Keywords
Abstract
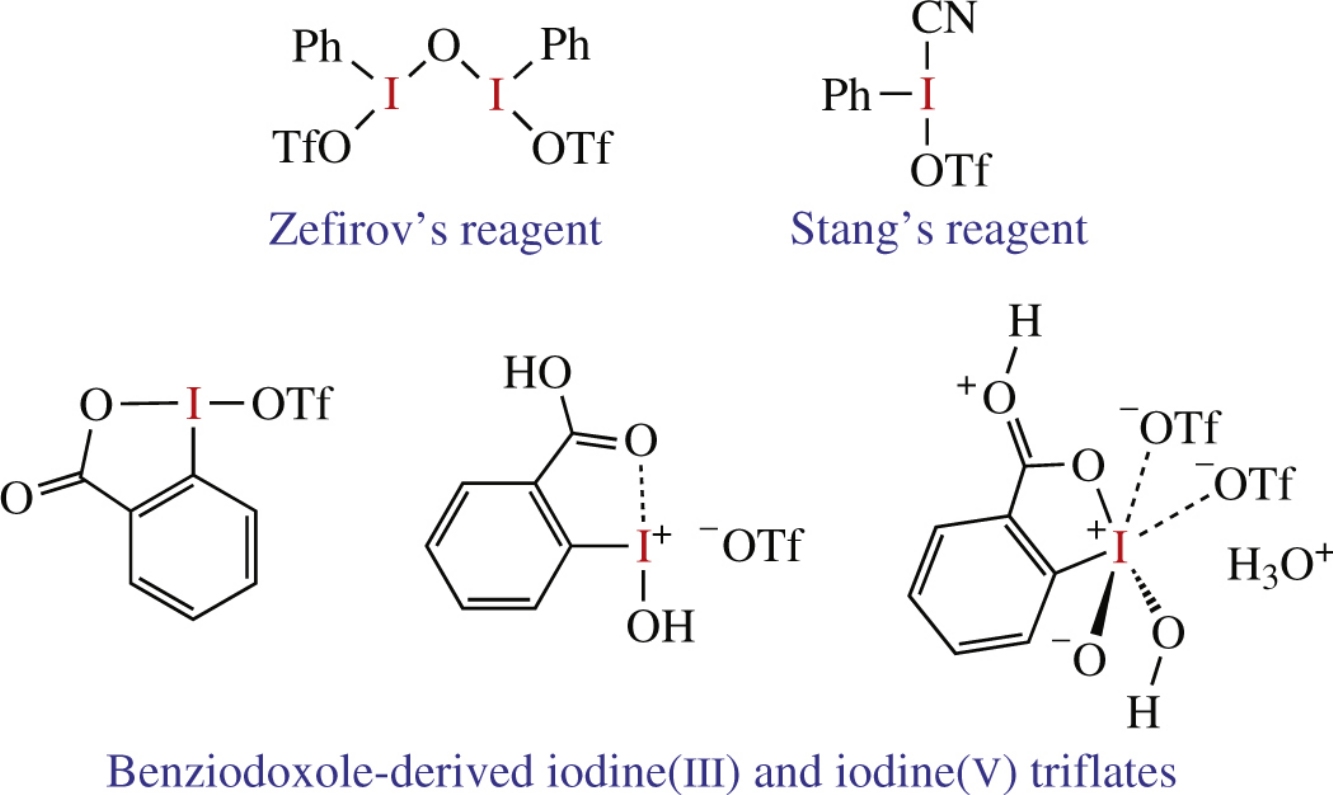
This minireview describes chemistry of hypervalent organoiodine triflates, which have received wide synthetic application as powerful electrophilic reagents and oxidants. The first representative of these compounds, μ-oxo-bis[(trifluoromethanesulfonato)(phenyl)iodine], was originally prepared and investigated in N. S. Zefirov's laboratory at Moscow State University in the early 1980s. This compound, now commonly known as Zefirov's reagent, is a useful reagent for the synthesis of various iodonium salts from the corresponding organic precursors. Recently, thermally stable and highly reactive triflates derived from cyclic hypervalent iodine compounds, benziodoxoles, have been reported and utilized in organic synthesis. The strongest iodine(V) oxidant, IBX-ditriflate, has been prepared from 2-iodoxybenzoic acid (IBX) and triflic acid. IBX-ditriflate can readily oxidize organic substrates that are generally resistant to oxidation.
References









![Conversion of olefins to ditriflates by .mu.-oxobis[(trifluoromethanesulfonato)(phenyl)iodine]](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)











![Selective ortho C−H Cyanoalkylation of (Diacetoxyiodo)arenes through [3,3]-Sigmatropic Rearrangement](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)

![Asymmetric Iodonio-[3,3]-Sigmatropic Rearrangement to Access Chiral α-Aryl Carbonyl Compounds](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)