Keywords
o-quinone
catechol
cyclic voltammetry
oxidation
pyrocatechuic acid
X-ray
Abstract
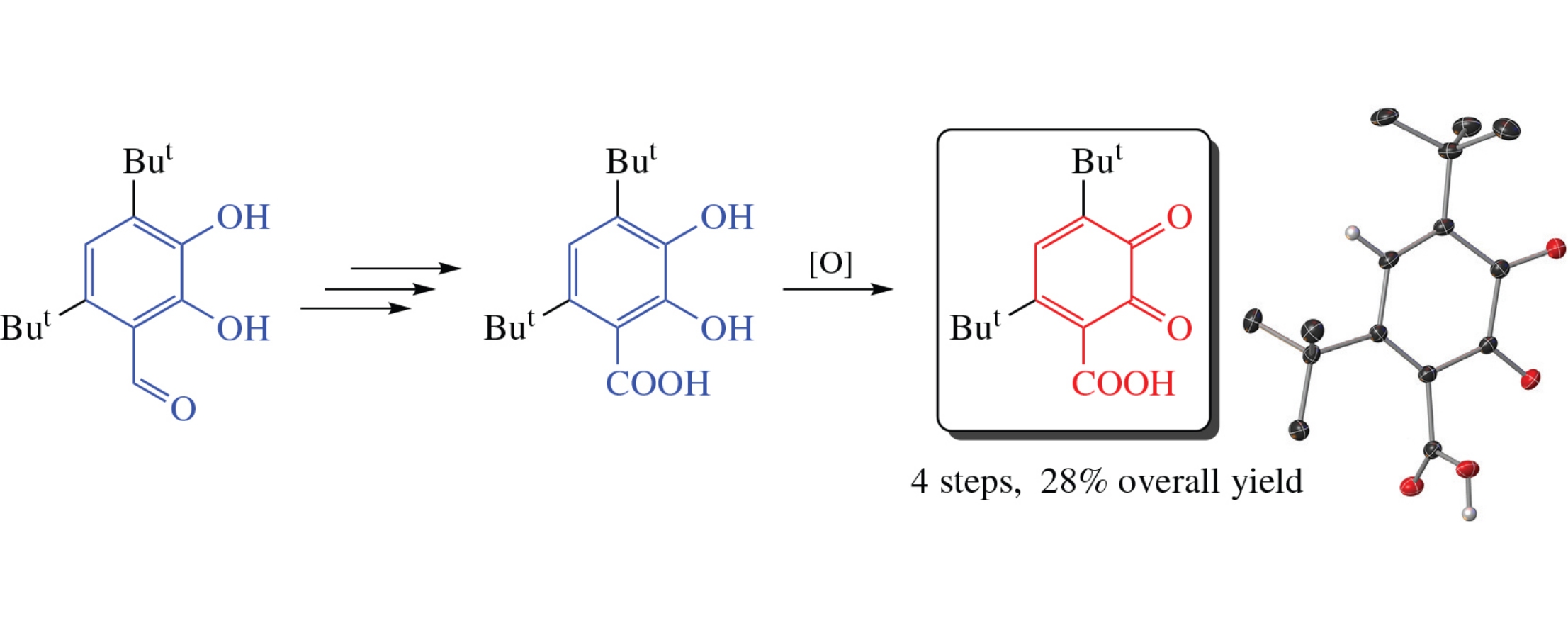
2,4-Di-tert-butyl-5,6-dioxocyclohexa-1,3-diene-1-carboxylic acid, an oxidized analogue of sterically hindered pyrocatechuic acid, was obtained from 4,6-di-tert-butyl-2,3-dihydroxybenzaldehyde in four stages. The electrochemical reduction of this compound proceeds in two stages: the first reduction wave (Ered1=−0.24V) is irreversible, while the second stage is partially reversible (E1/2, red2=−0.65V). The molecular structure of the o-quinone carboxylic acid in the crystalline state was comfirmed by single-crystal X-ray analysis.
References
1.

Aoki T., Yoshizawa H., Yamawaki K., Yokoo K., Sato J., Hisakawa S., Hasegawa Y., Kusano H., Sano M., Sugimoto H., Nishitani Y., Sato T., Tsuji M., Nakamura R., Nishikawa T., et. al.
European Journal of Medicinal Chemistry,
2018
2.

Liu R., Miller P.A., Vakulenko S.B., Stewart N.K., Boggess W.C., Miller M.J.
Journal of Medicinal Chemistry,
2018
3.

Hayashi T., Osawa A., Watanabe T., Murata Y., Nakayama A., Namba K.
Tetrahedron Letters,
2017
4.

Neumann W., Nolan E.M.
Journal of Biological Inorganic Chemistry,
2018
5.

Ivanović N., Jovanović L., Marković Z., Marković V., Joksović M.D., Milenković D., Djurdjević P.T., Ćirić A., Joksović L.
ChemistrySelect,
2016
6.

