Keywords
N-arylitaconimides
alkyl (3-oxopiperazin-2-ylidene)acetates
microwave irradiation
pyrido[1,2-a]pyrazin-1-ones
recyclization
Abstract
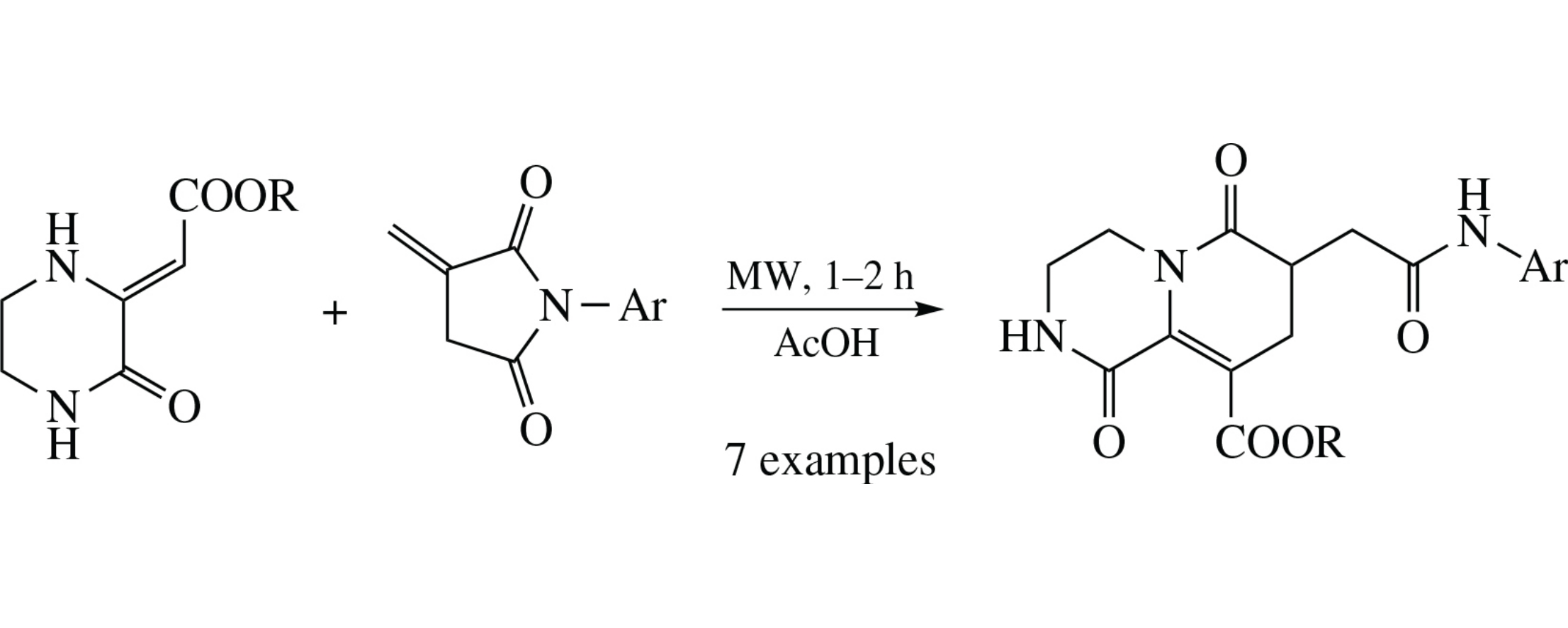
New polyfunctional hydrogenated pyrido[1,2-a]pyrazin- 1-ones were obtained by regioselective recyclization of N-arylitaconimides with alkyl (3-oxopiperazin-2-ylidene)acetates. The supposed cascade reaction pathway involves the Michael addition of the nucleophile to an activated double bond and subsequent intramolecular transamidation of the intermediate with simultaneous recycling.
References
1.

Podlogar B., Muegge I.
Current Topics in Medicinal Chemistry,
2001
2.
B. H. Lee and M. Kalamazoo, Patent WO 94/29319, 1994.
3.

Sakai R., Jares-Erijman E.A., Manzanares I., Silva Elipe M.V., Rinehart K.L.
Journal of the American Chemical Society,
1996
4.

Zewail-Foote M., Li V., Kohn H., Bearss D., Guzman M., Hurley L.H.
Chemistry & Biology,
2001
5.

