Keywords
acylation
CH-acidic compounds
diacylation
phosphonates
relative DpKa value
Abstract
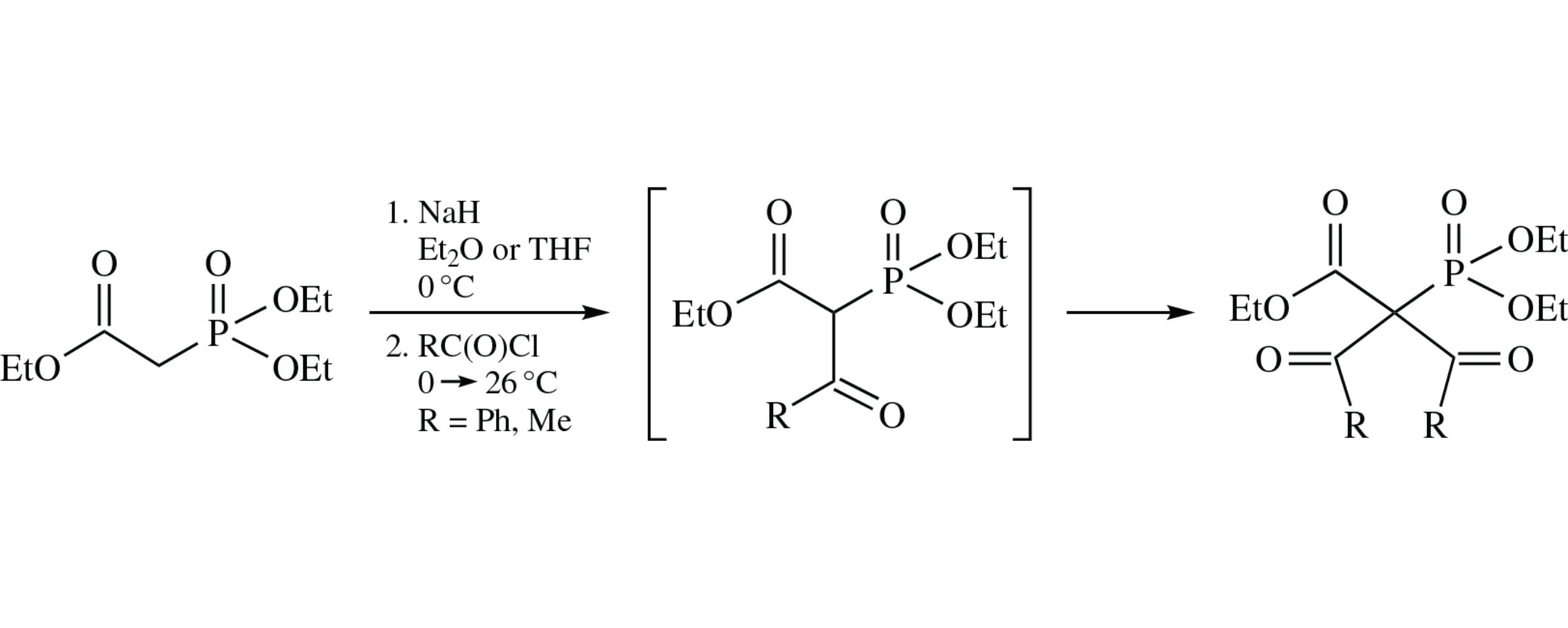
Deprotonation of diethyl (ethoxycarbonylmethyl)-phosphonate with NaH followed by treatment with acyl chlorides affords surprisingly the corresponding C,C-diacylated derivatives as the predominating products.
References
1.

Grün A., Bálint E., Keglevich G.
Catalysts,
2015
2.

Keglevich G., Novák T., Vida L., Greiner I.
Green Chemistry,
2006
3.

Sum F., Weiler L.
Canadian Journal of Chemistry,
1979
4.

Li C., Tao Z., Zhou Z., Bao X., Zhou S., Zou J.
Journal of Organic Chemistry,
2019
5.

Corbel B., L'Hostis-Kervella I., Haelters J.
Synthetic Communications,
1996
6.

Gough S.T., Trippett S.
Journal of the Chemical Society (Resumed),
1962
7.
10.1016/j.mencom.2021.03.033_bib0035
Cahiez
e-EROS Encyclopedia of Reagents for Organic Synthesis,
2001
8.

Durrant G., Sutherland J.K.
Journal of the Chemical Society Perkin Transactions 1,
1972
9.
10.1016/j.mencom.2021.03.033_bib0045
Sakhibullina
Zh. Obshch. Khim.,
1982
10.

SPEZIALE A.J., FREEMAN R.C.
Journal of Organic Chemistry,
1958
11.
![Synthesis of α-fluorinated phosphonoacetate derivatives using electrophilic fluorine reagents: Perchloryl fluoride versus 1-chloromethyl-4-fluoro-1,4-diazoniabicyclo[2.2.2]octane bis(tetrafluoroborate) (Selectfluor®)](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Marma M.S., Khawli L.A., Harutunian V., Kashemirov B.A., McKenna C.E.
Journal of Fluorine Chemistry,
2005
12.
10.1016/j.mencom.2021.03.033_bib0060
Frisch
Gaussian 16, Revision C. 01,
2016