Keywords
acridines
alkylation
anilines
catechols
heterocyclization
quinone methides
X-ray diffraction analysis
Abstract
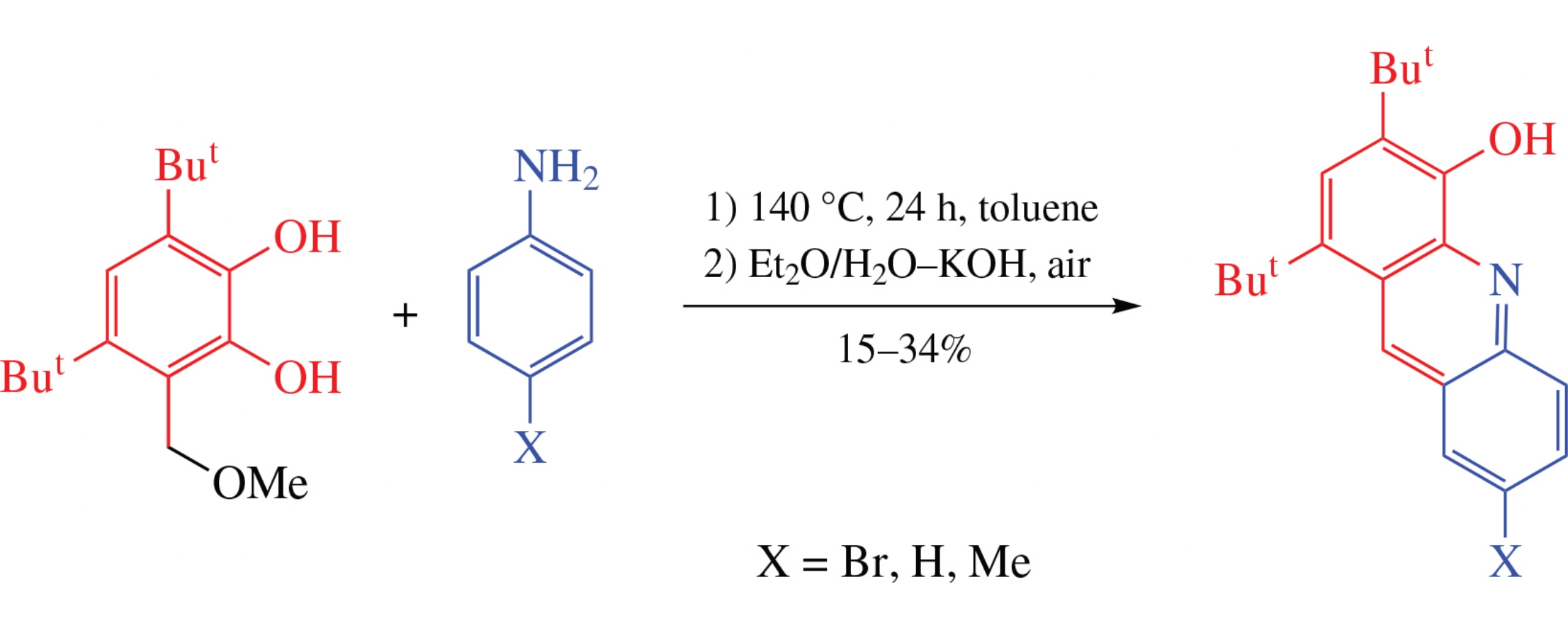
Three new sterically hindered acridin-4-ols have been prepared by alkylation of anilines with 3,5-di-tert-butyl-6-methoxymethylcatechol followed by oxidation of the reaction mixture. Formation of the acridine moiety was found to occur in the course of oxidation of the intermediate (anilinomethyl)catechol on contact with air in the Et2O/H2O–KOH system. The molecular structure of two acridin-4-ols was determined by single-crystal X-ray diffraction.
References
1.

Percivalle C., Mahmood T., Ladame S.
MedChemComm,
2013
2.

Rapi Z., Bakó P., Keglevich G., Baranyai P., Kubinyi M., Varga O.
Journal of Inclusion Phenomena and Macrocyclic Chemistry,
2014
3.

Kertész J., Huszthy P., Kormos A., Bezúr L.
Tetrahedron,
2011
4.
![Physicochemical and Electronic Properties of Cationic [6]Helicenes: from Chemical and Electrochemical Stabilities to Far-Red (Polarized) Luminescence](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bosson J., Labrador G.M., Pascal S., Miannay F., Yushchenko O., Li H., Bouffier L., Sojic N., Tovar R.C., Muller G., Jacquemin D., Laurent A.D., Le Guennic B., Vauthey E., Lacour J., et. al.
Chemistry - A European Journal,
2016
5.

