Keywords
Diels–Alder reaction
hydantoins
thiohydantoins
X-ray
α-amidoalkylation
Abstract
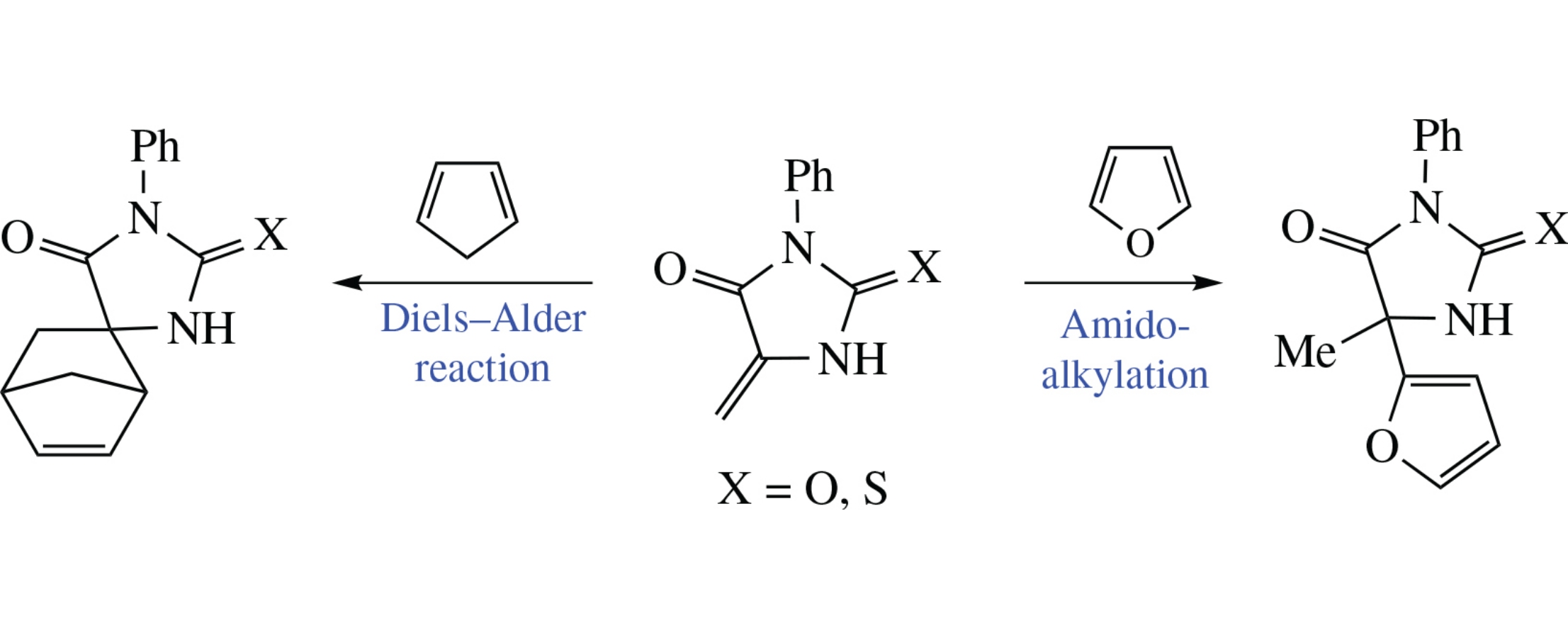
3-Methylidene-5-phenylhydantoin and 3-methylidene-5-phenylthiohydantoin react with cyclopentadiene forming spirocyclic Diels–Alder adducts. In contrast, their reactions with furan in the presence of AlCl3 give the products of the furan α-amidoalkylation.
References
1.

Wessels F.L., Schwan T.J., Pong S.F.
Journal of Pharmaceutical Sciences,
1980
2.

Meusel M., Gütschow M.
Organic Preparations and Procedures International,
2004
3.

Sondhi S.M., Singh J., Kumar A., Jamal H., Gupta P.P.
European Journal of Medicinal Chemistry,
2009
4.

Teno N., Gohda K., Wanaka K., Tsuda Y., Sueda T., Yamashita Y., Otsubo T.
Bioorganic and Medicinal Chemistry,
2014
5.

Lamothe M., Lannuzel M., Perez M.
Journal of Combinatorial Chemistry,
2001
6.

Handzlik J., Szymańska E., Chevalier J., Otrębska E., Kieć-Kononowicz K., Pagès J., Alibert S.
European Journal of Medicinal Chemistry,
2011
7.

Meyers M.J., Anderson E.J., McNitt S.A., Krenning T.M., Singh M., Xu J., Zeng W., Qin L., Xu W., Zhao S., Qin L., Eickhoff C.S., Oliva J., Campbell M.A., Arnett S.D., et. al.
Bioorganic and Medicinal Chemistry,
2015
8.

Ivanenkov Y.A., Vasilevski S.V., Beloglazkina E.K., Kukushkin M.E., Machulkin A.E., Veselov M.S., Chufarova N.V., Chernyaginab E.S., Vanzcool A.S., Zyk N.V., Skvortsov D.A., Khutornenko A.A., Rusanov A.L., Tonevitsky A.G., Dontsova O.A., et. al.
Bioorganic and Medicinal Chemistry Letters,
2015
9.

Beloglazkina A.А., Skvortsov D.А., Tafeenko V.A., Majouga А.G., Zyk N.V., Beloglazkina Е.К.
Russian Chemical Bulletin,
2018
10.

Beloglazkina A.A., Karpov N.A., Mefedova S.R., Polyakov V.S., Skvortsov D.A., Kalinina M.A., Tafeenko V.A., Majouga A.G., Zyk N.V., Beloglazkina E.K.
Russian Chemical Bulletin,
2019
11.

Kukushkin M.E., Skvortsov D.A., Kalinina M.A., Tafeenko V.A., Burmistrov V.V., Butov G.M., Zyk N.V., Majouga A.G., Beloglazkina E.K.
Phosphorus, Sulfur and Silicon and the Related Elements,
2020
12.

Beloglazkina A., Barashkin A., Polyakov V., Kotovsky G., Karpov N., Mefedova S., Zagribelny B., Ivanenkov Y., Kalinina M., Skvortsov D., Tafeenko V., Zyk N., Majouga A., Beloglazkina E.
Chemistry of Heterocyclic Compounds,
2020
13.

ABE N., FUJISAKI F., SUMOTO K.
Chemical and Pharmaceutical Bulletin,
2011
14.

Sumoto K., Fujisaki F., Shoji K.
Heterocycles,
2009
15.

Cernak T.A., Gleason J.L.
Journal of Organic Chemistry,
2007
16.

de la Hoz A., Dı́az-Ortiz A., Victoria Gómez M., Antonio Mayoral J., Moreno A., Sánchez-Migallón A.M., Vázquez E.
Tetrahedron,
2001