Keywords
1,3,4-oxadiazoles
alicyclic compounds
organozinc compounds
pyrano[2,3-c]pyrazoles
pyrazol-3-ones
Reformatsky reaction
spiro compounds
X-ray analysis
Abstract
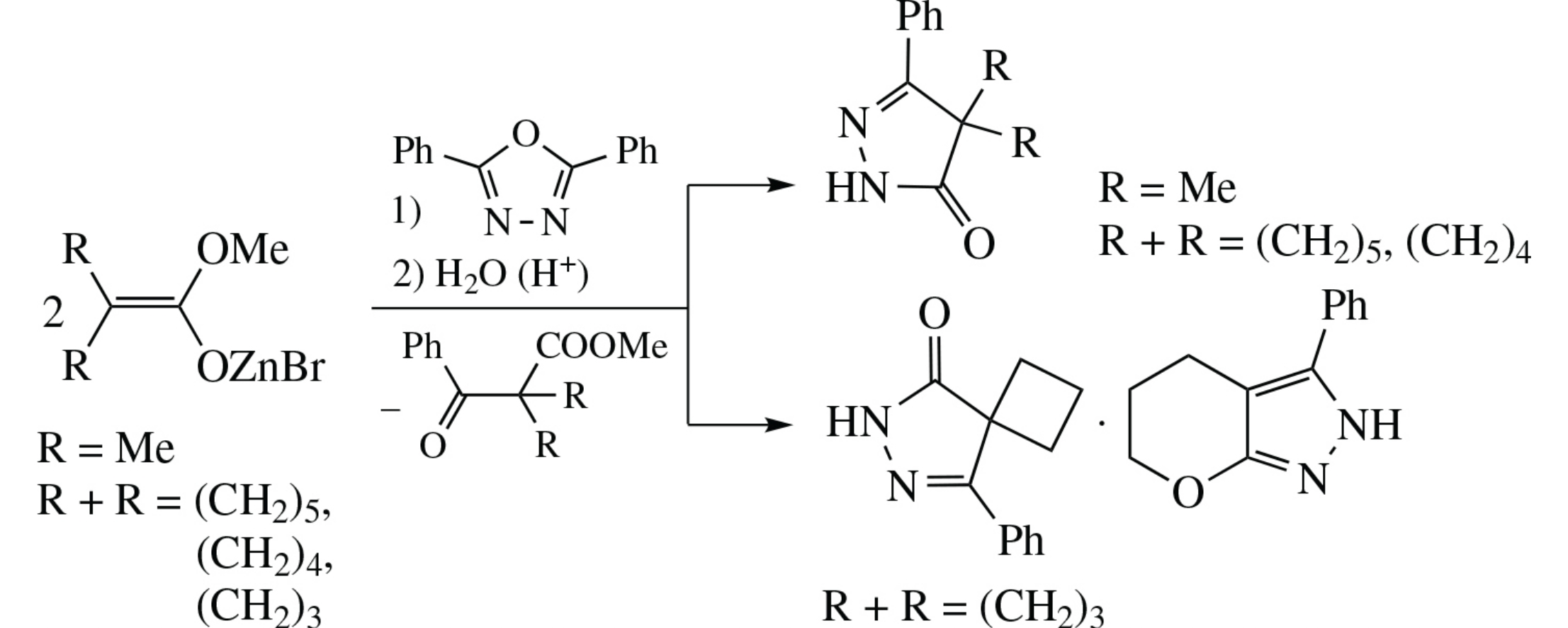
The reaction of methyl a-bromoisobutyrate or 1-bromocycloalkanecarboxylates with zinc and 2,5-diphenyl-1,3,4-oxadiazole leads to 5-phenyl-2,4-dihydro-3H-pyrazol-3-one derivatives, with the cycloalkane series affording products of spiro structure. In the case of cyclobutane reactant, 3-phenyl-2,4,5,6-tetrahydropyrano[2,3-c]pyrazole is also formed.
References
1.
10.1016/j.mencom.2021.03.035_bib0005
Dardoize
Bull. Soc. Chim. Fr.,
1969
2.

Brandi A., Cicchi S., Cordero F.M.
Chemical Reviews,
2008
3.

Kirillov N.F., Shchepin V.V.
Russian Journal of General Chemistry,
2005
4.
Nikiforova E.A., Kirillov N.F., Melekhin V.S., Slepukhin P.A.
Mendeleev Communications,
2019
5.

Singh G.S., D’hooghe M., De Kimpe N.
Tetrahedron,
2011
6.

Jarrahpour A., Rezaei S., Sinou V., Latour C., Brunel J.M.
Iranian Journal of Science and Technology, Transaction A: Science,
2016
7.

Synthesis of mono-, bis-spiro- and dispiro-β-lactams and evaluation of their antimalarial activities
Jarrahpour A., Ebrahimi E., De Clercq E., Sinou V., Latour C., Djouhri Bouktab L., Brunel J.M.
Tetrahedron,
2011
8.

Dagli D.J., Gorski R.A., Wemple J.
Journal of Organic Chemistry,
1975
9.

Etheredge S.J.
Journal of Organic Chemistry,
1966
10.
![Preparation of spiro[3.4]oct-1-ene](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Wilcox C.F., Whitney G.C.
Journal of Organic Chemistry,
1967
11.
CrysAlisPro Software System, version 1.171.37, Agilent Technologies, Yarnton, Oxfordshire, UK.
12.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
13.

Farrugia L.J.
Journal of Applied Crystallography,
2012
14.

Grimme S., Antony J., Ehrlich S., Krieg H.
Journal of Chemical Physics,
2010
15.
A. A. Granovsky, Firefly, version 8.2.0, http://classic.chem.msu.su/gran/firefly/index.html.