Keywords
1,5-diketones
bicyclo[3.3.1]nonanes
Cyclopropanes
cyclopropyl ketones
methyl(trifluoromethyl)dioxirane
oxidation
ozone
ruthenium(VIII) oxide
spiro compounds
Abstract
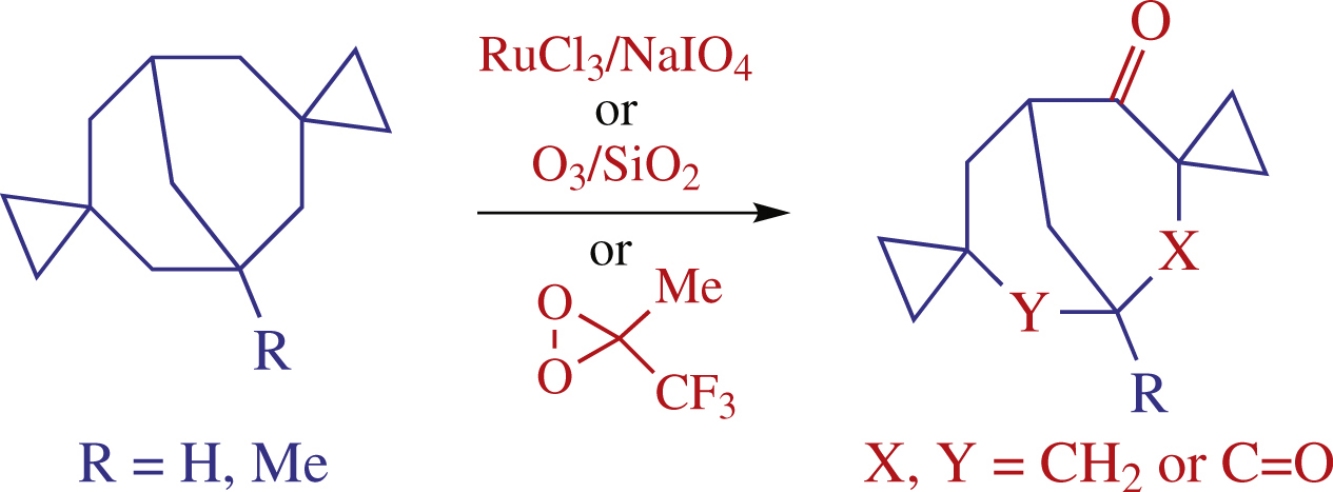
Two dispiro[cyclopropane-1,3’-bicyclo[3.3.1]nonane-7′,1″-cyclopropanes] were treated with various oxidative systems. The conditions providing mono- or dioxidation of the CH2 groups neighbouring to spiro-annulated cyclopropane moieties to afford the corresponding mono- or diketones were found.
References
1.

Kozikowski A.P., Tückmantel W.
Accounts of Chemical Research,
1999
2.

Wickerols A and B: novel anti-influenza virus diterpenes produced by Trichoderma atroviride FKI-3849
Yamamoto T., Izumi N., Ui H., Sueki A., Masuma R., Nonaka K., Hirose T., Sunazuka T., Nagai T., Yamada H., Ōmura S., Shiomi K.
Tetrahedron,
2012
3.

Ciochina R., Grossman R.B.
Chemical Reviews,
2006
4.
![Synthetic efforts toward [3.3.1] bridged bicyclic phloroglucinol natural products](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Njardarson J.T.
Tetrahedron,
2011
5.

Richard J., Pouwer R.H., Chen D.Y.
Angewandte Chemie - International Edition,
2012
6.
![Domino Michael-aldol annulations for the stereocontrolled synthesis of bicyclo[3.3.1]nonane and bicyclo[3.2.1]octane derivatives](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Promontorio R., Richard J., Marson C.
RSC Advances,
2016
7.

Wang L., Sun L., Wang X., Wu R., Zhou H., Zheng C., Xu H.
Organic Letters,
2019
8.

Smaligo A.J., Swain M., Quintana J.C., Tan M.F., Kim D.A., Kwon O.
Science,
2019
9.

Cianfanelli M., Olivo G., Milan M., Klein Gebbink R.J., Ribas X., Bietti M., Costas M.
Journal of the American Chemical Society,
2019
10.
![Chiral bipyridine-annulated bicyclo[3.3.1]nonane N-oxide organocatalysts for stereoselective allylation and hydrosilylation reactions](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Žeimytė S., Stončius S.
Tetrahedron,
2021
11.

Sedenkova K.N., Andriasov K.S., Kuznetsova T.S., Averina E.B.
Current Organic Synthesis,
2018
12.

Trost B.M.
Accounts of Chemical Research,
2002
13.

Wender P.A., Verma V.A., Paxton T.J., Pillow T.H.
Accounts of Chemical Research,
2007
14.
10.1016/j.mencom.2021.04.005_bib0070
Kozhushkov
Sci. Synth.,
2009
15.
![Beyond the Dimer and Trimer: Tetraspiro[2.1.2 5 .1.2 9 .1.2 13 .1 3 ] hexadecane‐1,3,5,7‐tetraone—the Cyclic Tetramer of Carbonylcyclopropane](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Sedenkova K.N., Averina E.B., Grishin Y.K., Andriasov K.S., Stepanova S.A., Roznyatovsky V.A., Kutateladze A.G., Rybakov V.B., Albov D.V., Kuznetsova T.S., Zefirov N.S.
Chemistry - A European Journal,
2016
16.

Sedenkova K.N., Andriasov K.S., Stepanova S.A., Gloriozov I.P., Grishin Y.K., Kuznetsova T.S., Averina E.B.
European Journal of Organic Chemistry,
2018
17.

Budynina E., Ivanov K., Sorokin I., Melnikov M.
Synthesis,
2017
18.

Afanasyev O.I., Tsygankov A.A., Usanov D.L., Chusov D.
Organic Letters,
2016
19.
![Catalytic [3 + 2] Cycloaddition through Ring Cleavage of Simple Cyclopropanes with Isocyanates](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Tsunoi S., Maruoka Y., Suzuki I., Shibata I.
Organic Letters,
2015
20.
![Brønsted acid mediated intramolecular cyclopropane ring expansion/[4 + 2]-cycloaddition](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Li J., Zhu S., Xu Q., Liu L., Yan S.
Organic and Biomolecular Chemistry,
2019
21.
10.1016/j.mencom.2021.04.005_bib0105
Krasutskii
J. Org. Chem. USSR (Engl. Transl.),
1984
22.

Averina E.B., Sedenkova K.N., Bakhtin S.G., Grishin Y.K., Kutateladze A.G., Roznyatovsky V.A., Rybakov V.B., Butov G.M., Kuznetsova T.S., Zefirov N.S.
Journal of Organic Chemistry,
2014
23.

de Meijere A., Kozhushkov S.I., Schill H.
Chemical Reviews,
2006
24.

D'Accolti L., Dinoi A., Fusco C., Russo A., Curci R.
Journal of Organic Chemistry,
2003
25.

Hasegawa T., Niwa H., Yamada K.
Chemistry Letters,
1985