Keywords
15d-prostaglandin J2
cyclopropanation
macrolactonization
silyl migration
Wittig reaction
Abstract
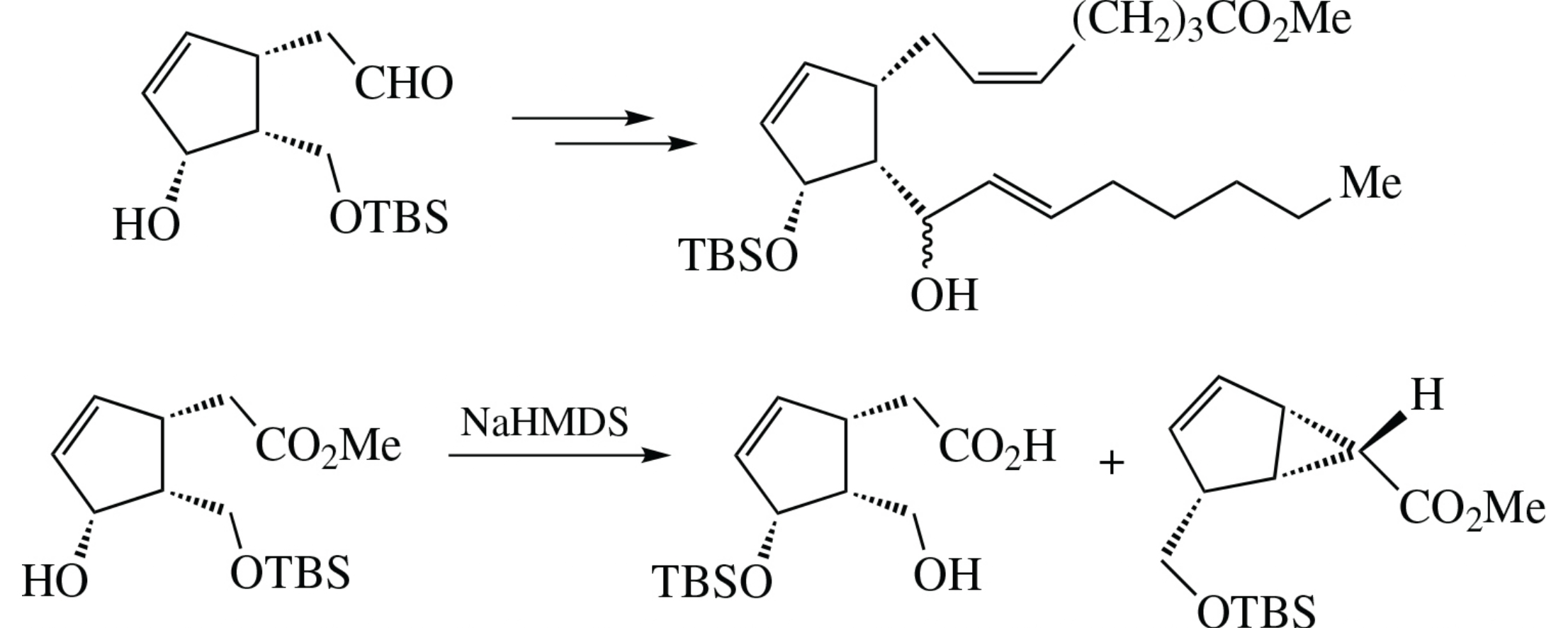
The Wittig reaction of {(1S,4R,5R)-5-[(tert-butyldimethylsilyloxy) methyl]-4-hydroxycyclopent-2-en-1-yl}acetaldehyde is accompanied by the migration of silyl protective group from the primary hydroxy group to the secondary one, which enables further synthesis of J-type prostaglandins. Study of this phenomemon on model relative ester comprising base variation revealed formation of unusual functionalized bicyclo[3.1.0]hex-2-ene-6-carboxylate derivative.
References
1.

Rouzer C.A., Marnett L.J.
Chemical Reviews,
2003
2.

Fitzpatrick F.A., Wynalda M.A.
Journal of Biological Chemistry,
1983
3.

Lefils-Lacourtablaise J., Socorro M., Géloën A., Daira P., Debard C., Loizon E., Guichardant M., Dominguez Z., Vidal H., Lagarde M., Bernoud-Hubac N.
PLoS ONE,
2013
4.

Straus D.S., Glass C.K.
Medicinal Research Reviews,
2001
5.

Brunoldi E.M., Zanoni G., Vidari G., Sasi S., Freeman M.L., Milne G.L., Morrow J.D.
Chemical Research in Toxicology,
2007
6.

Uchida K., Shibata T.
Chemical Research in Toxicology,
2007
7.

Pande V., Ramos M.J.
Bioorganic and Medicinal Chemistry Letters,
2005
8.

Kudva A.K., Kaushal N., Mohinta S., Kennett M.J., August A., Paulson R.F., Prabhu K.S.
PLoS ONE,
2013
9.

Loza V.V., Gimazetdinov A.M., Miftakhov M.S.
Russian Journal of Organic Chemistry,
2018
10.

Li J., Ahmed T.S., Xu C., Stoltz B.M., Grubbs R.H.
Journal of the American Chemical Society,
2018
11.

Li J., Stoltz B.M., Grubbs R.H.
Organic Letters,
2019
12.

Nicolaou K.C., Pulukuri K.K., Rigol S., Peitsinis Z., Yu R., Kishigami S., Cen N., Aujay M., Sandoval J., Zepeda N., Gavrilyuk J.
Journal of Organic Chemistry,
2018
13.
B. K. Goering, PhD Thesis, Cornell University, 1995.
14.
![A simple and efficient synthesis of enantiomeric (3aRS,4RS,6aSR)-4-hydroxy-3,3a,4,6a-tetrahydro-1H-cyclopenta[c]furan-1-ones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Gimazetdinov A.M., Gataullin S.S., Bushmarinov I.S., Miftakhov M.S.
Tetrahedron,
2012
15.
![Synthetic Approaches to 15-Deoxy-Δ12,14-prostaglandin J2. A New Key Building Block Based on (3aR,6R,6aS)-6-Trimethylsilyl)-3,3a,6,6a-tetrahydro-1H-cyclopenta[c]furan-1-one](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Gimazetdinov A.M., Al’mukhametov A.Z., Miftakhov M.S.
Russian Journal of Organic Chemistry,
2019
16.
Al’mukhametov A.Z., Gimazetdinov A.M., Miftakhov M.S.
Mendeleev Communications,
2020
17.
Al’mukhametov A.Z., Gimazetdinov A.M., Miftakhov M.S.
Mendeleev Communications,
2018
18.
Enantiopure vicinally trisubstituted all-cis-bis(hydroxymethyl)-cyclopentenols and their derivatives
Gimazetdinov A.M., Almukhametov A.Z., Loza V.V., Spirikhin L.V., Miftakhov M.S.
Mendeleev Communications,
2018
19.
10.1016/j.mencom.2021.03.031_bib0095
Greene
Greene's Protective Groups in Organic Synthesis,
2007
20.

Torisawa Y., Shibasaki M., Ikegami S.
Tetrahedron Letters,
1979
21.
10.1016/j.mencom.2021.03.031_bib0105
Jones
J. Chem. Soc., Perkin Trans.,
1979
22.

Sullivan D.F., Woodbury R.P., Rathke M.W.
Journal of Organic Chemistry,
1977