Keywords
2-cyanomethylbenzimidazole
heterocyclization
itaconimides
pyrido[1,2-a]benzimidazoles
recyclization
Abstract
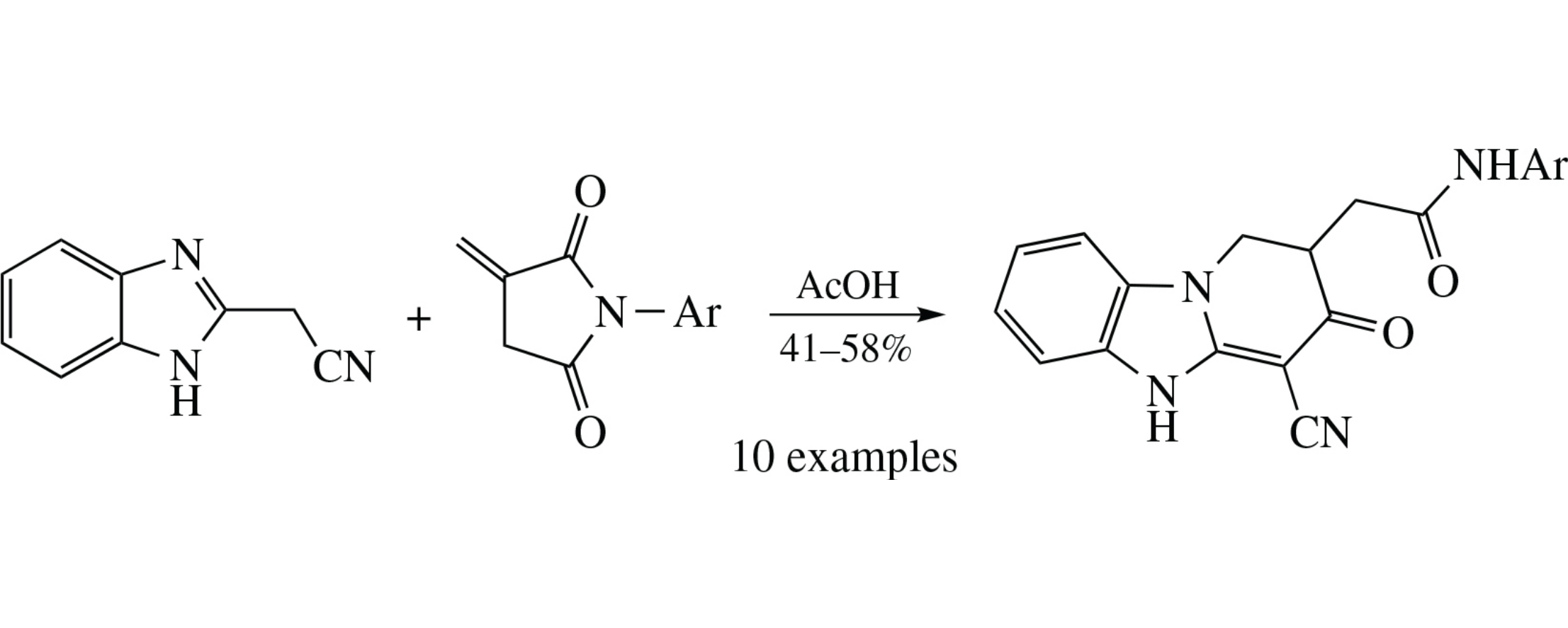
A convenient preparative synthesis of N-aryl-2-(4-cyano-3-oxo-1,2,3,5-tetrahydropyrido[1,2-a]benzimidazol-2-yl)acetamides is based on the reflux of N-arylitaconimides with 2-cyanomethylbenzimidazole in acetic acid.
References
1.

Kitamura A., Someya K., Hata M., Nakajima R., Takemura M.
Antimicrobial Agents and Chemotherapy,
2009
2.
K. I. N. Kerkvlie and S. Kolluri, Patent WO 2016/069780 Al, 2016.
3.

Sondhi S.M., Rani R., Singh J., Roy P., Agrawal S.K., Saxena A.K.
Bioorganic and Medicinal Chemistry Letters,
2010
4.

5.
![4-Deoxypyrido[1',2':1,2]imidazo[5,4-c]rifamycin SV derivatives. A new series of semisynthetic rifamycins with high antibacterial activity and low gastroenteric absorption](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Marchi E., Mascellani G., Montecchi L., Venturini A.P., Brufani M., Cellai L.
Journal of Medicinal Chemistry,
1985
6.
![Tautomerism and Physical Properties of Pyrido[1,2-a]benzimidazole (PBI) GABA-A Receptor Ligands](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Reitz A.B., Gauthier D.A., Ho W., Maryanoff B.E.
Tetrahedron,
2000
7.
![Amino substituted benzimidazo[1,2- a ]quinolines: Antiproliferative potency, 3D QSAR study and DNA binding properties](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Perin N., Nhili R., Cindrić M., Bertoša B., Vušak D., Martin-Kleiner I., Laine W., Karminski-Zamola G., Kralj M., David-Cordonnier M., Hranjec M.
European Journal of Medicinal Chemistry,
2016
8.

