Keywords
acetylene–allene rearrangement
cotarnine acetylene derivatives
iodomethylation
isoquinolinium compounds
quantum-chemical calculations
X-ray
Abstract
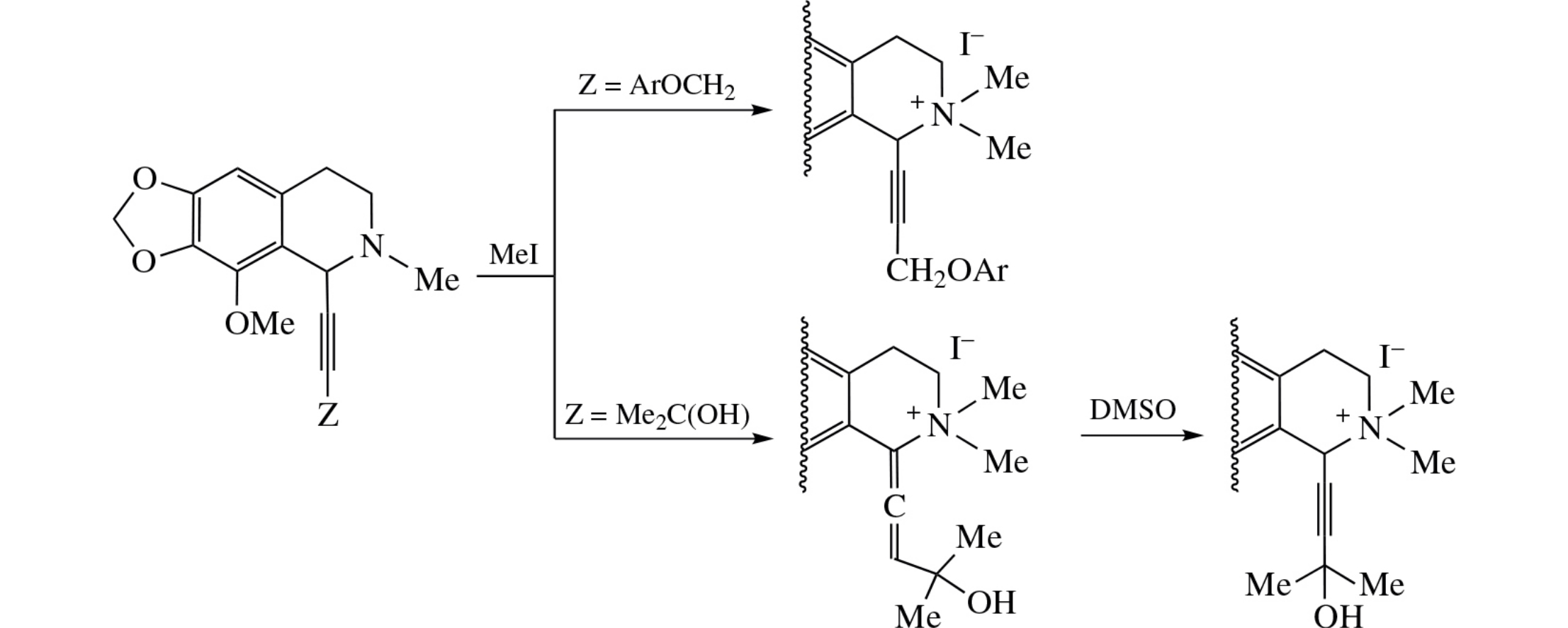
N-Methylation of cotarnine–phenol propargyl ethers conjugates with MeI affords the corresponding quaternary ammonium derivatives with retained acetylenic moiety. 3-Hydroxyalk-1-yn-1-yl analogues in the course of such iodomethylation undergo isomerization into allenic derivatives which are transformed into acetylenic ones upon dissolution in DMSO.
References
1.
10.1016/j.mencom.2021.03.036_bib0005
Mashkovsky
Lekarstvennye sredstva (Medicines),
2005
2.

Zhao D., Mao Y., Song S., Cheng M.
Heterocycles,
2013
3.

Zubenko A.A., Kartsev V.G., Morkovnik A.S., Divaeva L.N., Alexeenko D.V., Suponitsky K.Y., Borodkin G.S., Klimenko A.I.
Tetrahedron Letters,
2017
4.
10.1016/j.mencom.2021.03.036_bib0020
Anh
Acta Crystallogr.,
2017
5.

6.

Reddy Nagireddy P.K., Kommalapati V.K., Manchukonda N.K., Sridhar B., Tangutur A.D., Kantevari S.
ChemistrySelect,
2019
7.

Kartsev V.G., Zubenko A.A., Divaeva L.N., Morkovnik A.S., Baryshnikova T.K., Shirinian V.Z.
Russian Journal of General Chemistry,
2020
8.
L. Yu. Ukhin, K. Yu. Suponitskii and V. G. Kartsev, in Abstracts of the First International Conference ‘Chemistry and Biological Activity of Nitrogen Heterocycles and Alkaloids’, Moscow, 2001, vol. 1, p. 552.
9.

Ukhin L.Y., Suponitskii K.Y., Kartsev V.G.
Chemistry of Natural Compounds,
2003
10.

Ukhin L.Y., Gol'ding I.R., Kartsev V.G.
Chemistry of Natural Compounds,
2004
11.

Ukhin L.Y., Shepelenko E.N., Belousova L.V., Popova O.S., Alekseenko D.V.
Russian Chemical Bulletin,
2016
12.
10.1016/j.mencom.2021.03.036_bib0060
Temnikova
Molekulyarnye peregruppirovki v organicheskoi khimii (Molecular Rearrangements in Organic Chemistry),
1983
13.

Phadtare S., Kessel D., Corbett T.H., Renis H.E., Court B.E., Zemlicka J.
Journal of Medicinal Chemistry,
1991
14.

Phadtare S., Zemlicka J.
Journal of Organic Chemistry,
1989
15.

Chen Y., Wang L., Sun N., Xie X., Zhou X., Chen H., Li Y., Liu Y.
Chemistry - A European Journal,
2014
16.
10.1016/j.mencom.2021.03.036_sbref0080a
Smith
March's Advanced Organic Chemistry: Reactions, Mechanisms, and Structure,
2007
17.
10.1016/j.mencom.2021.03.036_sbref0080b
Smith
March's Advanced Organic Chemistry: Reactions, Mechanisms, and Structure,
2007
18.
10.1016/j.mencom.2021.03.036_sbref0080c
Smith
March's Advanced Organic Chemistry: Reactions, Mechanisms, and Structure,
2007
19.

Danilkina N.A., Vasileva A.A., Balova I.A.
Russian Chemical Reviews,
2020