Wang Y., Huang Z., Leng X., Zhu H., Liu G., Huang Z.
Journal of the American Chemical Society,
2018
6.

Ando S., Fukushima T., Yamaguchi T.
Chemistry of Materials,
2016
7.

Su Q., Li P., He M., Wu Q., Ye L., Mu Y.
Organic Letters,
2013
8.

Wen J., Tang S., Zhang F., Shi R., Lei A.
Organic Letters,
2016
9.

Szymanska A., Wiczk W., Lankiewicz L.
Chemistry of Heterocyclic Compounds,
2000
10.
![Cycloisomerization – a straightforward way to benzo[h]quinolines and benzo[c]acridines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Shestakov A.N., Pankova A.S., Kuznetsov M.A.
Chemistry of Heterocyclic Compounds,
2017
11.

12.

Brikci-Nigassa N.M., Bentabed-Ababsa G., Erb W., Chevallier F., Picot L., Vitek L., Fleury A., Thiéry V., Souab M., Robert T., Ruchaud S., Bach S., Roisnel T., Mongin F.
Tetrahedron,
2018
13.

Mastropietro T.F., Szerb E.I., La Deda M., Crispini A., Ghedini M., Aiello I.
European Journal of Inorganic Chemistry,
2013
14.

Crispini A., Pucci D., Sessa S., Cataldi A., Napoli A., Valentini A., Ghedini M.
New Journal of Chemistry,
2003
15.

Paul G.C., Sarkar P., Mukherjee C.
Inorganica Chimica Acta,
2019
16.
Arsenyev M.V., Baranov E.V., Shurygina M.P., Chesnokov S.A., Abakumov G.A.
Mendeleev Communications,
2016
17.
Zhiganshina E.R., Arsenyev M.V., Shavyrin A.S., Baranov E.V., Chesnokov S.A.
Mendeleev Communications,
2019
18.

Poddel'sky A.I., Arsenyev M.V., Astaf'eva T.V., Chesnokov S.A., Fukin G.K., Abakumov G.A.
Journal of Organometallic Chemistry,
2017
19.

Smolyaninov I.V., Poddel’sky A.I., Smolyaninova S.A., Arsenyev M.V., Fukin G.K., Berberova N.T.
Molecules,
2020
20.
Arsenyev M.V., Astafeva T.V., Baranov E.V., Poddelsky A.I., Chesnokov S.A.
Mendeleev Communications,
2018
21.

Asherson J.L., Bilgic O., Young D.W.
Journal of the Chemical Society Perkin Transactions 1,
1980
22.

Radhika S., Saranya S., Harry N.A., Anilkumar G.
ChemistrySelect,
2019
23.

Astaf’eva T.V., Arsenyev M.V., Rumyantcev R.V., Fukin G.K., Cherkasov V.K., Poddel’sky A.I.
ACS Omega,
2020
24.
CrysAlisPro 1.171.40.71a, Data Collection, Reduction and Correction Program, Rigaku Oxford Diffraction, 2020.
25.
SAINT, Data Reduction and Correction Program, Bruker AXS, Madison, WI, 2014.
26.

Krause L., Herbst-Irmer R., Sheldrick G.M., Stalke D.
Journal of Applied Crystallography,
2015
27.

Clark R.C., Reid J.S.
Acta Crystallographica Section A Foundations of Crystallography,
1995
28.
10.1016/j.mencom.2021.03.040_bib0140
Sheldrick
Acta Crystallogr.,
2015
29.
10.1016/j.mencom.2021.03.040_bib0145
Sheldrick
Acta Crystallogr.,
2015
30.

Miozzo L., Papagni A., Casalbore-Miceli G., Del Buttero P., Girotti C., Moret M., Trabattoni S.
Chemistry of Materials,
2004
31.

Steiner T.
Angewandte Chemie - International Edition,
2002
32.

Papagni A., Del Buttero P., Moret M., Sassella A., Miozzo L., Ridolfi G.
Chemistry of Materials,
2003
33.

Chowdhury P., Panja S., Chakravorti S.
Journal of Physical Chemistry A,
2002
34.

Singh R.B., Mahanta S., Guchhait N.
Journal of Photochemistry and Photobiology A: Chemistry,
2008
35.

Bos R., Barnett N.W., Dyson G.A., Russell R.A.
Analytica Chimica Acta,
2002
36.
10.1016/j.mencom.2021.03.040_bib0180
Mataga
Molecular Interactions and Electronic Spectra,
1970
37.

Albrecht M., Fröhlich R.
Journal of the American Chemical Society,
1997