He L., Yang D., Li S., Zhang Y., Tang Y., Lei J., Frett B., Lin H., Li H., Chen Z., Xu Z.
Bioorganic and Medicinal Chemistry,
2018
9.

Liang G., Yu Y., Wei J., Kuang W., Chen Z., Zhang Y.
European Journal of Medicinal Chemistry,
2020
10.
![Potential anxiolytic agents. 3. Novel A-ring modified pyrido[1,2-a]benzimidazoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Maryanoff B.E., Nortey S.O., McNally J.J., Sanfilippo P.J., McComsey D.F., Dubinsky B., Shank R.P., Reitz A.B.
Bioorganic and Medicinal Chemistry Letters,
1999
11.

Mamada M., Pérez-Bolívar C., Kumaki D., Esipenko N.A., Tokito S., Anzenbacher P.
Chemistry - A European Journal,
2014
12.

Liu Q., Fang Y., Yi X., Chen R., Zhang Y.
Asian Journal of Chemistry,
2013
13.

Tozlu C., Erten-Ela S., Icli S.
Sensors and Actuators, A: Physical,
2010
14.

15.

Ortiz R.P., Herrera H., Blanco R., Huang H., Facchetti A., Marks T.J., Zheng Y., Segura J.L.
Journal of the American Chemical Society,
2010
16.
M. Engel, W. Fröhner, A. Stroba and R. M. Biondi, Patent WO 2010/043711 A1, 2010.
17.

Sun X., Lv X., Ye L., Hu Y., Chen Y., Zhang X., Yan M.
Organic and Biomolecular Chemistry,
2015
18.

Liao W., Li S., Wang J., Zhang Z., Yang Z., Xu D., Xu C., Lan H., Chen Z., Xu Z.
ACS Combinatorial Science,
2015
19.
![A novel method for the synthesis of pyrimido[1,2-a]benzimidazoles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Vandyshev D.Y., Shikhaliev K.S., Kokonova A.V., Potapov A.Y., Kolpakova M.G., Sabynin A.L., Zubkov F.I.
Chemistry of Heterocyclic Compounds,
2016
20.
![A novel synthetic approach to hydroimidazo[1,5-b]pyridazines by the recyclization of itaconimides and HPLC–HRMS monitoring of the reaction pathway](/storage/images/resized/ex6KJoZujZOZFZh7jGfeHauiftuB3CI7iwJVFRDg_small_thumb.webp)
Vandyshev D.Y., Shikhaliev K.S., Potapov A.Y., Krysin M.Y., Zubkov F.I., Sapronova L.V.
Beilstein Journal of Organic Chemistry,
2017
21.

22.

Ferris R.G., Hazen R.J., Roberts G.B., St. Clair M.H., Chan J.H., Romines K.R., Freeman G.A., Tidwell J.H., Schaller L.T., Cowan J.R., Short S.A., Weaver K.L., Selleseth D.W., Moniri K.R., Boone L.R., et. al.
Antimicrobial Agents and Chemotherapy,
2005
23.

Gu S., Zhang X., He Q., Yang L., Ma X., Zheng Y., Yang S., Chen F.
Bioorganic and Medicinal Chemistry,
2011
24.
![Benzimidazole condensed ring systems. 2. New synthesis of substituted 1-oxo-1H,5H-pyrido[1,2-a]benzimidazole-4-carbonitriles and related derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Rida S.M., Soliman F.S., Badawey E.A., Kappe T.
Journal of Heterocyclic Chemistry,
1988
25.

Rational Design and Synthesis of Thioridazine Analogues as Enhancers of the Antituberculosis Therapy
Pieroni M., Machado D., Azzali E., Santos Costa S., Couto I., Costantino G., Viveiros M.
Journal of Medicinal Chemistry,
2015
26.

Cai Q., Li Z., Wei J., Fu L., Ha C., Pei D., Ding K.
Organic Letters,
2010
27.

Kovygin Y.A., Shikhaliev K.S., Krysin M.Y., Potapov A.Y., Ledenyova I.V., Kosheleva Y.A., Vandyshev D.Y.
Chemistry of Heterocyclic Compounds,
2019
28.
![Synthesis of condensed pyrrolo[b]pyrazines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Volovenko Y.M., Dubinina G.G.
Chemistry of Heterocyclic Compounds,
1999
29.
![Synthesis and X-ray crystal structure of pyrrolo[1,2-a]benzimidazoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Awadallah A.M., Seppelt K., Shorafa H.
Tetrahedron,
2006
30.

Dawood K.M., Elwan N.M., Farahat A.A., Abdel-Wahab B.F.
Journal of Heterocyclic Chemistry,
2010
31.

Rudenko R.V., Komykhov S.A., Musatov V.I., Konovalova I.S., Shishkin O.V., Desenko S.M.
Journal of Heterocyclic Chemistry,
2011