Keywords
cancer cell morphology
cycloalka[d]thiazoles
heterocyclization
isothioureas
lung carcinoma A549
thiazolin-2-amines
thioureas
verubulin
Abstract
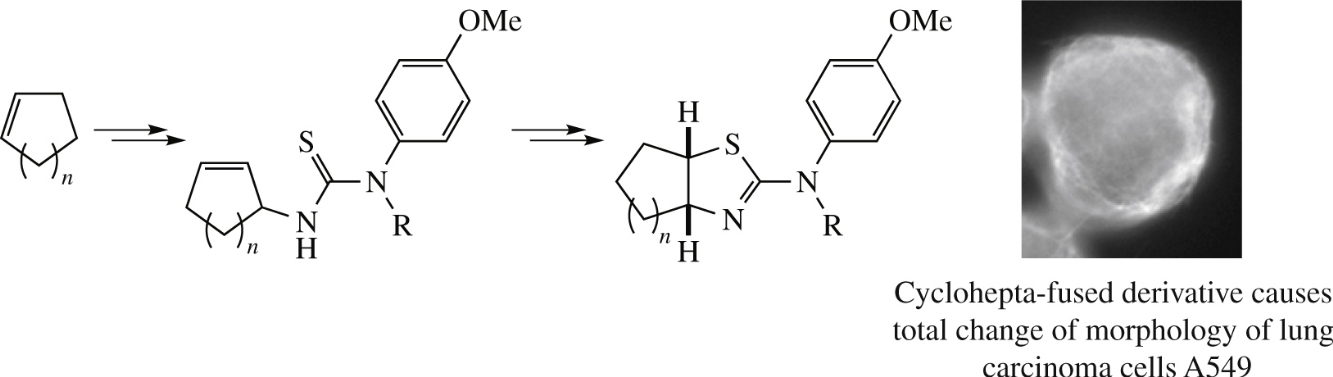
Bicyclic isothioureas of N-(4-methoxyphenyl)-2-aminocycloalkane[d]thiazole type were obtained using intramolecular electrophilic cyclization of N-(cycloalk-2-enyl)-N′-(4-methoxyphenyl)thioureas. Isothiourea fused with sevenmembered ring caused noticeable changes of the morphology of human lung carcinoma cells A549, but without affecting their microtubule net.
References
1.

Langdon S.R., Ertl P., Brown N.
Molecular Informatics,
2010
2.

Patani G.A., LaVoie E.J.
Chemical Reviews,
1996
3.

Meanwell N.A.
Journal of Medicinal Chemistry,
2011
4.
10.1016/j.mencom.2021.04.003_bib0020
The Practice of Medicinal Chemistry,
2003
5.

Lovering F., Bikker J., Humblet C.
Journal of Medicinal Chemistry,
2009
6.

Grembecka J., He S., Shi A., Purohit T., Muntean A.G., Sorenson R.J., Showalter H.D., Murai M.J., Belcher A.M., Hartley T., Hess J.L., Cierpicki T.
Nature Chemical Biology,
2012
7.

Giraudo A., Krall J., Bavo F., Nielsen B., Kongstad K.T., Rolando B., De Blasio R., Gloriam D.E., Löffler R., Thiesen L., Harpsøe K., Frydenvang K., Boschi D., Wellendorph P., Lolli M.L., et. al.
Journal of Medicinal Chemistry,
2019
8.

Ueda S., Terauchi H., Yano A., Matsumoto M., Kubo T., Kyoya Y., Suzuki K., Ido M., Kawasaki M.
Bioorganic and Medicinal Chemistry,
2004
9.

Sirisoma N., Pervin A., Zhang H., Jiang S., Willardsen J.A., Anderson M.B., Mather G., Pleiman C.M., Kasibhatla S., Tseng B., Drewe J., Cai S.X.
Journal of Medicinal Chemistry,
2009
10.

Wang S., Wang X., Qin B., Ohkoshi E., Hsieh K., Hamel E., Cui M., Zhu D., Goto M., Morris-Natschke S.L., Lee K., Xie L.
Bioorganic and Medicinal Chemistry,
2015
11.

McLoughlin E.C., O’Boyle N.M.
Pharmaceuticals,
2020
12.

Gracheva I.A., Shchegravina E.S., Schmalz H., Beletskaya I.P., Fedorov A.Y.
Journal of Medicinal Chemistry,
2020
13.

Current Medicinal Chemistry
14.

Haider K., Rahaman S., Yar M.S., Kamal A.
Expert Opinion on Therapeutic Patents,
2019
15.

La Regina G., Coluccia A., Naccarato V., Silvestri R.
European Journal of Pharmaceutical Sciences,
2019
16.
Nurieva E.V., Trofimova T.P., Alexeev A.A., Proshin A.N., Chesnakova E.A., Grishin Y.K., Lyssenko K.A., Filimonova M.V., Bachurin S.O., Zefirova O.N.
Mendeleev Communications,
2018
17.
Alexeev A.A., Nurieva E.V., Trofimova T.P., Chesnakova E.A., Grishin Y.K., Lyssenko K.A., Filimonova M.V., Zefirova O.N.
Mendeleev Communications,
2019
18.
![Novel structural type of bridged urea derivatives bearing azatricyclo[4.3.1.03,8]decane moiety](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Alexeev A.A., Nurieva E.V., Lyssenko K.A., Grishin Y.K., Zefirova O.N.
Structural Chemistry,
2018
19.
Nurieva E.V., Alexeev A.A., Grishin Y.K., Tafeenko V.A., Zefirova O.N.
Mendeleev Communications,
2020
20.

Al-Haddad A., Shonn M.A., Redlich B., Blocker A., Burkhardt J.K., Yu H., Hammer J.A., Weiss D.G., Steffen W., Griffiths G., Kuznetsov S.A.
Molecular Biology of the Cell,
2001
21.
Zefirov N.A., Evteeva Y.A., Krasnoperova A.I., Mamaeva A.V., Milaeva E.R., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2020
22.
Nurieva E.V., Zefirov N.A., Fritsch N., Milaeva E.R., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2020