Prangé T., Billion M., Vuilhorgne M., Pascard C., Polonsky J., Moreau S.
Tetrahedron Letters,
1981
6.

Pettit G.R., Knight J.C., Collins J.C., Herald D.L., Pettit R.K., Boyd M.R., Young V.G.
Journal of Natural Products,
2000
7.

Scott J.D., Williams R.M.
Chemical Reviews,
2002
8.
![Agents acting on the central nervous system. XIII. 2,3,4,4a,5,6-Hexahydro-1(H)-pyrazino[1,2-a]quinolines. A new class of hypotensive agents](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Rao V.A., Jain P.C., Anand N., Srimal R.C., Dua P.R.
Journal of Medicinal Chemistry,
1970
9.
C. W. am Ende, J.M. Humphrey, D.S. Johnson, G.W. Kauffman, M.Y. Pettersson, D.A. Rankic, A.F. Stepan and P. R. Verhoest, Patent WO 2015/150957, 2015.
10.
C. C. Kong and B. Liu, Patent WO 2006/066414, 2006.
11.

Yoshinaga T., Kobayashi M., Seki T., Miki S., Wakasa-Morimoto C., Suyama-Kagitani A., Kawauchi-Miki S., Taishi T., Kawasuji T., Johns B.A., Underwood M.R., Garvey E.P., Sato A., Fujiwara T.
Antimicrobial Agents and Chemotherapy,
2015
12.

Kobayashi M., Yoshinaga T., Seki T., Wakasa-Morimoto C., Brown K.W., Ferris R., Foster S.A., Hazen R.J., Miki S., Suyama-Kagitani A., Kawauchi-Miki S., Taishi T., Kawasuji T., Johns B.A., Underwood M.R., et. al.
Antimicrobial Agents and Chemotherapy,
2010
13.

Cai G., Zhu W., Ma D.
Tetrahedron,
2006
14.
![A novel method for the synthesis of pyrimido[1,2-a]benzimidazoles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Vandyshev D.Y., Shikhaliev K.S., Kokonova A.V., Potapov A.Y., Kolpakova M.G., Sabynin A.L., Zubkov F.I.
Chemistry of Heterocyclic Compounds,
2016
15.
10.1016/j.mencom.2021.03.039_bib0075
Vandyshev
J. Org. Chem.,
2017
16.

Kovygin Y.A., Shikhaliev K.S., Krysin M.Y., Potapov A.Y., Ledenyova I.V., Kosheleva Y.A., Vandyshev D.Y.
Chemistry of Heterocyclic Compounds,
2019
17.

18.

Ferris R.G., Hazen R.J., Roberts G.B., St. Clair M.H., Chan J.H., Romines K.R., Freeman G.A., Tidwell J.H., Schaller L.T., Cowan J.R., Short S.A., Weaver K.L., Selleseth D.W., Moniri K.R., Boone L.R., et. al.
Antimicrobial Agents and Chemotherapy,
2005
19.

Gu S., Zhang X., He Q., Yang L., Ma X., Zheng Y., Yang S., Chen F.
Bioorganic and Medicinal Chemistry,
2011
20.
![Synthesis of alkyl hexahydropyrazino-[1,2-c]pyrimidine-9-carboxylates](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Vovk M.V., Kushnir O.V., Melˈnichenko N.V., Tsymbal I.F.
Chemistry of Heterocyclic Compounds,
2011
21.

KAWAHARA N., SHIMAMORI T., ITOH T., TAKAYANAGI H., OGURA H.
Chemical and Pharmaceutical Bulletin,
2011
22.
![One-pot synthesis of pyrrolo[1,2-a]pyrazines via three component reaction of ethylenediamine, acetylenic esters and nitrostyrene derivatives](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Moradi L., Piltan M., Rostami H., Abasi G.
Chinese Chemical Letters,
2013
23.
![A one-pot catalyst-free synthesis of functionalized pyrrolo[1,2-a]quinoxaline derivatives from benzene-1,2-diamine, acetylenedicarboxylates and ethyl bromopyruvate](/storage/images/resized/ex6KJoZujZOZFZh7jGfeHauiftuB3CI7iwJVFRDg_small_thumb.webp)
Piltan M., Moradi L., Abasi G., Zarei S.A.
Beilstein Journal of Organic Chemistry,
2013