Keywords
arylthiolation
chiral template
cysteine derivatives
Michael addition
nickel complexes
Schiff bases
stereoselective synthesis
sulfides
Abstract
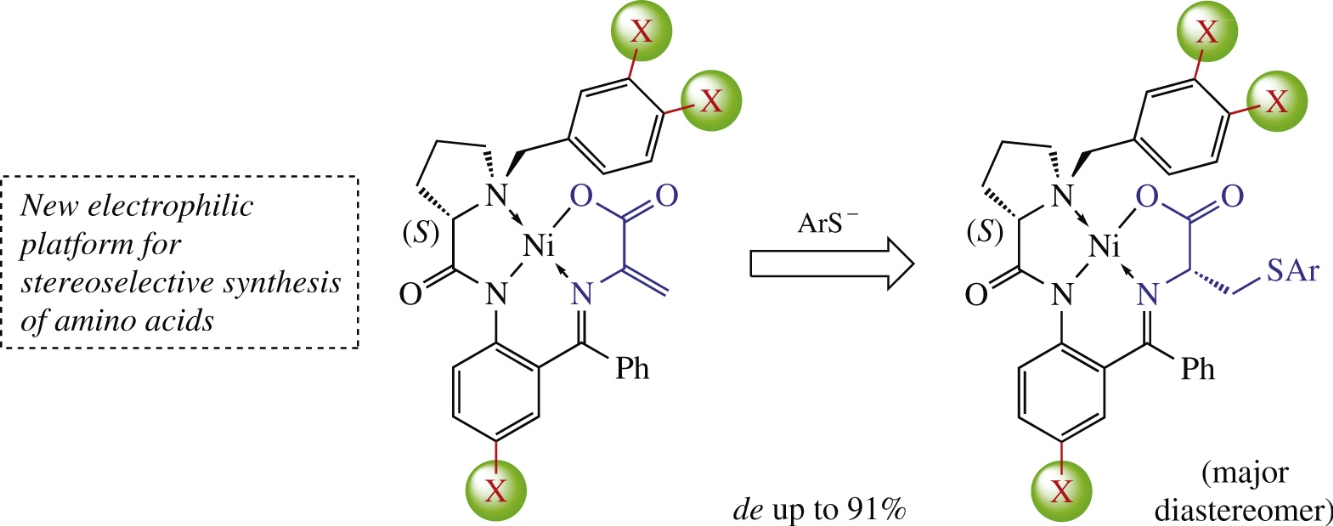
Arylthiolation of new dehydroalanine NiII Schiff-base complex containing (S)-2-[N-(N′-2,3-dichlorobenzylprolyl)amino]-5-chlorobenzophenone auxiliary affords (S,R)-cysteine derivatives in high chemical yield (65–88%) and with excellent diastereoselectivity (de up to 91%), which significantly exceeds that for the commonly used analogue depriving of three chlorine atoms.
References
1.

Wan Q., Danishefsky S.
Angewandte Chemie,
2007
2.

Vamisetti G.B., Satish G., Sulkshane P., Mann G., Glickman M.H., Brik A.
Journal of the American Chemical Society,
2020
3.
![Calix[4]arene decorated with four Tn antigen glycomimetic units and P3CS immunoadjuvant: synthesis, characterization, and anticancer immunological evaluation.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Geraci C., Consoli G.M., Galante E., Bousquet E., Pappalardo M., Spadaro A.
Bioconjugate Chemistry,
2008
4.

Wang Q., Guo Z.
ACS Medicinal Chemistry Letters,
2011
5.

Liu C., Ye X.
Glycoconjugate Journal,
2012
6.

Kaldor S.W., Kalish V.J., Davies J.F., Shetty B.V., Fritz J.E., Appelt K., Burgess J.A., Campanale K.M., Chirgadze N.Y., Clawson D.K., Dressman B.A., Hatch S.D., Khalil D.A., Kosa M.B., Lubbehusen P.P., et. al.
Journal of Medicinal Chemistry,
1997
7.
Atmaca G.
Yonsei Medical Journal,
2004
8.

Kang-Sickel J.C., Fox D.D., Nam T., Jayaraj K., Ball L.M., French J.E., Klapper D.G., Gold A., Nylander-French L.A.
Chemical Research in Toxicology,
2008
9.

Emre Y., Imhof B.A.
Seminars in Immunopathology,
2014
10.

Mologni L.
Current Medicinal Chemistry,
2011
11.
10.1016/j.mencom.2021.04.018_bib0055
Inoue
Mediators Inflammation,
2009
12.

Belokon Y.N., Bulychev A.G., Vitt S.V., Struchkov Y.T., Batsanov A.S., Timofeeva T.V., Tsyryapkin V.A., Ryzhov M.G., Lysova L.A.
Journal of the American Chemical Society,
1985
13.

