raneri E.
Il Farmaco,
2001
22.

Kim S., Pudzianowski A.T., Leavitt K.J., Barbosa J., McDonnell P.A., Metzler W.J., Rankin B.M., Liu R., Vaccaro W., Pitts W.
Bioorganic and Medicinal Chemistry Letters,
2005
23.

Fraser W., Suckling C.J., Wood H.C.
Journal of the Chemical Society Perkin Transactions 1,
1990
24.
J. Duan, B. Jiang, L. Chen, Z. Lu, J. Barbosa and W. J. Pitts, US Patent 7294624 B2, 2007.
25.

Elinson M.N., Dorofeeva E.O., Vereshchagin A.N., Nasybullin R.F., Egorov M.P.
Catalysis Science and Technology,
2015
26.

Elinson M.N., Dorofeeva E.O., Vereshchagin A.N., Nikishin G.I.
Russian Chemical Reviews,
2015
27.

Vereshchagin A.N., Elinson M.N., Dorofeeva E.O., Stepanov N.O., Zaimovskaya T.A., Nikishin G.I.
Tetrahedron,
2013
28.
![The electrocatalytic cascade assembling of isatins, malononitrile and N-alkyl barbiturates: An efficient multicomponent approach to the spiro[indole-3,5′-pyrano[2,3-d]pyrimidine] framework](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Elinson M.N., Ilovaisky A.I., Merkulova V.M., Demchuk D.V., Belyakov P.A., Ogibin Y.N., Nikishin G.I.
Electrochimica Acta,
2008
29.
Elinson M.N., Feducovich S.K., Bushuev S.G., Zakharenkov A.A., Pashchenko D.V., Nikishin G.I.
Mendeleev Communications,
1998
30.
Elinson M.N., Vereshchagin A.N., Anisina Y.E., Leonova N.A., Egorov M.P.
Mendeleev Communications,
2020
31.
Vereshchagin A.N., Dorofeeva E.O., Elinson M.N., Korolev V.A., Egorov M.P.
Mendeleev Communications,
2019
32.

Vereshchagin A.N., Elinson M.N., Egorov M.P.
RSC Advances,
2015
33.
![Stereoselective electrocatalytic transformation of arylidene- or alkylidenemalononitriles and malonate into alkyl (1R,5R,6R)* 6-substituted 5-cyano-4,4-dialkoxy-2-oxo-3-azabicyclo[3.1.0]hexane-1-carboxylates](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Elinson M.N., Feducovich S.K., Starikova Z.A., Vereshchagin A.N., Gorbunov S.V., Nikishin G.I.
Tetrahedron Letters,
2005