Keywords
DU8+
isoserrins
natural products
NMR calculations
structure revision
Abstract
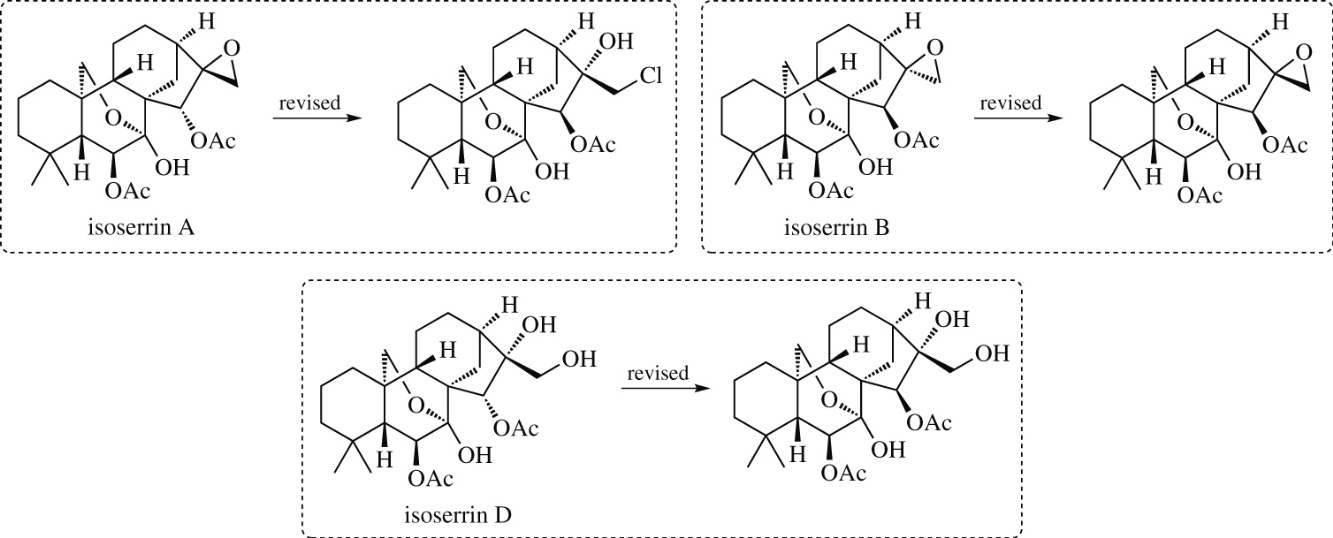
DU8+ calculations of 13C NMR chemical shifts suggested that the structures of isoserrins A, B, and D – which were recently isolated from medicinal plant Isodon serra – are misassigned. Computationally driven structure revisions are presented in this communication.
References
1.

Lodewyk M.W., Siebert M.R., Tantillo D.J.
Chemical Reviews,
2011
2.

Krivdin L.B.
Magnetic Resonance in Chemistry,
2019
3.

Krivdin L.B.
Magnetic Resonance in Chemistry,
2019
4.

Krivdin L.B.
Magnetic Resonance in Chemistry,
2019
5.

Rusakov Y.Y., Krivdin L.B.
Russian Chemical Reviews,
2013
6.
Chemical Shift Repository, http://cheshirenmr.info.
7.

Kutateladze A.G., Reddy D.S.
Journal of Organic Chemistry,
2017
8.

Addressing the Challenges of Structure Elucidation in Natural Products Possessing the Oxirane Moiety
Kutateladze A.G., Kuznetsov D.M., Beloglazkina A.A., Holt T.
Journal of Organic Chemistry,
2018
9.

Kutateladze A.G., Holt T., Reddy D.S.
Journal of Organic Chemistry,
2019
10.

Kutateladze A.G., Holt T.
Journal of Organic Chemistry,
2019
11.

Xing H., An L., Song Z., Li S., Wang H., Wang C., Zhang J., Tuerhong M., Abudukeremu M., Li D., Lee D., Xu J., Lall N., Guo Y.
Journal of Natural Products,
2020
12.

McAlpine J.B., Chen S., Kutateladze A., MacMillan J.B., Appendino G., Barison A., Beniddir M.A., Biavatti M.W., Bluml S., Boufridi A., Butler M.S., Capon R.J., Choi Y.H., Coppage D., Crews P., et. al.
Natural Product Reports,
2019