Sorochinsky A.E., Aceña J.L., Moriwaki H., Sato T., Soloshonok V.A.
Amino Acids,
2013
14.

Sorochinsky A.E., Aceña J.L., Moriwaki H., Sato T., Soloshonok V.
Amino Acids,
2013
15.

Zhou S., Wang J., Chen X., Aceña J.L., Soloshonok V.A., Liu H.
Angewandte Chemie - International Edition,
2014
16.

Wang Y., Song X., Wang J., Moriwaki H., Soloshonok V.A., Liu H.
Amino Acids,
2017
17.

Zou Y., Han J., Saghyan A.S., Mkrtchyan A.F., Konno H., Moriwaki H., Izawa K., Soloshonok V.A.
Molecules,
2020
18.

Belokon' Y.N., Sagyan A.S., Djamgaryan S.M., Bakhmutov V.I., Belikov V.M.
Tetrahedron,
1988
19.

Saghiyan A.S., Geolchanyan A.V., Djamgaryan S.M., Vardapetyan S.M., Tararov V.I., Kuz'mina N.A., Ikonnikov N.S., Belokon Y.N., North M.
Russian Chemical Bulletin,
2000
20.

Saghiyan A.S., Geolchanyan A.V., Manasyan L.L., Mkrtchyan G.M., Martirosyan N.R., Dadayan S.A., Kochickyan T.V., Harutyunyan V.S., Avetisyan A.A., Tararov V.I., Maleev V.I., Belokon" Y.N.
Russian Chemical Bulletin,
2004
21.

Yamamoto J., Kawashima A., Kawamura A., Abe H., Moriwaki H., Shibata N., Soloshonok V.A.
European Journal of Organic Chemistry,
2017
22.

Aydillo C., Compañón I., Avenoza A., Busto J.H., Corzana F., Peregrina J.M., Zurbano M.M.
Journal of the American Chemical Society,
2014
23.

Gutiérrez-Jiménez M.I., Aydillo C., Navo C.D., Avenoza A., Corzana F., Jiménez-Osés G., Zurbano M.M., Busto J.H., Peregrina J.M.
Organic Letters,
2016
24.

West H.D., Mathura G.R.
Journal of Biological Chemistry,
1954
25.

Sandin R.B., Christiansen R.G., Brown R.K., Kirkwood S.
Journal of the American Chemical Society,
1947
26.

Belokon Y.N., Bakhmutov V.I., Chernoglazova N.I., Kochetkov K.A., Vitt S.V., Garbalinskaya N.S., Belikov V.M.
Journal of the Chemical Society Perkin Transactions 1,
1988
27.
10.1016/j.mencom.2021.04.018_bib0135
Organic Electrochemistry,
2015
28.

Magdesieva T.V., Levitskiy O.A., Grishin Y.K., Ambartsumyan A.A., Kiskin M.A., Churakov A.V., Babievsky K.K., Kochetkov K.A.
Organometallics,
2014
29.

Levitskiy O.A., Grishin Y.K., Paseshnichenko K.A., Kochetkov K.A., Magdesieva T.V.
Tetrahedron Letters,
2018
30.

Magdesieva T.V., Levitskiy O.A., Grishin Y.K., Ambartsumyan A.A., Paseshnichenko K.A., Kolotyrkina N.G., Kochetkov K.A.
Organometallics,
2014
31.

Levitskiy O.A., Grishin Y.K., Semivrazhskaya O.O., Ambartsumyan A.A., Kochetkov K.A., Magdesieva T.V.
Angewandte Chemie - International Edition,
2017
32.

Levitskiy O.A., Aglamazova O.I., Soloshonok V.A., Moriwaki H., Magdesieva T.V.
Chemistry - A European Journal,
2020
33.

Nian Y., Wang J., Zhou S., Wang S., Moriwaki H., Kawashima A., Soloshonok V.A., Liu H.
Angewandte Chemie - International Edition,
2015
34.

Han J., Takeda R., Liu X., Konno H., Abe H., Hiramatsu T., Moriwaki H., Soloshonok V.A.
Molecules,
2019
35.

Levitskiy O.A., Grishin Y.K., Magdesieva T.V.
European Journal of Organic Chemistry,
2019
36.

du Vigneaud V., Wood J.L., Binkley F.
Journal of Biological Chemistry,
1941
37.

Navo C.D., Asín A., Gómez-Orte E., Gutiérrez-Jiménez M.I., Compañón I., Ezcurra B., Avenoza A., Busto J.H., Corzana F., Zurbano M.M., Jiménez-Osés G., Cabello J., Peregrina J.M.
Chemistry - A European Journal,
2018