Keywords
alloy
Cd
electron–hole pairs
hot electrons
hydrogen evolution reaction
lifetime
Mn
nanocrystals
ZnS
Abstract
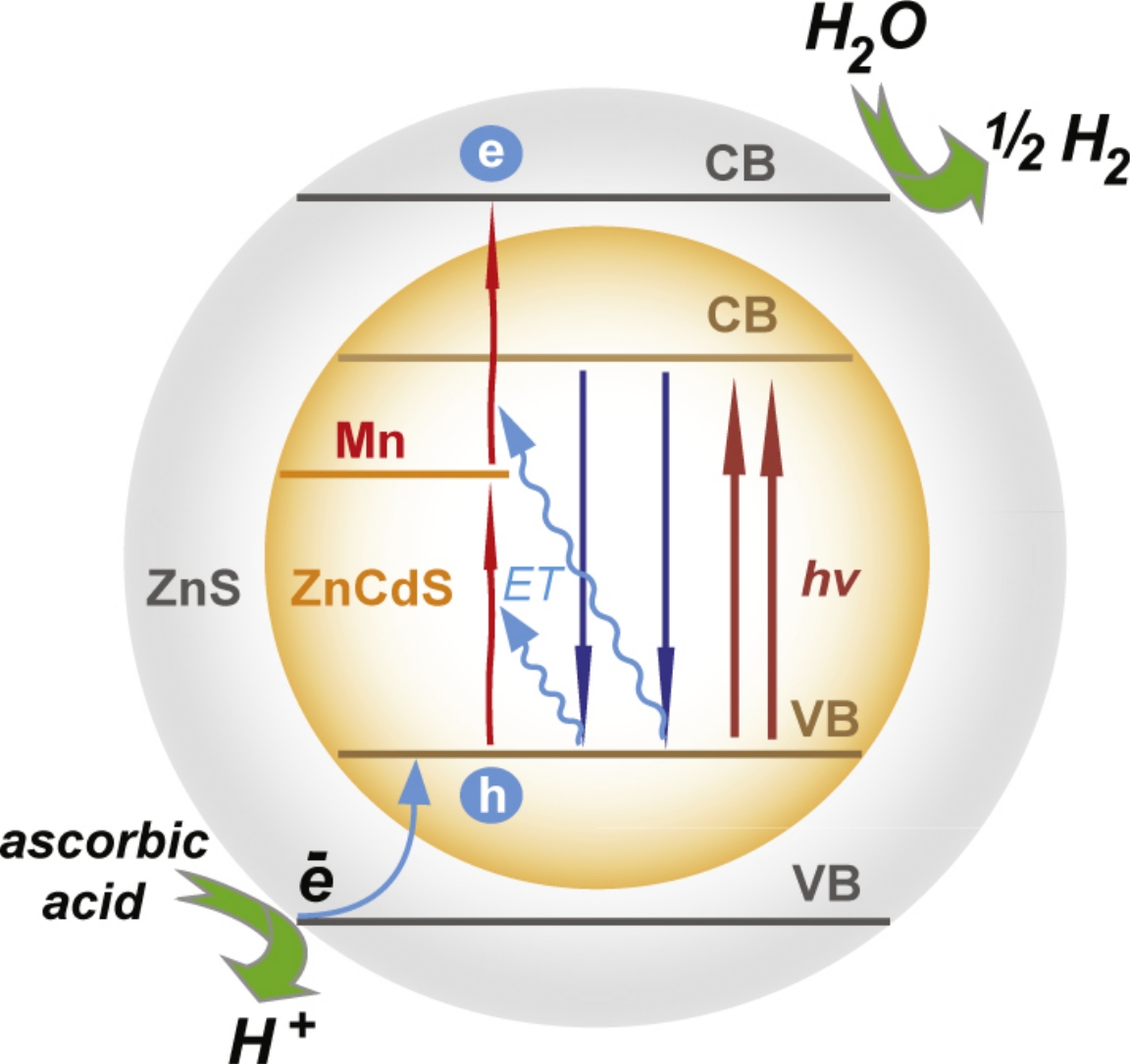
The effect of doping of the ZnxCd1−xS (x=0.37–0.50) alloy nanocrystals with Mn2+ ions on the rate and apparent quantum yield of the photocatalytic H2 evolution catalyzed by this alloy has been investigated. It has been found that the ZnS shell significantly increases the lifetime of the Mn2+ excited state, which leads to the generation of ‘hot’ electrons and the two-photon photocatalytic reduction of hydrogen ions. An analysis of the characteristics of nanocrystals without a ZnS shell with similar excitation energies of the edge exciton revealed that the efficiency of the alloy doped with Mn2+ ions is 1.2–1.3 times higher due to an increase in the lifetime of photoinduced electron–hole pairs.
References
1.