Mihailović N., Marković V., Matić I.Z., Stanisavljević N.S., Jovanović Ž.S., Trifunović S., Joksović L.
RSC Advances,
2017
7.
![Majority-Rules Effect and Allostery in Molecular Recognition of Calix[4]arene-Based Triple-Stranded Metallohelicates](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Yamasaki Y., Shio H., Amimoto T., Sekiya R., Haino T.
Chemistry - A European Journal,
2018
8.

Hart-Cooper W.M., Zhao C., Triano R.M., Yaghoubi P., Ozores H.L., Burford K.N., Toste F.D., Bergman R.G., Raymond K.N.
Chemical Science,
2015
9.

Hong C.M., Kaphan D.M., Bergman R.G., Raymond K.N., Toste F.D.
Journal of the American Chemical Society,
2017
10.

Pierre V.C., Botta M., Aime S., Raymond K.N.
Journal of the American Chemical Society,
2006
11.

Levin M.D., Kaphan D.M., Hong C.M., Bergman R.G., Raymond K.N., Toste F.D.
Journal of the American Chemical Society,
2016
12.

Dalton D.M., Ellis S.R., Nichols E.M., Mathies R.A., Toste F.D., Bergman R.G., Raymond K.N.
Journal of the American Chemical Society,
2015
13.

Pham T.A., Altman A.B., Stieber S.C., Booth C.H., Kozimor S.A., Lukens W.W., Olive D.T., Tyliszczak T., Wang J., Minasian S.G., Raymond K.N.
Inorganic Chemistry,
2016
14.

Pham T.A., Xu J., Raymond K.N.
Journal of the American Chemical Society,
2014
15.

16.

Baramov T., Keijzer K., Irran E., Mösker E., Baik M., Süssmuth R.
Chemistry - A European Journal,
2013
17.

Günther K., Filby W.G., Eiben K.
Tetrahedron Letters,
1971
18.

Nematollahi D., Khoshsafar H.
Tetrahedron,
2009
19.
10.1016/j.mencom.2021.03.042_bib0095
Nematollahi
J. Chem. Sci. (Amritsar, India),
2011
20.
Arsenyev M.V., Baranov E.V., Fedorov A.Y., Chesnokov S.A., Abakumov G.A.
Mendeleev Communications,
2015
21.
Arsenyev M.V., Astafeva T.V., Baranov E.V., Poddelsky A.I., Chesnokov S.A.
Mendeleev Communications,
2018
22.
Zhiganshina E.R., Arsenyev M.V., Shavyrin A.S., Baranov E.V., Chesnokov S.A.
Mendeleev Communications,
2019
23.

Kocherova T.N., Druzhkov N.O., Martyanov K.A., Shavyrin A.S., Arsenyev M.V., Kulikova T.I., Baranov E.V., Kuropatov V.A., Cherkasov V.K.
Russian Chemical Bulletin,
2020
24.

Arsenyev M.V., Baranov E.V., Chesnokov S.A., Cherkasov V.K., Abakumov G.A.
Russian Chemical Bulletin,
2013
25.

Arsenyev M.V., Khamaletdinova N.M., Baranov E.V., Chesnokov S.A., Cherkasov V.K.
Russian Chemical Bulletin,
2016
26.

Druzhkov N.O., Egorova E.N., Arsen’ev M.V., Baranov E.V., Cherkasov V.K.
Russian Chemical Bulletin,
2016
27.
Arsenyev M.V., Baranov E.V., Shurygina M.P., Chesnokov S.A., Abakumov G.A.
Mendeleev Communications,
2016
28.

Baryshnikova S.V., Bellan E.V., Poddel'sky A.I., Arsenyev M.V., Smolyaninov I.V., Fukin G.K., Piskunov A.V., Berberova N.T., Cherkasov V.K., Abakumov G.A.
European Journal of Inorganic Chemistry,
2016
29.

Do T.H., Brown S.N.
Dalton Transactions,
2019
30.

Do T.H., Brown S.N.
Dalton Transactions,
2020
31.

Astaf’eva T.V., Arsenyev M.V., Rumyantcev R.V., Fukin G.K., Cherkasov V.K., Poddel’sky A.I.
ACS Omega,
2020
32.

Bogdanov A.V., Iskhakova K.R., Voloshina A.D., Sapunova A.S., Kulik N.V., Terekhova N.V., Arsenyev M.V., Ziyatdinova G.K., Bukharov S.V.
Chemistry and Biodiversity,
2020
33.

René A., Evans D.H.
Journal of Physical Chemistry C,
2012
34.
CrysAlisPro 1.171.38.46, Data Collection, Reduction and Correction Program, Rigaku Oxford Diffraction, 2015.
35.
CrysAlisPro 1.171.38.46, SCALE3 ABSPACK: Empirical Absorption Correction, Rigaku Oxford Diffraction, 2015.
36.
10.1016/j.mencom.2021.03.042_bib0180
Sheldrick
Acta Crystallogr.,
2015
37.
G. M. Sheldrick, Structure Determination Software Suite, Bruker AXS, Madison, WI, 2003.