Keywords
N,O-acetals
aminals
arylamines
chroman-2-amines
nucleophilic substitution
transacetalization
transamination
Abstract
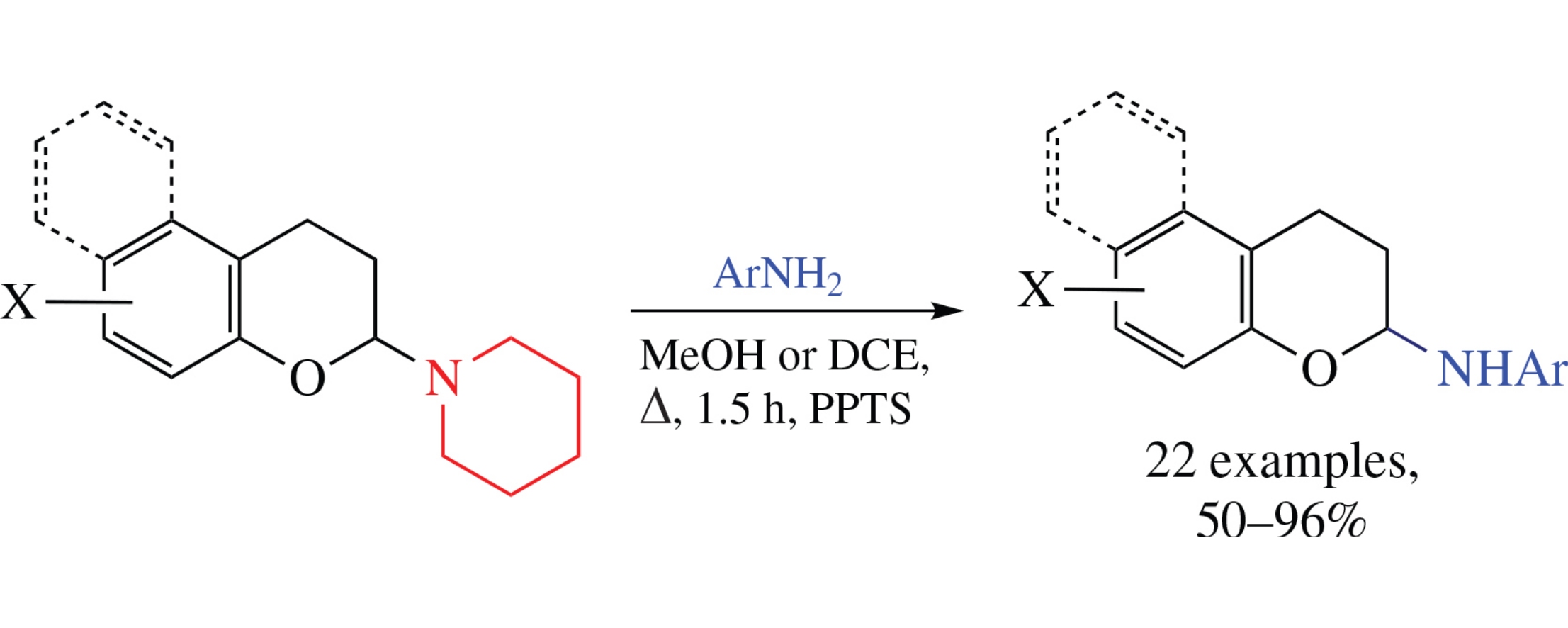
Reaction between 2-piperidinochromane and (hetero)aromatic amines in the presence of pyridinium tosylate affords the products of replacement of the piperidine fragment by the (hetero)arylamino moiety in good yields. This single-stage process opens facile and efficient way to the functionalized N-arylchroman-2-amines.
References
1.
![Synthesis of 5H-Dibenzo[c,g]chromen-5-ones via FeCl3-Mediated Tandem C–O Bond Cleavage/6π Electrocyclization/Oxidative Aromatization](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Miao M., Jin M., Xu H., Chen P., Zhang S., Ren H.
Organic Letters,
2018
2.
Levchenko K.S., Chudov K.A., Zinoviev E.V., Lyssenko K.A., Fakhrutdinov A.N., Demin D.U., Poroshin N.O., Zhukova A.A., Shmelin P.S., Grebennikov E.P.
Mendeleev Communications,
2019
3.

Levchenko K.S., Chudov K.A., Demin D.Y., Adamov G.E., Zinoviev E.V., Lyssenko K.A., Shokurov A.V., Shmelin P.S., Grebennikov E.P.
Russian Chemical Bulletin,
2019
4.

Korotaev V.Y., Kutyashev I.B., Barkov A.Y., Sosnovskikh V.Y.
Russian Chemical Reviews,
2019
5.
![Synthesis and pharmacological evaluation of 2,5-cycloamino-5H-[1]benzopyrano[4,3-d]pyrimidines endowed with in vitro antiplatelet activity](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Bruno O., Brullo C., Ranise A., Schenone S., Bondavalli F., Barocelli E., Ballabeni V., Chiavarini M., Tognolini M., Impicciatore M.
Bioorganic and Medicinal Chemistry Letters,
2001
6.

Bruno O., Schenone S., Ranise A., Bondavalli F., Barocelli E., Ballabeni V., Chiavarini M., Bertoni S., Tognolini M., Impicciatore M.
Bioorganic and Medicinal Chemistry,
2001
7.

Jiang N., Wang X., Yang Y., Dai W.
Mini-Reviews in Medicinal Chemistry,
2006
8.

Beyer C., Lin Q., Platt B., Malberg J., Hornby G., Sullivan K., Smith D., Lock T., Mitchell P., Hatzenbuhler N., Evrard D., Harrison B., Magolda R., Pangalos M., Schechter L., et. al.
British Journal of Pharmacology,
2009
9.

Li Q., Zhang M., Hallis T.M., DeRosier T.A., Yue J., Ye Y., Mais D.E., Wang M.
Biochemical and Biophysical Research Communications,
2010
10.

Mori T., Akioka Y., Onodera G., Kimura M.
Molecules,
2014
11.

Wang X., Liu Y., Ma R., Si C., Wei B.
Journal of Organic Chemistry,
2019
12.

Yu C., Huang H., Li X., Zhang Y., Li H., Wang W.
Chemistry - A European Journal,
2016
13.

Xu G., Feng Z., Xu J., Wang Z., Qin Y., Xu P.
Chemistry - A European Journal,
2018
14.

Lukashenko A.V., Osipov D.V., Osyanin V.А., Klimochkin Y.N.
Chemistry of Heterocyclic Compounds,
2020
15.

Osyanin V.A., Lukashenko A.V., Osipov D.V.
Russian Chemical Reviews,
2021
16.

He L., Zhao L., Wang D., Wang M.
Organic Letters,
2014
17.

He L., Liu H., Zhao L., Wang D., Wang M.
Tetrahedron,
2015
18.

Liu J., Xu J., Li Z., Huang Y., Wang H., Gao Y., Guo T., Ouyang P., Guo K.
European Journal of Organic Chemistry,
2017
19.

Liu Y., Li Z., Li J., Feng H., Tong Z.
Organic Letters,
2015
20.

Semenova I.А., Korzhenko K.S., Osipov D.V., Osyanin V.А., Klimochkin Y.N.
Chemistry of Heterocyclic Compounds,
2020
21.

Osyanin V.A., Osipov D.V., Melnikova I.V., Korzhenko K.S., Semenova I.A., Klimochkin Y.N.
Synthesis,
2020
22.

Osipov D.V., Melnikova I.V., Korzhenko K.S., Osyanin V.A., Klimochkin Y.N.
Chemistry of Heterocyclic Compounds,
2020