2.

Beaulac R., Archer P.I., van Rijssel J., Meijerink A., Gamelin D.R.
Nano Letters,
2008
3.

Singhal M., Sharma J.K., Jeon H.C., Kang T.W., Kumar S.
Advances in Applied Ceramics,
2019
4.

Parobek D., Qiao T., Son D.H.
Journal of Chemical Physics,
2019
5.

Chen H., Chen T., Berdugo E., Park Y., Lovering K., Son D.H.
Journal of Physical Chemistry C,
2011
6.

Dong Y., Choi J., Jeong H., Son D.H.
Journal of the American Chemical Society,
2015
7.
10.1016/j.mencom.2021.04.012_bib0035
Hydrogen as a Future Energy Carrier,
2008
8.

Wang Q., Domen K.
Chemical Reviews,
2019
9.

Wang H., Li Y., Shu D., Chen X., Liu X., Wang X., Zhang J., Wang H.
International Journal of Energy Research,
2016
10.

Shen C., Liu Y., Zhou X., Guo H., Zhao Z., Liang K., Xu A.
Catalysis Science and Technology,
2017
11.

Li Q., Meng H., Zhou P., Zheng Y., Wang J., Yu J., Gong J.
ACS Catalysis,
2013
12.

Nag A., Chakraborty S., Sarma D.D.
Journal of the American Chemical Society,
2008
13.

Kabachii Y.A., Golub’ A.S., Goloveshkin A.S., Abramchuk S.S., Shapovalov A.V., Buzin M.I., Valetskii P.M., Kochev S.Y.
Russian Chemical Bulletin,
2014
14.

Nien Y., Chen P., Chen I.
Journal of Alloys and Compounds,
2008
15.

Gulin A., Shakhov A., Vasin A., Astafiev A., Antonova O., Kochev S., Kabachii Y., Golub A., Nadtochenko V.
Applied Surface Science,
2019
16.

Jones M., Lo S.S., Scholes G.D.
Proceedings of the National Academy of Sciences of the United States of America,
2009
17.

Stroyuk A.L., Dzhagan V.M., Kuchmii S.Y., Valakh M.Y., Zahn D.R., von Borczyskowski C.
Theoretical and Experimental Chemistry,
2007
18.

Jing L., Kershaw S.V., Li Y., Huang X., Li Y., Rogach A.L., Gao M.
Chemical Reviews,
2016
19.

Sadovnikov S.I.
Russian Chemical Reviews,
2019
20.

Leatherdale C.A., Woo W.-., Mikulec F.V., Bawendi M.G.
Journal of Physical Chemistry B,
2002
21.

Ihara T., Kanemitsu Y.
Physical Review B,
2015
22.

Nanda J., Ivanov S.A., Htoon H., Bezel I., Piryatinski A., Tretiak S., Klimov V.I.
Journal of Applied Physics,
2006
23.

Yu P., Beard M.C., Ellingson R.J., Ferrere S., Curtis C., Drexler J., Luiszer F., Nozik A.J.
Journal of Physical Chemistry B,
2005
24.

Wheeler D.A., Zhang J.Z.
Advanced Materials,
2013